Advanced Pd-Catalyzed Synthesis of 2-Methyl-2,3-Diarylpropionaldehyde Derivatives for Commercial Scale
Advanced Pd-Catalyzed Synthesis of 2-Methyl-2,3-Diarylpropionaldehyde Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex molecular architectures, particularly those serving as critical building blocks for active pharmaceutical ingredients. Patent CN109438205B introduces a groundbreaking synthesis method for 2-methyl-2,3-diaryl propionaldehyde derivatives, a structural motif frequently encountered in bioactive natural products and medicinal chemistry campaigns. This innovation leverages a palladium-catalyzed C(sp3)-H arylation strategy that utilizes inexpensive amino acids as transient directing groups, effectively overcoming the limitations of traditional cross-coupling reactions which often rely on pre-functionalized substrates or prohibitively expensive ligands. By enabling the direct functionalization of unactivated C-H bonds adjacent to the carbonyl group, this technology offers a streamlined pathway to high-value intermediates with exceptional atom economy and operational simplicity.
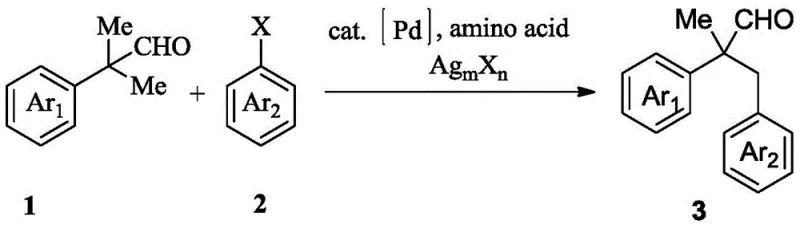
The core of this technological breakthrough lies in its ability to merge high efficiency with economic viability, addressing the dual pressures of R&D speed and manufacturing cost that define the modern chemical supply chain. The process operates under relatively mild thermal conditions compared to extreme high-pressure alternatives, utilizing a binary solvent system of acetic acid and hexafluoroisopropanol to stabilize the catalytic cycle and enhance solubility of diverse organic substrates. For procurement managers and supply chain directors, the implication is profound: a reliable pharmaceutical intermediate supplier can now offer these complex aldehydes with reduced lead times and lower raw material input costs, as the method eliminates the need for multi-step pre-functionalization sequences. Furthermore, the broad substrate tolerance described in the patent ensures that this platform technology can be adapted for the commercial scale-up of complex pharmaceutical intermediates without extensive re-optimization for each new derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-methyl-2,3-diarylpropanal skeletons has relied heavily on alpha-benzylation reactions of acetaldehyde derivatives or palladium-catalyzed couplings that necessitate the use of sophisticated, chiral phosphine ligands. A significant bottleneck in these conventional approaches is the reliance on binaphthyl-type phosphine ligands, which are not only synthetically challenging to produce but also command exorbitant market prices, thereby inflating the overall cost of goods sold for the final API intermediate. Additionally, earlier attempts to utilize amino acids as catalysts for similar transformations suffered from critically poor kinetics, with reported reaction times extending beyond 140 hours to achieve acceptable conversion levels. Such prolonged processing times are commercially untenable, as they tie up reactor capacity, increase energy consumption, and introduce significant risks of product degradation or side-reaction formation over extended heating periods. These factors collectively render traditional methods inefficient for large-scale manufacturing, creating a supply chain vulnerability for companies dependent on these specific chemical scaffolds.
The Novel Approach
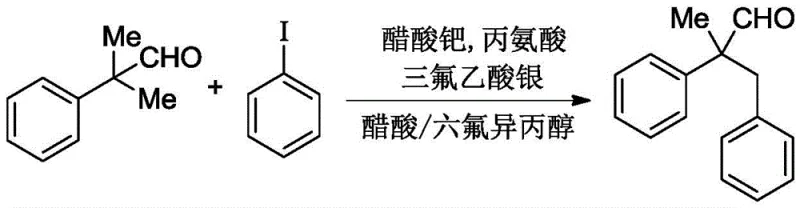
In stark contrast to these legacy techniques, the novel approach detailed in the patent employs a divalent palladium catalyst in conjunction with a transient directing group strategy that dramatically accelerates the reaction kinetics while maintaining high selectivity. By optimizing the reaction parameters, specifically the choice of silver salts and the solvent ratio of acetic acid to hexafluoroisopropanol, the process achieves completion within a practical window of 10 to 15 hours at temperatures between 120°C and 130°C. This represents a paradigm shift in process efficiency, reducing the reactor occupancy time by an order of magnitude compared to previous amino acid-catalyzed methods. The use of cheap, commercially available amino acids like alanine replaces the need for custom-synthesized phosphine ligands, effectively decoupling the reaction performance from volatile specialty chemical markets. This strategic pivot not only simplifies the operational workflow but also ensures cost reduction in pharmaceutical intermediate manufacturing by minimizing the expense associated with catalytic systems and purification steps.
Mechanistic Insights into Pd-Catalyzed C-H Arylation with Transient Directing Groups
The mechanistic elegance of this transformation relies on the in situ formation of an imine intermediate between the aldehyde substrate and the amino acid, which serves as a transient directing group to coordinate the palladium center. This coordination brings the metal catalyst into close proximity with the specific beta-C-H bond targeted for activation, facilitating a concerted metalation-deprotonation (CMD) process that cleaves the inert carbon-hydrogen bond with high regioselectivity. Once the C-Pd bond is formed, the catalytic cycle proceeds through oxidative addition of the aryl halide, followed by reductive elimination to forge the new carbon-carbon bond and release the desired 2-methyl-2,3-diarylpropionaldehyde product. The hydrolysis of the imine linkage regenerates the free amino acid, allowing it to participate in subsequent catalytic cycles, thereby functioning truly as a transient mediator rather than a stoichiometric reagent. This mechanism effectively bypasses the need for pre-installed directing groups that would otherwise require additional synthetic steps to install and remove, streamlining the overall synthetic route.
From an impurity control perspective, the transient directing group strategy offers superior selectivity profiles compared to non-directed radical pathways which often lead to mixtures of regioisomers. The rigid coordination geometry imposed by the amino acid-palladium complex ensures that arylation occurs exclusively at the intended position, minimizing the formation of ortho- or gamma-arylated byproducts that are difficult to separate via standard chromatography. Furthermore, the inclusion of silver salts, such as silver trifluoroacetate, plays a crucial role in scavenging halide ions generated during the oxidative addition step, preventing catalyst poisoning and maintaining high turnover numbers throughout the reaction duration. For R&D directors focused on purity and杂质谱 (impurity profiles), this level of mechanistic control translates to cleaner crude reaction mixtures and higher isolated yields, reducing the burden on downstream purification units. The robustness of this catalytic system across various electronic environments on the aryl halide partner further underscores its utility as a general-purpose tool for late-stage functionalization in drug discovery pipelines.
How to Synthesize 2-Methyl-2,3-Diarylpropionaldehyde Efficiently
The implementation of this synthesis protocol requires careful attention to the molar ratios of reagents and the specific sequence of addition to maximize catalyst longevity and reaction homogeneity. The patent outlines a generalized procedure where the aldehyde substrate, aryl halide, palladium source, amino acid, and silver oxidant are combined in a pressure-resistant vessel under an inert atmosphere to prevent oxidative degradation of sensitive intermediates. The choice of solvent mixture is critical, with hexafluoroisopropanol acting as a hydrogen-bond donor to stabilize charged transition states while acetic acid provides the necessary acidic medium for imine formation. Following the reaction period, standard workup procedures involving solvent evaporation and silica gel column chromatography are sufficient to isolate the target molecules in high purity, demonstrating the practical feasibility of this method for both laboratory and pilot plant settings.
- Dissolve 2-methyl-2-arylpropionaldehyde, aryl halide, palladium catalyst, amino acid, and silver salt in a mixed solvent of acetic acid and hexafluoroisopropanol.
- Stir the reaction mixture at room temperature for 20-40 minutes to ensure complete dissolution and initial complexation.
- Heat the mixture to 120-130°C and react for 10-15 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend far beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the raw material portfolio; by replacing expensive, specialized ligands with commodity amino acids, manufacturers can insulate their production costs from the volatility of the fine chemical market. This shift towards abundant, bio-based catalysts ensures a more stable pricing structure for the final intermediates, facilitating long-term budget planning and contract negotiations with downstream API producers. Moreover, the reduction in reaction time from days to less than a single shift significantly enhances asset utilization rates, allowing existing manufacturing infrastructure to produce greater volumes without capital expenditure on new reactors. This increased throughput capability directly addresses the challenge of reducing lead time for high-purity pharmaceutical intermediates, ensuring that critical supply chains remain resilient against demand surges.
- Cost Reduction in Manufacturing: The elimination of costly binaphthyl phosphine ligands results in substantial cost savings per kilogram of product, as the catalytic system now relies on widely available agricultural or fermentation-derived amino acids. Additionally, the shortened reaction cycle reduces energy consumption for heating and stirring, contributing to a lower overall carbon footprint and utility cost profile for the manufacturing site. The simplified purification process, driven by higher selectivity, further lowers the consumption of chromatography media and solvents, compounding the economic advantages throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Sourcing risks are mitigated because the key reagents, such as palladium acetate and alanine, are standard catalog items with multiple global suppliers, preventing single-source bottlenecks. The robustness of the reaction conditions means that minor variations in raw material quality do not catastrophicly impact the outcome, providing a buffer against supply chain disruptions. This reliability is crucial for maintaining continuous production schedules and meeting just-in-time delivery commitments to major pharmaceutical clients who demand strict adherence to timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in pressure tubes that mimic the conditions of larger autoclaves, ensuring a smooth transition from gram-scale R&D to ton-scale commercial production. The use of acetic acid and hexafluoroisopropanol, while requiring appropriate recovery systems, avoids the generation of heavy metal waste streams associated with stoichiometric organometallic reagents. This alignment with green chemistry principles facilitates easier regulatory approval and environmental permitting, accelerating the time-to-market for new drug candidates utilizing this synthetic route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H arylation technology, derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the transient directing group strategy over traditional cross-coupling methods.
Q: What is the primary advantage of using amino acids as transient directing groups in this synthesis?
A: Using amino acids as transient directing groups significantly reduces raw material costs compared to expensive binaphthyl-type phosphine ligands, while also simplifying the reaction operation and improving economic efficiency.
Q: How does this method improve upon previous C-H arylation strategies regarding reaction time?
A: Unlike prior art methods utilizing amino acid catalysts which required reaction times exceeding 140 hours, this optimized protocol achieves high conversion within 10 to 15 hours, making it viable for industrial production.
Q: What types of substrates are compatible with this palladium-catalyzed arylation process?
A: The method demonstrates a broad substrate scope, accommodating various aryl halides including those with electron-withdrawing or electron-donating groups, as well as different substitution patterns on the aldehyde starting material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-2,3-Diarylpropionaldehyde Supplier
As the global demand for complex pharmaceutical intermediates continues to rise, partnering with a CDMO expert capable of executing advanced C-H functionalization chemistries is a strategic imperative. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-methyl-2,3-diarylpropionaldehyde derivative meets the exacting standards required for GMP API synthesis. We understand that consistency and quality are non-negotiable in the pharmaceutical sector, and our technical team is dedicated to maintaining the highest levels of process control and documentation.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic platform for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development programs.
