Advanced Copper-Catalyzed Synthesis of 8-Acylquinoline Derivatives for Commercial Scale-Up
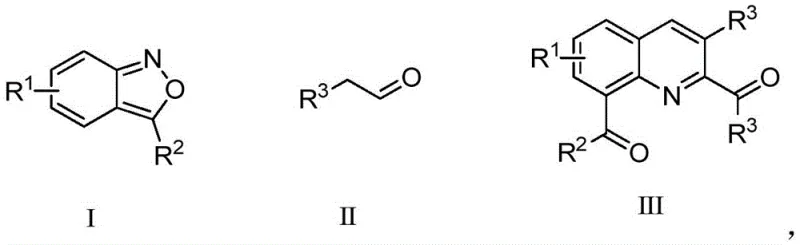
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for constructing complex heterocyclic scaffolds. Patent CN112500339A discloses a groundbreaking synthesis method for 8-acylquinoline derivatives, a core structural motif prevalent in numerous bioactive substances and synthetic drugs. This innovation addresses the critical need for efficient access to polysubstituted quinolines, which serve as pivotal intermediates for further modification into ligands and therapeutic agents. The disclosed methodology utilizes a copper-catalyzed coupling reaction between a 2,1-benzisoxazole derivative and a phenylacetaldehyde derivative under mild oxidative conditions. By leveraging inexpensive and readily available catalysts alongside a wide range of substrates, this technology offers a transformative approach to quinoline synthesis that aligns perfectly with modern green chemistry principles and commercial manufacturing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aldehyde-functionalized quinoline compounds has been fraught with significant technical and economic challenges that hinder large-scale production. Conventional strategies often rely on the oxidation of methyl or hydroxymethyl groups positioned on the quinoline ring, a process that typically necessitates the use of strong acids, stoichiometric amounts of hazardous oxidizing agents, or extremely toxic reagents. These harsh reaction conditions not only pose severe safety risks to operators but also generate substantial amounts of hazardous waste, leading to serious environmental pollution and increased disposal costs. Furthermore, existing methods for obtaining 8-hydroxyquinolines or related derivatives often suffer from limited functional group tolerance, restricting the diversity of molecules that can be synthesized. The inability to introduce sensitive substituents without degradation or side reactions severely limits the utility of these traditional pathways in the rapid development of new drug candidates and advanced materials.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a direct conversion strategy from aldehyde to quinoline aldehyde via a copper-catalyzed cyclization. This method employs a 2,1-benzisoxazole derivative and a phenylacetaldehyde derivative as substrates, reacting under the influence of a copper catalyst and a silver additive in an oxygen atmosphere. The process operates under significantly milder conditions, typically around 110°C in common organic solvents like toluene, eliminating the need for corrosive acids or toxic oxidants. The reaction exhibits high efficiency with yields reaching up to 70% in optimized examples, and it completes within a practical timeframe of roughly 12 hours. This shift represents a paradigm change in quinoline synthesis, offering a cleaner, safer, and more economically viable pathway that is inherently suitable for the rigorous demands of industrial pharmaceutical manufacturing.
Mechanistic Insights into Copper-Catalyzed Cyclization
The mechanistic underpinning of this transformation involves a sophisticated interplay between the copper catalyst, the silver additive, and molecular oxygen. The reaction likely proceeds through a radical-mediated pathway or a C-H activation mechanism where the copper species facilitates the cleavage of the weak N-O bond in the benzisoxazole ring. This ring-opening event generates a reactive nitrile oxide or similar intermediate in situ, which subsequently undergoes cycloaddition or insertion with the phenylacetaldehyde component. The presence of oxygen is critical, serving as the terminal oxidant to regenerate the active copper species and drive the catalytic cycle forward, thereby ensuring high atom economy. The silver additive, such as silver triflate, plays a crucial role in activating the catalyst or stabilizing intermediates, enhancing the overall reaction rate and selectivity. This synergistic catalytic system allows for the construction of the quinoline core with precise regiocontrol, specifically installing the acyl group at the 8-position, which is notoriously difficult to access via electrophilic substitution due to steric hindrance.
From an impurity control perspective, the mildness of this catalytic system is a distinct advantage for producing high-purity intermediates. Traditional oxidation methods often lead to over-oxidation of the aldehyde group to carboxylic acids or degradation of sensitive aromatic rings, creating complex impurity profiles that are difficult to separate. In this copper-catalyzed protocol, the reaction conditions are sufficiently gentle to preserve delicate functional groups such as halogens (fluorine, chlorine, bromine) and alkoxy groups, as evidenced by the successful synthesis of various substituted derivatives in the patent examples. The use of standard silica gel column chromatography for purification further indicates that the crude reaction mixture contains manageable levels of byproducts, facilitating the isolation of the target 8-acylquinoline derivative with high purity. This clean reaction profile minimizes the need for extensive downstream processing, directly contributing to cost reduction and improved process reliability.
How to Synthesize 8-Acylquinoline Derivatives Efficiently
The practical execution of this synthesis is designed for straightforward implementation in both laboratory and pilot plant settings. The general procedure involves charging a reaction vessel, such as a Schlenk tube, with the 2,1-benzisoxazole substrate, the phenylacetaldehyde derivative, copper powder (or other copper salts), a silver additive, and water. The mixture is dissolved in an organic solvent like toluene, with a typical solvent-to-substrate ratio of roughly 6-14 mL/mmol. A critical operational step is the replacement of the atmosphere with oxygen, usually performed by purging the vessel three times under reduced pressure to ensure an adequate supply of the oxidant. The reaction is then heated to approximately 110°C and stirred for about 12 hours. Upon completion, the solvent is removed under reduced pressure, and the residue is purified via silica gel column chromatography using a petroleum ether and ethyl acetate gradient. For a comprehensive, step-by-step standardized operating procedure including specific molar ratios and safety precautions, please refer to the detailed guide below.
- Prepare the reaction mixture by dissolving 2,1-benzisoxazole derivative and phenylacetaldehyde derivative in an organic solvent such as toluene, adding copper catalyst and silver additive.
- Purge the reaction vessel with oxygen gas three times under reduced pressure to ensure an oxidative atmosphere essential for the catalytic cycle.
- Heat the mixture to 110°C for 12 hours, then purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
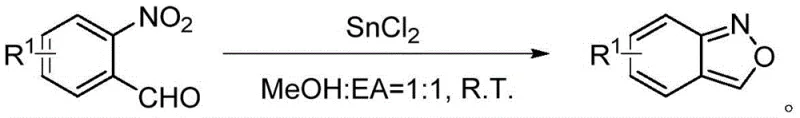
For procurement managers and supply chain directors, the adoption of this copper-catalyzed synthesis route offers compelling strategic advantages that extend beyond mere chemical novelty. The primary benefit lies in the drastic simplification of the supply chain for raw materials. The starting materials, 2,1-benzisoxazoles and phenylacetaldehydes, are commercially available or easily synthesized from commodity chemicals like o-nitrobenzaldehydes and alcohols, ensuring a stable and continuous supply source. Unlike methods requiring exotic reagents or precious metal catalysts like palladium or rhodium, this process relies on earth-abundant copper and silver salts, which are significantly cheaper and less subject to volatile market price fluctuations. This stability in raw material sourcing translates directly into predictable costing models and reduced risk of production delays caused by material shortages.
- Cost Reduction in Manufacturing: The economic impact of switching to this methodology is substantial, driven primarily by the elimination of expensive and hazardous reagents. By utilizing cheap copper powder or copper acetate instead of noble metals, the direct material cost of the catalyst system is minimized. Furthermore, the avoidance of strong acids and toxic oxidants reduces the overhead costs associated with specialized corrosion-resistant equipment and hazardous waste treatment facilities. The reaction's high efficiency and relatively short reaction time mean that reactor occupancy is optimized, allowing for higher throughput without the need for capital-intensive infrastructure upgrades. These factors combine to deliver a manufacturing process that is inherently leaner and more cost-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of the reaction conditions. The method tolerates a wide variety of substituents, meaning that a single platform technology can be used to produce a diverse library of 8-acylquinoline derivatives simply by swapping out the starting aldehyde or benzisoxazole. This modularity reduces the need for developing entirely new processes for each new analog, accelerating time-to-market for new drug candidates. Additionally, the use of common solvents like toluene and standard purification techniques ensures that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-validation efforts, securing the continuity of supply.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by its operational simplicity and green chemistry profile. The reaction does not generate excessive heat or pressure, reducing the engineering controls required for safe scale-up. From an environmental compliance standpoint, the use of oxygen as the terminal oxidant produces water as the primary byproduct, significantly lowering the E-factor (mass of waste per mass of product) compared to traditional stoichiometric oxidations. This alignment with increasingly stringent environmental regulations minimizes the risk of regulatory shutdowns and enhances the corporate sustainability profile, making the final product more attractive to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and scope of the method. Understanding these details is crucial for R&D teams evaluating the technology for pipeline integration and for procurement teams assessing the long-term viability of the supply chain. The responses cover aspects ranging from catalyst selection and optimization to the handling of specific substrate variations, ensuring a comprehensive understanding of the process capabilities.
Q: What are the advantages of this copper-catalyzed method over traditional oxidation routes?
A: Unlike traditional methods that require harsh oxidants or toxic reagents to convert methyl groups to aldehydes, this copper-catalyzed approach uses mild conditions, inexpensive catalysts, and readily available substrates, significantly reducing environmental impact and operational costs.
Q: Can this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate scope, tolerating various substituents including halogens (F, Cl, Br), alkoxy groups (OMe), and alkyl groups on both the benzisoxazole and phenylacetaldehyde components, making it highly versatile for medicinal chemistry applications.
Q: Is the purification process scalable for industrial production?
A: The purification involves standard silica gel column chromatography using common solvent systems like petroleum ether and ethyl acetate, which are easily scalable and cost-effective for large-scale manufacturing compared to complex crystallization or distillation processes required by other methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Acylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including oxygen atmosphere reactions and rigorous purification protocols. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 8-acylquinoline derivative meets the highest quality standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Together, we can optimize your supply chain and accelerate the delivery of life-saving medicines to the market.
