Advanced Peroxide-Free Synthesis of Beta-Iodoalkenyl Sulfones for Commercial Scale-Up
Introduction to Novel Beta-Iodoalkenyl Sulfone Synthesis
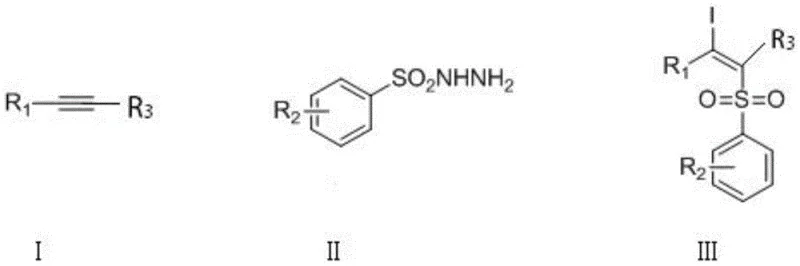
The development of efficient and safe methodologies for constructing functionalized olefin compounds remains a critical priority in modern organic synthesis, particularly for the pharmaceutical and agrochemical industries. Patent CN108997178B introduces a groundbreaking approach for the synthesis of β-iodoalkenyl sulfone compounds, addressing long-standing safety and efficiency challenges associated with traditional protocols. This technology leverages diiodine pentoxide ($I_2O_5$) as a dual-function reagent, serving simultaneously as the iodine source and the reaction initiator. By shifting away from hazardous peroxide-based oxidation systems, this method offers a robust pathway for generating high-purity intermediates essential for downstream drug discovery and material science applications.
The significance of this innovation lies in its ability to streamline the 1,2-difunctionalization of alkynes, a transformation that installs both iodine and sulfonyl groups in a single operational step. Functionalized olefins are pivotal building blocks, yet their synthesis often requires harsh conditions or toxic catalysts. The disclosed method operates under mild thermal conditions, typically between 60°C and 100°C, and utilizes readily available starting materials. For procurement managers and supply chain directors, this represents a shift towards more sustainable and manageable chemical processes that reduce regulatory burdens while maintaining high product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-iodoalkenyl sulfones has relied heavily on the addition of sodium sulfinates to alkynes, catalyzed by iodide salts and driven by strong organic peroxides such as tert-butyl hydroperoxide (TBHP) or di-tert-butyl peroxide (DTBP). While these methods are chemically feasible, they present substantial drawbacks for industrial scale-up. The use of strong peroxides introduces significant safety hazards due to their explosive potential and thermal instability, necessitating specialized handling equipment and rigorous safety protocols that inflate operational costs. Furthermore, peroxide-mediated reactions often generate complex impurity profiles, complicating the purification process and reducing the overall atom economy of the synthesis.
Additionally, conventional routes frequently suffer from limited substrate scope or require excessive amounts of oxidants to drive the reaction to completion. The reliance on separate iodine sources, such as elemental iodine or iodide salts, alongside distinct oxidants, adds complexity to the reagent sourcing and inventory management. For R&D teams, the presence of residual peroxides in the final product can be detrimental, requiring extensive quenching and washing steps that increase solvent consumption and waste generation. These factors collectively hinder the economic viability and environmental sustainability of traditional iodosulfonylation processes.
The Novel Approach
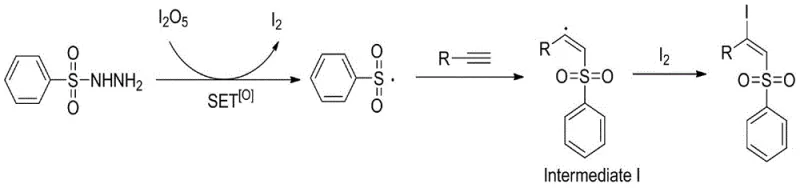
The methodology described in patent CN108997178B fundamentally reimagines the oxidative iodosulfonylation landscape by employing diiodine pentoxide as a singular, multifunctional reagent. This inorganic oxide acts as a mild yet effective oxidant that facilitates the generation of sulfonyl radicals from sulfonyl hydrazides without the need for dangerous organic peroxides. This substitution drastically improves the safety profile of the reaction, making it far more suitable for commercial manufacturing environments where risk mitigation is paramount. The reaction proceeds smoothly in common organic solvents, with 1,4-dioxane identified as the optimal medium, delivering exceptional yields that often surpass 90%.
Beyond safety, this novel approach offers superior operational simplicity. The dual role of $I_2O_5$ eliminates the need for balancing multiple oxidant and iodine source equivalents, simplifying the stoichiometry and reducing raw material costs. The mild reaction conditions preserve sensitive functional groups on the alkyne and sulfonyl hydrazide substrates, allowing for the synthesis of a diverse array of derivatives including those with halogen, alkoxy, and heteroaromatic substituents. This versatility ensures that the process can be adapted for the production of various high-value fine chemicals and pharmaceutical intermediates without extensive re-optimization.
Mechanistic Insights into Radical Iodosulfonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific targets. The reaction initiates through a single electron transfer (SET) process mediated by diiodine pentoxide under thermal conditions. The sulfonyl hydrazide undergoes oxidation, resulting in the extrusion of nitrogen gas and the formation of a highly reactive sulfonyl radical species. Concurrently, the diiodine pentoxide is reduced, releasing molecular iodine into the reaction matrix. This in situ generation of iodine is critical, as it serves as the trapping agent for the subsequent radical intermediate, ensuring high regioselectivity and stereoselectivity.
Once generated, the sulfonyl radical adds across the carbon-carbon triple bond of the alkyne substrate. Due to steric hindrance and electronic factors, this addition occurs in a trans-selective manner, forming a vinyl radical intermediate. This intermediate is then rapidly intercepted by the molecular iodine present in the solution, yielding the final β-iodoalkenyl sulfone product with high E-selectivity. This radical cascade mechanism avoids the formation of carbocation intermediates that might lead to rearrangement side products, thereby ensuring a clean impurity profile. The efficiency of this radical propagation cycle is a key driver of the high yields observed across a broad range of substrates.
How to Synthesize Beta-Iodoalkenyl Sulfones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal control to maximize efficiency. The standard protocol involves combining the alkyne derivative, the sulfonyl hydrazide derivative, and diiodine pentoxide in a reaction vessel equipped with a heating mantle and stirrer. The choice of solvent is pivotal, with 1,4-dioxane providing the best balance of solubility and reaction rate, although other polar aprotic solvents can be utilized if necessary. The reaction mixture is heated to approximately 80°C and maintained under stirring for a period ranging from 6 to 10 hours, depending on the specific electronic nature of the substrates involved.
- Mix alkyne derivatives, sulfonyl hydrazide derivatives, and diiodine pentoxide in a suitable solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 60°C and 100°C, preferably around 80°C, and stir for 6 to 10 hours.
- Upon completion, cool the mixture to room temperature and purify the crude product via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this peroxide-free synthesis method offers tangible strategic benefits beyond mere chemical curiosity. The elimination of hazardous organic peroxides from the supply chain significantly reduces the costs associated with special storage, transportation, and insurance compliance. Handling non-peroxide oxidants simplifies warehouse logistics and minimizes the risk of supply disruptions caused by strict regulations on explosive precursors. Furthermore, the high atom economy and simplified workup procedures translate directly into reduced solvent usage and lower waste disposal fees, contributing to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The use of diiodine pentoxide as a dual-purpose reagent streamlines the bill of materials by removing the need for separate iodine sources and expensive peroxide oxidants. This consolidation of reagents lowers the direct material cost per kilogram of product. Additionally, the high yields achieved, often exceeding 90%, mean that less raw material is wasted on side reactions or lost during purification. The simplified post-reaction processing, which avoids complex peroxide quenching steps, further reduces labor hours and utility consumption, driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: Sourcing stable inorganic oxidants like diiodine pentoxide is generally more reliable than procuring regulated organic peroxides, which are subject to fluctuating availability and stringent transport restrictions. By adopting a chemistry that relies on commercially abundant and stable reagents, manufacturers can secure a more resilient supply chain. This stability ensures consistent production schedules and reduces the likelihood of delays caused by regulatory hold-ups or supplier shortages, thereby enhancing the reliability of delivery to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of volatile, explosive peroxides make this process inherently safer to scale from gram to ton quantities. The reduced toxicity and environmental impact align with increasingly strict global environmental regulations, facilitating easier permitting for new production lines. The generation of nitrogen gas as a benign byproduct, rather than toxic organic waste, simplifies effluent treatment. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly valued by international partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What is the primary safety advantage of this synthesis method?
A: Unlike conventional methods that rely on hazardous organic peroxides like TBHP or DTBP as oxidants, this novel approach utilizes diiodine pentoxide. This eliminates the significant safety risks associated with handling strong peroxides, making the process safer and more environmentally friendly for large-scale manufacturing.
Q: What yields can be expected with this protocol?
A: The method demonstrates excellent substrate universality and high efficiency. Experimental data indicates that yields can reach up to 95% for optimized substrates, with many examples consistently achieving yields above 80% under mild reaction conditions.
Q: Is this method suitable for diverse chemical structures?
A: Yes, the reaction shows broad tolerance for various functional groups. It is effective for alkynes substituted with electron-withdrawing or electron-donating groups, as well as heteroaromatic systems, providing a versatile route for constructing complex functionalized olefin intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Iodoalkenyl Sulfones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation pharmaceuticals and fine chemicals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the peroxide-free iodosulfonylation described here can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-iodoalkenyl sulfones meets the exacting standards required for API intermediate manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel derivatives or large-scale supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to safety and efficiency can drive value for your organization.
