Advanced Gold-Catalyzed Synthesis of N-Sulfonyl Ketimines for Commercial Scale Production
Advanced Gold-Catalyzed Synthesis of N-Sulfonyl Ketimines for Commercial Scale Production
The chemical landscape for synthesizing complex nitrogen-containing heterocycles is constantly evolving, driven by the need for more efficient and environmentally benign processes in the fine chemical sector. A pivotal advancement in this domain is documented in patent CN101314581B, which discloses a robust methodology for the preparation of N-sulfonyl ketimine compounds. These compounds serve as critical building blocks in the synthesis of high-value active pharmaceutical ingredients (APIs) and agrochemical agents. The patented technology leverages a transition metal-catalyzed addition reaction between alkyne compounds and sulfonamides, offering a distinct departure from traditional synthetic routes that often suffer from harsh conditions and poor atom economy. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this gold-catalyzed pathway is essential for securing a stable supply of high-purity precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-sulfonyl ketimines has been plagued by significant operational challenges that hinder efficient commercial scale-up. Traditional methodologies often rely on the condensation of ketones with sulfenamides followed by oxidation, or the direct dehydration of sulfonamides and ketones under strong Lewis acid conditions. These legacy processes frequently require rigorous exclusion of moisture, utilize corrosive reagents, and generate substantial quantities of hazardous waste, complicating the cost reduction in pharmaceutical intermediate manufacturing. Furthermore, alternative routes involving palladium-catalyzed oxidative addition of ketones and styrenes can be prohibitively expensive due to the high cost of palladium and the complexity of removing trace metal residues from the final product. Such limitations not only impact the purity profile required for sensitive biological applications but also introduce variability in yield and reproducibility, creating bottlenecks for supply chain managers aiming for consistent output.
The Novel Approach
In stark contrast, the method outlined in CN101314581B introduces a streamlined catalytic cycle that directly couples terminal alkynes with sulfonamides. This innovative approach utilizes a gold-based catalyst system to activate the alkyne triple bond towards nucleophilic attack by the sulfonamide nitrogen. The reaction proceeds smoothly in inert organic solvents at moderate temperatures ranging from 35°C to 140°C, with a preferred operating window of 50°C to 120°C. This mild thermal profile significantly reduces energy consumption and minimizes the risk of thermal degradation of sensitive functional groups. By employing readily available starting materials and avoiding the multi-step sequences associated with older technologies, this novel route enhances the overall process mass intensity (PMI). The result is a cleaner reaction profile that simplifies downstream purification, thereby supporting the production of high-purity N-sulfonyl ketimines essential for downstream drug synthesis.
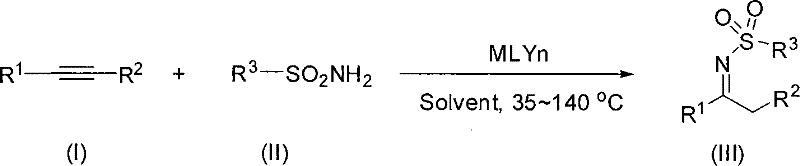
Mechanistic Insights into Gold-Catalyzed Hydroamination
The core of this technological breakthrough lies in the unique ability of cationic gold species to act as soft Lewis acids. In the catalytic cycle, the gold center, typically coordinated with phosphine ligands such as triphenylphosphine, activates the pi-system of the alkyne substrate. This activation increases the electrophilicity of the alkyne carbons, facilitating the nucleophilic attack by the nitrogen atom of the sulfonamide. The subsequent proton transfer and protodeauration steps regenerate the active gold catalyst, allowing the cycle to continue with high turnover numbers. This mechanism is particularly advantageous because it avoids the formation of unstable enol intermediates that often plague acid-catalyzed condensations. The use of a silver salt, such as silver trifluoromethanesulfonate, as a co-catalyst further enhances the reaction efficiency by abstracting the halide ligand from the gold precursor, generating the highly active cationic gold species in situ. This precise control over the catalytic species ensures that the reaction proceeds with high regioselectivity and minimal side reactions.
From an impurity control perspective, the atom-economic nature of this addition reaction is a major benefit. Unlike substitution reactions that generate stoichiometric amounts of leaving groups, this hydroamination process incorporates all atoms of the reactants into the final product structure, theoretically yielding 100% atom economy. In practice, this translates to a simplified impurity profile, primarily consisting of unreacted starting materials which are easily removed during workup. The absence of heavy metal oxidants or strong dehydrating agents means that the final product is less likely to contain difficult-to-remove inorganic salts or toxic organic by-products. For quality control teams, this implies a more straightforward analytical validation process and a reduced burden on purification resources, ultimately leading to a more robust and reliable manufacturing process for complex pharmaceutical intermediates.
How to Synthesize N-Sulfonyl Ketimines Efficiently
The practical implementation of this synthesis involves a straightforward procedure that balances reaction kinetics with operational simplicity. The process begins by combining the alkyne and sulfonamide substrates in a suitable solvent, with the gold catalyst and silver co-catalyst added in precise molar ratios to ensure optimal activity. The reaction mixture is then heated to the target temperature and monitored via thin-layer chromatography (TLC) to determine the endpoint, typically achieved within 2 to 10 hours. Upon completion, the reaction is quenched and subjected to a standard aqueous workup involving extraction and drying, followed by purification via column chromatography to isolate the pure N-sulfonyl ketimine. Detailed standardized synthetic steps for replicating this high-yield process are provided in the guide below.
- Mix alkyne compound, sulfonamide compound, (PPh3)AuCl catalyst, and silver trifluoromethanesulfonate co-catalyst in an organic solvent such as tetrahydrofuran or toluene.
- Heat the reaction mixture to a temperature between 50°C and 120°C and maintain stirring for 2 to 10 hours until TLC indicates completion.
- Cool the reaction to room temperature, quench with saturated sodium bicarbonate, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this gold-catalyzed technology offers tangible strategic benefits beyond mere chemical elegance. The shift towards this methodology addresses several critical pain points associated with the sourcing and production of fine chemical intermediates. By utilizing a catalyst system that operates effectively at low loadings (0.001 to 0.2 mol relative to substrate), the process mitigates the cost impact of using precious metals. Furthermore, the reliance on commercially available alkynes and sulfonamides ensures that raw material sourcing remains stable and unaffected by niche supply constraints. The mild reaction conditions also extend the lifespan of reactor equipment and reduce the safety risks associated with high-pressure or high-temperature operations, contributing to a safer and more sustainable manufacturing environment.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and simplified workup procedures. By eliminating the need for expensive oxidants, strong bases, or complex multi-step sequences, the overall cost of goods sold (COGS) is significantly optimized. The removal of transition metal catalysts, while requiring attention, is streamlined by the low loading levels and the use of standard filtration or extraction techniques, avoiding the need for specialized scavenging resins often required for palladium removal. This efficiency translates into substantial cost savings over the lifecycle of the product, making it a financially attractive option for large-scale production runs without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Because the reaction tolerates a range of temperatures and uses common organic solvents like tetrahydrofuran or toluene, it is less susceptible to disruptions caused by utility fluctuations or solvent shortages. The high yields reported in the patent examples, often exceeding 80% and reaching near-quantitative levels in optimized cases, ensure that material throughput is maximized. This reliability allows supply chain planners to forecast production volumes with greater confidence, reducing the need for excessive safety stock and minimizing the risk of stockouts for critical downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or tonne scale is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous gas evolution. The process aligns well with green chemistry principles, generating minimal waste and utilizing reagents that are easier to handle and dispose of compared to traditional alternatives. This environmental compatibility simplifies regulatory compliance and waste management logistics, which are increasingly critical factors for global supply chains. The ability to run the reaction in standard glass-lined or stainless steel reactors without specialized pressure vessels further lowers the barrier to entry for contract manufacturing organizations looking to expand their capacity for nitrogen-heterocycle intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the gold-catalyzed method over traditional Lewis acid methods?
A: The gold-catalyzed method described in patent CN101314581B operates under significantly milder conditions (50-120°C) compared to harsh Lewis acid protocols, resulting in higher product quality, better reproducibility, and reduced formation of toxic by-products.
Q: Is this synthesis method suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available raw materials and simple post-treatment steps like extraction and column chromatography, making it highly scalable for industrial production while maintaining atom economy and environmental safety.
Q: What specific catalyst system is required for this transformation?
A: The reaction employs a transition metal catalyst system comprising a gold cation complex, preferably (PPh3)AuCl, often used in conjunction with a silver trifluoromethanesulfonate co-catalyst to activate the alkyne substrate efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Sulfonyl Ketimine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN101314581B for the modern pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify identity and assay. Our capability to handle gold-catalyzed reactions safely and efficiently positions us as a preferred partner for clients demanding high-quality intermediates for complex drug synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic method for your specific target molecules. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your development timelines and production budgets. Let us collaborate to drive innovation and efficiency in your chemical supply chain.
