Scalable Hydroxylation of Aryl Halides Using Recyclable Copper Phthalocyanine Catalysts for Industrial Manufacturing
Scalable Hydroxylation of Aryl Halides Using Recyclable Copper Phthalocyanine Catalysts for Industrial Manufacturing
The chemical industry continuously seeks robust methodologies for constructing carbon-oxygen bonds, particularly for the synthesis of phenols and hydroxylated heteroaromatics which serve as critical building blocks in drug discovery and material science. A significant breakthrough in this domain is documented in Chinese patent CN114805031A, which discloses a highly efficient hydroxylation method for aryl and heteroaryl halides utilizing copper phthalocyanine and its derivatives as catalysts. This technology represents a paradigm shift from traditional homogeneous systems to a more sustainable heterogeneous catalytic regime, addressing long-standing challenges regarding catalyst recovery and operational costs. By leveraging the unique electronic properties of macrocyclic copper complexes, this invention enables the conversion of diverse halide substrates into valuable phenolic compounds under relatively mild thermal conditions. For procurement leaders and technical directors alike, understanding the nuances of this patent is essential for evaluating next-generation supply chain strategies for high-purity pharmaceutical intermediates.
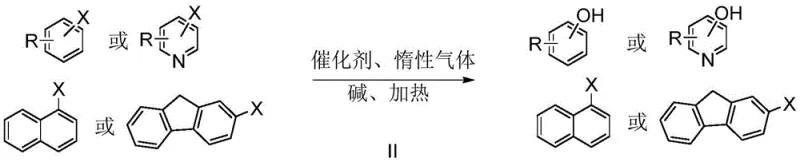
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroxylation of aryl halides has relied heavily on homogeneous copper catalytic systems that necessitate the use of specialized and often expensive ligands to stabilize the active metal species. Prominent literature, such as works by Tlili and Zhao, utilized ligands like 1,3-diketones or 1,10-phenanthroline to facilitate CuI-catalyzed coupling, yet these systems invariably suffer from high catalytic loadings and complex downstream processing. The homogeneous nature of these traditional catalysts makes separation from the final product arduous, often requiring extensive chromatography or extraction steps that generate significant chemical waste and increase production lead times. Furthermore, many existing heterogeneous copper catalysts, such as Cu-Zagronas or Cu3N nanoparticles reported in recent years, exhibit disappointingly low activity towards less reactive aryl bromides and chlorides, limiting their utility in broad-spectrum manufacturing. These technical bottlenecks translate directly into higher operational expenditures and reduced throughput for facilities aiming to produce cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast to the limitations of prior art, the methodology described in CN114805031A employs copper phthalocyanine derivatives, such as CuPcF16 and CuPcCl16, which function as highly active and stable heterogeneous catalysts. This innovative approach eliminates the need for external expensive ligands because the phthalocyanine macrocycle itself provides a robust coordination environment for the copper center, ensuring sustained catalytic turnover. The reaction proceeds efficiently in a mixed solvent system of organic polar solvents like DMSO or DMF with water, utilizing common inorganic bases such as sodium hydroxide or potassium carbonate to drive the nucleophilic substitution. Crucially, the heterogeneous nature of the copper phthalocyanine catalyst allows for straightforward recovery via simple filtration after the reaction concludes, enabling multiple reuse cycles without significant loss of activity. This technological advancement offers a reliable pharmaceutical intermediates supplier pathway that drastically simplifies workup procedures and enhances overall process sustainability.
Mechanistic Insights into Copper Phthalocyanine-Catalyzed Hydroxylation
The catalytic cycle likely involves the activation of the aryl halide bond by the copper center within the phthalocyanine framework, facilitating oxidative addition or a radical-mediated pathway depending on the specific substrate electronics. The electron-withdrawing or donating groups on the phthalocyanine periphery, such as fluorine or chlorine atoms in CuPcF16 or CuPcCl16, modulate the Lewis acidity of the central copper ion, thereby tuning its reactivity towards different halide leaving groups. In the presence of a strong base, the hydroxide ion acts as the nucleophile, attacking the activated aryl-copper intermediate to form the carbon-oxygen bond while regenerating the catalyst species. The stability of the phthalocyanine ring system prevents the leaching of copper into the solution, which is a common failure mode in other heterogeneous systems that leads to product contamination and catalyst deactivation. This mechanistic robustness ensures that the impurity profile of the resulting phenols remains clean, a critical factor for meeting stringent purity specifications in API synthesis.
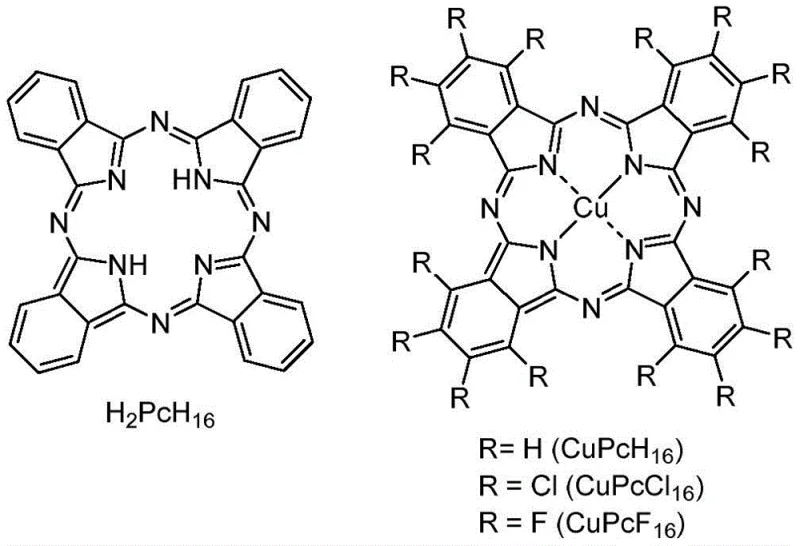
Furthermore, the tolerance of this catalytic system towards various functional groups is attributed to the mild reaction conditions and the specific steric environment provided by the planar macrocyclic ligand. Unlike aggressive homogeneous conditions that might degrade sensitive moieties like ketones or nitro groups, this heterogeneous protocol maintains chemoselectivity, allowing for the hydroxylation of halides bearing competing electrophilic sites. The use of water as a co-solvent not only aids in the solubility of the inorganic base but also participates in the proton transfer steps necessary for product release, enhancing the green chemistry profile of the transformation. Detailed analysis of the reaction kinetics suggests that the rate-determining step may involve the diffusion of the substrate to the catalyst surface or the initial activation of the carbon-halogen bond, which is accelerated by the elevated temperatures ranging from 90°C to 150°C. Understanding these mechanistic details empowers R&D teams to optimize reaction parameters for commercial scale-up of complex polymer additives or electronic chemicals.
How to Synthesize Hydroxylated Aryl Compounds Efficiently
Implementing this technology requires careful attention to the molar ratios of reactants and the selection of the appropriate solvent system to maximize yield and catalyst longevity. The patent outlines a generalized procedure where the aryl halide, catalyst, and base are combined in a reactor purged with inert gas to prevent oxidative degradation of sensitive intermediates. Following the addition of the solvent mixture, typically DMSO and water in a ratio such as 1:1, the reaction is heated to the specified temperature for a duration of 3 to 10 hours depending on the substrate reactivity. Upon completion, the mixture is cooled, acidified to neutralize excess base, and extracted with an organic solvent like ethyl acetate to isolate the crude phenolic product. The detailed standardized synthesis steps see the guide below for specific operational parameters tailored to different substrate classes.
- Combine aryl halide substrate, copper phthalocyanine catalyst (e.g., CuPcF16), and inorganic base in a reaction vessel under inert atmosphere.
- Add a mixed solvent system comprising a polar aprotic organic solvent like DMSO and water, then heat the mixture to 90-150°C.
- After completion, cool the reaction, extract with organic solvent, and recover the heterogeneous catalyst via filtration for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper phthalocyanine catalyzed hydroxylation method presents tangible strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which removes the dependency on scarce and costly homogeneous ligands that often plague traditional cross-coupling supply chains. By transitioning to a heterogeneous catalyst that can be filtered and reused, facilities can significantly reduce the consumption of raw materials and minimize the generation of heavy metal waste, aligning with increasingly strict environmental compliance regulations. This operational efficiency translates into a more resilient supply chain capable of sustaining high-volume production without the volatility associated with specialized reagent sourcing. Moreover, the broad substrate scope ensures that a single catalytic platform can service multiple product lines, reducing the need for diverse inventory management and specialized equipment configurations.
- Cost Reduction in Manufacturing: The elimination of expensive ligand systems and the ability to recycle the copper phthalocyanine catalyst multiple times leads to substantial cost savings in raw material expenditure over the lifecycle of the process. Traditional methods often require stoichiometric or near-stoichiometric amounts of additives that are lost during workup, whereas this novel approach retains the catalytic species for subsequent batches, effectively lowering the cost per kilogram of the final product. Additionally, the use of inexpensive inorganic bases like sodium hydroxide instead of bulky organic bases further drives down the bill of materials, making the process economically attractive for large-scale commodity chemical production. These cumulative savings allow manufacturers to offer more competitive pricing for high-purity OLED material or agrochemical intermediate clients without compromising on quality margins.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable catalyst precursors mitigates the risk of supply disruptions that are common with custom-synthesized homogeneous ligands. The robustness of the reaction conditions, which tolerate the presence of water and operate at moderate temperatures, ensures consistent batch-to-batch reproducibility even in facilities with varying levels of infrastructure sophistication. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the occurrence of failed batches and the need for reprocessing. Consequently, supply chain planners can forecast production timelines with greater accuracy, ensuring timely delivery to downstream customers who depend on just-in-time inventory models for their own manufacturing schedules.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst facilitates seamless scale-up from laboratory gram-scale experiments to multi-ton industrial reactors without the mass transfer limitations often encountered in homogeneous systems. The simplified workup procedure, which avoids complex chromatographic separations in favor of filtration and extraction, reduces the volume of organic solvents required, thereby lowering the environmental footprint and waste disposal costs. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly a deciding factor for global partners selecting a reliable fine chemical intermediates supplier. The ability to handle diverse substrates including chlorides and bromides further expands the addressable market, allowing for the flexible production of various derivative portfolios.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this hydroxylation technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational boundaries and potential applications for technical teams evaluating this process for integration into their existing manufacturing pipelines. Understanding these specifics is vital for assessing the feasibility of adapting this method to proprietary compound libraries and ensuring alignment with internal quality standards.
Q: Can the copper phthalocyanine catalyst be recycled?
A: Yes, the patent demonstrates that the heterogeneous copper phthalocyanine catalyst can be recovered by simple filtration and drying, maintaining high activity even after six consecutive reuse cycles.
Q: What types of halides are compatible with this hydroxylation method?
A: The method exhibits broad substrate adaptability, successfully converting aryl iodides, bromides, and even some chlorides, as well as heteroaryl halides like pyridines, into their corresponding hydroxylated products.
Q: Does this process require expensive ligands?
A: No, unlike conventional homogeneous copper systems that require costly ligands like 1,10-phenanthroline, this novel approach utilizes stable copper phthalocyanine derivatives which act as both catalyst and ligand framework.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Copper Phthalocyanine Derivative Supplier
As the demand for efficient and sustainable synthetic routes grows, NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in handling sensitive catalytic systems and optimizing reaction parameters to meet stringent purity specifications required by top-tier global pharmaceutical and electronic material companies. We maintain rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of copper phthalocyanine catalyst or hydroxylated intermediate meets the highest standards of quality and consistency. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and commercial reliability, ensuring your projects move from concept to market without interruption.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and target molecules. Our experts are prepared to provide specific COA data and route feasibility assessments to demonstrate how this patented hydroxylation technology can be integrated into your current operations. Let us collaborate to unlock the full potential of this innovative catalytic system, driving down costs and accelerating your time to market for critical chemical intermediates.
