Advanced Synthesis of 3-Fluoro-4-Trifluoromethyl Benzonitrile for Commercial Pharmaceutical Applications
Advanced Synthesis of 3-Fluoro-4-Trifluoromethyl Benzonitrile for Commercial Pharmaceutical Applications
The global demand for fluorinated aromatic nitriles has surged due to their critical role as building blocks in modern medicinal chemistry, particularly for kinase inhibitors and agrochemical active ingredients. Patent CN112979498A, published in mid-2021, introduces a robust and scalable preparation method for 3-fluoro-4-trifluoromethyl benzonitrile that addresses significant bottlenecks found in legacy synthetic routes. This technical breakthrough utilizes a strategic two-step sequence starting from 2-chloro-3-fluoro-4-trifluoromethylbenzaldehyde, leveraging oximation followed by catalytic hydrogenolysis to achieve superior outcomes. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology represents a paradigm shift towards greener, more efficient manufacturing. By avoiding highly toxic reagents like cuprous cyanide and complex diazotization procedures, the process not only enhances operator safety but also streamlines the purification workflow, resulting in a final product with exceptional quality metrics suitable for GMP environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated benzonitriles has relied on arduous multi-step pathways that pose severe challenges for industrial scale-up and environmental compliance. As illustrated in prior art such as Patent CN101337911A, traditional routes often commence with nitration and reduction sequences followed by bromination and diazotization, culminating in a Rosenmund-von Braun reaction using cuprous cyanide. 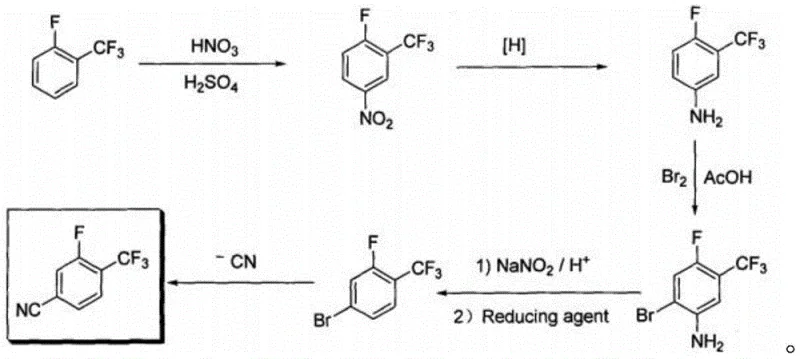 This classical approach is fraught with inefficiencies; the use of cuprous cyanide generates substantial amounts of heavy metal waste, necessitating expensive disposal protocols and rigorous wastewater treatment to meet regulatory standards. Furthermore, the diazotization step requires precise temperature control and generates unstable intermediates, increasing the risk of thermal runaway incidents. The cumulative yield across these five or six steps is often compromised, leading to higher raw material consumption and inflated production costs that erode profit margins for downstream API manufacturers.
This classical approach is fraught with inefficiencies; the use of cuprous cyanide generates substantial amounts of heavy metal waste, necessitating expensive disposal protocols and rigorous wastewater treatment to meet regulatory standards. Furthermore, the diazotization step requires precise temperature control and generates unstable intermediates, increasing the risk of thermal runaway incidents. The cumulative yield across these five or six steps is often compromised, leading to higher raw material consumption and inflated production costs that erode profit margins for downstream API manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN112979498A offers a streamlined alternative that drastically reduces the number of unit operations while enhancing overall process safety. The new route bypasses the need for hazardous cyanation reagents entirely by employing a catalytic hydrogenolysis strategy to install the nitrile functionality indirectly via a chloro-nitrile intermediate. This innovation allows for the use of commercially available and relatively inexpensive starting materials, such as substituted benzaldehydes, which are easier to source than specialized halogenated anilines. By condensing the synthesis into two primary stages, the process minimizes solvent usage and energy consumption, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The elimination of heavy metal catalysts like copper simplifies the workup procedure, as filtration and crystallization become sufficient to achieve high purity, thereby reducing the burden on quality control laboratories and accelerating batch release times for supply chain teams.
Mechanistic Insights into Catalytic Hydrogenolysis and Oximation
The core of this innovative synthesis lies in the precise orchestration of two distinct chemical transformations: the conversion of an aldehyde to a nitrile via an oxime intermediate, and the subsequent reductive dehalogenation. In the first stage, 2-chloro-3-fluoro-4-trifluoromethylbenzaldehyde reacts with a hydroxylamine salt in the presence of a carboxylic acid solvent, such as formic or acetic acid. This environment facilitates the formation of the aldoxime, which subsequently undergoes dehydration to yield 2-chloro-3-fluoro-4-trifluoromethylbenzonitrile. The choice of solvent is critical here, as formic acid acts dually as a reaction medium and a dehydrating agent, driving the equilibrium towards the nitrile product without requiring additional harsh dehydrating agents like phosphorus oxychloride. This mechanistic nuance ensures that the reaction proceeds smoothly at temperatures between 60°C and 180°C, yielding the chloro-intermediate with high selectivity and minimal byproduct formation.
The second stage involves a sophisticated catalytic hydrogenolysis where the carbon-chlorine bond is cleaved and replaced by a hydrogen atom, preserving the sensitive nitrile and trifluoromethyl groups. 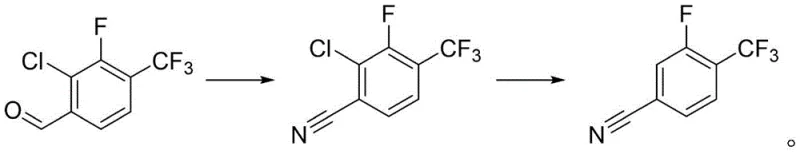 This transformation is mediated by heterogeneous catalysts such as palladium on carbon or Raney Nickel under mild hydrogen pressures ranging from 0.1 to 1 MPa. The presence of an acid-binding agent, such as triethylamine or potassium acetate, is essential to neutralize the hydrochloric acid generated during the dehalogenation, preventing catalyst poisoning and equipment corrosion. From a mechanistic standpoint, the catalyst surface adsorbs the chloro-nitrile and hydrogen gas, facilitating the oxidative addition of the C-Cl bond and its subsequent reductive elimination. This pathway is highly advantageous for impurity control, as it avoids the radical mechanisms often associated with homogenous reductions, thereby ensuring that the final 3-fluoro-4-trifluoromethyl benzonitrile maintains a purity profile exceeding 95 percent, which is critical for high-purity pharmaceutical intermediate applications.
This transformation is mediated by heterogeneous catalysts such as palladium on carbon or Raney Nickel under mild hydrogen pressures ranging from 0.1 to 1 MPa. The presence of an acid-binding agent, such as triethylamine or potassium acetate, is essential to neutralize the hydrochloric acid generated during the dehalogenation, preventing catalyst poisoning and equipment corrosion. From a mechanistic standpoint, the catalyst surface adsorbs the chloro-nitrile and hydrogen gas, facilitating the oxidative addition of the C-Cl bond and its subsequent reductive elimination. This pathway is highly advantageous for impurity control, as it avoids the radical mechanisms often associated with homogenous reductions, thereby ensuring that the final 3-fluoro-4-trifluoromethyl benzonitrile maintains a purity profile exceeding 95 percent, which is critical for high-purity pharmaceutical intermediate applications.
How to Synthesize 3-Fluoro-4-Trifluoromethyl Benzonitrile Efficiently
Implementing this synthesis protocol requires careful attention to reaction parameters to maximize yield and ensure reproducibility on a commercial scale. The process begins with the dissolution of the aldehyde substrate and hydroxylamine salt in a protic solvent, followed by heating to facilitate the cyclization and dehydration steps. Once the chloro-nitrile intermediate is isolated, it is subjected to hydrogenation in a pressurized reactor equipped with efficient agitation to ensure proper gas-liquid-solid contact. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and workup techniques described in the patent, provide a clear roadmap for process chemists to follow.
- Dissolve 2-chloro-3-fluoro-4-trifluoromethylbenzaldehyde and hydroxylamine salt in a solvent like formic acid, add an acid-binding agent, and heat to 60-180°C for 2-4 hours to obtain the chloro-nitrile intermediate.
- Dissolve the intermediate in a second solvent such as toluene, add a palladium or nickel catalyst and a base, then perform hydrogen replacement and react at 0-180°C under 0.1-1 MPa pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical efficiency. The shift away from toxic cuprous cyanide and phosphorus pentachloride eliminates the need for specialized hazardous material handling licenses and reduces the liability associated with storing dangerous reagents. This transition significantly lowers the barrier to entry for manufacturing facilities, allowing for broader geographic distribution of production sites and enhancing supply chain resilience against regional disruptions. Furthermore, the use of common solvents like toluene, xylene, and acetic acid ensures that raw material sourcing remains stable and cost-effective, even during periods of market volatility. The simplified purification process, which relies on standard filtration and recrystallization rather than complex chromatography or distillation, translates directly into reduced utility costs and shorter cycle times per batch.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of expensive and toxic reagents that require costly disposal methods. By replacing cuprous cyanide with catalytic hydrogenation, manufacturers avoid the generation of heavy metal sludge, which is a significant cost center in traditional fine chemical production. Additionally, the high yield of over 85 percent means that less raw material is wasted, optimizing the atom economy of the process. The ability to recycle solvents like toluene and xylene further contributes to substantial cost savings, making the overall cost of goods sold (COGS) much more competitive compared to legacy methods. This efficiency allows suppliers to offer more aggressive pricing structures to downstream API clients without compromising on margin.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted benzaldehydes and hydroxylamine salts mitigates the risk of supply bottlenecks that often plague specialty chemical markets. Unlike complex halogenated anilines which may have limited suppliers, the precursors for this route are commodity chemicals produced by multiple global vendors. This diversification of the supply base ensures continuity of supply even if one vendor faces production issues. Moreover, the robustness of the catalytic step, which tolerates a range of temperatures and pressures, reduces the likelihood of batch failures due to minor process deviations. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by multinational pharmaceutical companies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is inherently scalable because it avoids exothermic hazards associated with diazotization and the use of pyrophoric reagents. The three wastes generated are minimal and primarily consist of aqueous salt solutions and spent catalyst, which are easier to treat than the acidic and heavy metal-laden waste streams of conventional routes. This alignment with green chemistry principles facilitates smoother regulatory approvals and permits for plant expansion. The simplicity of the equipment requirements—standard stainless steel reactors and filtration units—means that scale-up from pilot plant to 100 MT annual production can be achieved with minimal capital expenditure, ensuring that the technology can be rapidly deployed to meet surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the purity and yield specifications for this new synthesis method?
A: According to patent CN112979498A, the product yield reaches more than 85 percent, and the purity is consistently maintained above 95 percent, ensuring suitability for downstream pharmaceutical applications.
Q: How does this method improve safety compared to traditional cuprous cyanide routes?
A: This novel approach eliminates the use of toxic cuprous cyanide and hazardous phosphorus pentachloride, replacing them with safer catalytic hydrogenolysis and common organic solvents, significantly reducing environmental hazards.
Q: Which catalysts are suitable for the hydrogenolysis step?
A: The process supports various effective catalysts including 5% to 20% palladium on carbon, palladium hydroxide on carbon, or Raney Nickel, providing flexibility for cost optimization and supply chain management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Fluoro-4-Trifluoromethyl Benzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our facility is equipped to handle the specific catalytic hydrogenation and oximation processes required for this synthesis, guaranteeing a consistent supply of 3-fluoro-4-trifluoromethyl benzonitrile that adheres to the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development programs.
