Advanced Metal-Free Synthesis of Diarylbenzylphosphine Oxides for Industrial Scale-up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies that align with stringent regulatory standards for pharmaceutical and agrochemical intermediates. Patent CN110143982B introduces a groundbreaking approach to the synthesis of diarylbenzylphosphine oxide compounds, a class of molecules critical for applications ranging from flame retardants to Horner-Wadsworth-Emmons olefination precursors. This innovation addresses long-standing challenges in P-C(sp3) bond formation by utilizing a transition-metal-free protocol that relies on cesium carbonate mediation. By leveraging readily available benzaldehyde derivatives and diarylphosphine oxides, this method offers a robust pathway to high-purity intermediates while generating valuable by-products, marking a substantial leap forward in sustainable fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of P-C(sp3) bonds has relied heavily on classical reactions such as the Michaelis-Arbuzov and Michaelis-Becker protocols, both of which present significant operational and safety hurdles for modern industrial applications. The Michaelis-Arbuzov reaction typically involves trivalent phosphorus compounds that are notoriously unstable and emit unpleasant, hazardous odors, complicating containment and worker safety in large-scale facilities. Furthermore, the Michaelis-Becker reaction necessitates the use of strong bases and often requires prolonged reaction times, which can lead to substrate decomposition and limited scope regarding functional group tolerance. More recent advancements involving transition metal catalysts like copper or palladium have attempted to mitigate these issues but introduce new complications, specifically the inevitable contamination of the final product with toxic metal residues.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a simple yet highly effective system comprising benzaldehyde derivatives, diarylphosphine oxides, and cesium carbonate in a dioxane solvent. This methodology eliminates the need for expensive and toxic transition metal catalysts, thereby removing the complex and costly downstream purification steps required to meet strict residual metal specifications (such as ICH Q3D guidelines). The reaction operates under a nitrogen atmosphere at moderate temperatures, ensuring high stereo- and position selectivity while maintaining a simple reaction profile. Crucially, this system transforms the diarylphosphine oxide into a dual-function reagent, acting as both a coupling partner and a reducing agent, which streamlines the stoichiometry and enhances the overall efficiency of the synthetic route.
Mechanistic Insights into Cs2CO3-Mediated P-C Bond Formation
The mechanistic elegance of this synthesis lies in the redox-neutral coupling facilitated by the inorganic base, which activates the P(O)-H bond without inducing harsh side reactions common with alkali metal hydroxides. The cesium cation likely plays a crucial role in stabilizing the transition state through favorable ion-pairing interactions, promoting the nucleophilic attack of the phosphorus species onto the carbonyl carbon of the benzaldehyde derivative. This initial addition forms a hydroxy-phosphonium intermediate, which subsequently undergoes a reduction-elimination sequence to yield the final benzylphosphine oxide. Unlike traditional cross-coupling reactions that require external reductants or oxidants, this system internally balances the redox potential by consuming two equivalents of the phosphine oxide for every equivalent of aldehyde.
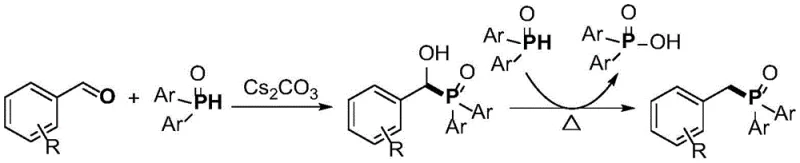
A distinctive feature of this mechanism is the generation of diarylphosphinic acid as a stoichiometric by-product, rather than useless waste salts. As illustrated in the reaction scheme, one molecule of the diarylphosphine oxide is incorporated into the target benzylphosphine oxide structure, while the second molecule is oxidized to the corresponding phosphinic acid. This by-product is not merely waste; it is a purifiable and chemically valuable compound that can be isolated and potentially utilized in other synthetic applications, thereby improving the overall atom economy of the process. This dual-output capability significantly reduces the environmental footprint of the synthesis, aligning with green chemistry principles by maximizing the utility of all input materials and minimizing the generation of non-recyclable waste streams.
How to Synthesize Diarylbenzylphosphine Oxide Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemists aiming to establish robust manufacturing lines for phosphorus-containing intermediates. The procedure involves mixing the benzaldehyde derivative, diarylphosphine oxide, and cesium carbonate in a reaction vessel, followed by the addition of a solvent such as 1,4-dioxane or tetrahydrofuran under an inert atmosphere. The mixture is then heated to temperatures ranging from 120°C to 160°C, with optimal results often observed around 140°C, and stirred for a period of 12 to 24 hours. Upon completion, the workup is straightforward, involving cooling, washing with saturated sodium chloride, extraction with ethyl acetate, and final purification via column chromatography to achieve purities exceeding 98%.
- Mix benzaldehyde derivative, diarylphosphine oxide compound, and cesium carbonate (Cs2CO3) in a reaction vessel under an inert gas atmosphere such as nitrogen or argon.
- Add a solvent system comprising 1,4-dioxane or tetrahydrofuran and heat the mixture to a reaction temperature between 120°C and 160°C for 12 to 24 hours with continuous stirring.
- Upon completion, cool the reaction to room temperature, wash with saturated NaCl, extract with ethyl acetate, and purify the crude product via column chromatography to isolate the target oxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling advantages by fundamentally restructuring the cost drivers associated with organophosphorus manufacturing. The elimination of precious metal catalysts such as palladium or copper not only reduces the direct material cost but also removes the dependency on volatile commodity markets for these strategic metals. Furthermore, the use of cesium carbonate, while a specialized base, is used in stoichiometric amounts that are predictable and manageable, avoiding the complexities of handling pyrophoric reagents or sensitive organometallic complexes that often require specialized storage and handling infrastructure.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts leads to significant cost savings by eliminating the need for expensive ligands and the rigorous purification steps required to remove trace metals from the final product. This simplification of the downstream processing directly translates to lower operational expenditures and reduced solvent consumption during the purification phase, enhancing the overall economic viability of producing high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on bench-stable, commercially available starting materials like benzaldehyde derivatives and diarylphosphine oxides ensures a resilient supply chain that is less susceptible to disruptions compared to routes requiring custom-synthesized catalysts. The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens and electron-withdrawing substituents, allows for the flexible sourcing of diverse raw materials without compromising the integrity of the final output.
- Scalability and Environmental Compliance: The generation of diarylphosphinic acid as a valuable co-product rather than toxic waste simplifies waste management protocols and reduces disposal costs, facilitating easier compliance with increasingly stringent environmental regulations. The reaction's compatibility with standard solvents like dioxane and THF, combined with moderate temperature requirements, ensures that the process can be safely scaled from kilogram to multi-ton production levels using existing reactor infrastructure without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding the implementation of this novel synthesis method, derived directly from the experimental data and background analysis of the patent documentation. These insights are intended to clarify the operational parameters and strategic benefits for stakeholders evaluating this technology for integration into their existing manufacturing portfolios.
Q: What are the primary advantages of this Cs2CO3-mediated method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts such as copper or palladium, which removes the risk of toxic metal residues in the final API intermediate and simplifies the purification process significantly.
Q: Can this synthesis method be scaled for commercial production of flame retardant precursors?
A: Yes, the method utilizes cheap and readily available raw materials like benzaldehyde derivatives and operates under relatively mild conditions (100-180°C), making it highly suitable for large-scale commercial manufacturing of flame retardant precursors.
Q: What is the nature of the by-product generated in this reaction system?
A: The reaction generates diarylphosphinic acid as a stoichiometric by-product, which is a purifiable and chemically valuable compound, thereby enhancing the overall atom economy and reducing waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylbenzylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value organophosphorus intermediates used in pharmaceuticals and advanced materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of diarylbenzylphosphine oxide meets the exacting standards required for downstream drug substance manufacturing.
We invite you to collaborate with our technical team to explore how this innovative Cs2CO3-mediated protocol can optimize your supply chain and reduce your overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project's unique requirements. Let us help you leverage this cutting-edge chemistry to secure a competitive advantage in the global market for fine chemical intermediates.
