Advanced NHC-Metal Catalysis for Efficient Phosphine Ligand Manufacturing and Commercial Scale-Up
Introduction to Next-Generation Phosphine Synthesis Technology
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the need for more sustainable and efficient synthetic methodologies. Patent CN101824052A introduces a groundbreaking approach for the catalytic synthesis of phosphine compounds utilizing N-heterocyclic carbene (NHC) metal complexes. This technology represents a paradigm shift from traditional harsh synthetic routes to a mild, highly selective catalytic process that operates effectively at temperatures ranging from -78°C to 100°C, with optimal performance observed between -20°C and 40°C. For R&D directors and procurement specialists in the fine chemical sector, this innovation offers a robust pathway to access high-purity phosphine ligands which are critical components in homogeneous catalysis, pharmaceutical intermediates, and advanced material science. The core breakthrough lies in the ability to directly couple substituted phosphine hydrides with various electron-deficient olefins without the necessity for cumbersome protecting group strategies that have historically plagued this chemical space.
The significance of this patent extends beyond mere academic interest; it addresses fundamental bottlenecks in the commercial manufacturing of organophosphorus compounds. By leveraging the unique electronic and steric properties of NHC ligands coordinated to transition metals such as copper, silver, or gold, the method achieves high reaction efficiency with catalyst loadings as low as 0.1% to 2%. This level of efficiency is paramount for industrial applications where catalyst cost and metal residue limits are critical constraints. Furthermore, the broad substrate scope encompasses a wide array of electron-withdrawing groups including carbonyls, nitro groups, and cyano groups, enabling the synthesis of diverse phosphine architectures. As a reliable pharmaceutical intermediate supplier, understanding and adopting such advanced catalytic technologies is essential for maintaining competitiveness in the global market, ensuring that supply chains are resilient against the volatility of raw material costs and regulatory pressures regarding hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trivalent phosphine compounds via hydrophosphination of unsaturated olefins has been fraught with significant technical challenges that impede scalable manufacturing. Traditional protocols frequently rely on the use of borane-protected phosphine hydrides as starting materials to prevent oxidation and control reactivity. While effective in small-scale laboratory settings, this protection-deprotection sequence adds substantial steps to the synthetic route, increasing both the time and cost of production. Moreover, conventional methods often necessitate the use of strong bases to activate the phosphine-hydrogen bond, which can lead to compatibility issues with substrates containing acid-sensitive functional groups. Alternative approaches involving radical mechanisms or microwave heating, while sometimes effective, often suffer from poor reproducibility and safety concerns when scaled to multi-kilogram or tonne quantities. The requirement for harsh reaction conditions not only elevates energy consumption but also generates complex impurity profiles that are difficult to remove, thereby compromising the purity specifications required for high-value applications in the electronics and pharmaceutical industries.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes N-heterocyclic carbene metal complexes to catalyze the hydrophosphination reaction under remarkably mild conditions. This methodology eliminates the strict requirement for borane protection at the outset, allowing for the direct use of simple phosphine hydride raw materials. The catalytic system demonstrates exceptional versatility, accommodating a wide range of substrates including those with sensitive functionalities that would typically decompose under basic or radical conditions. The operational simplicity is another key advantage; reactions can be conducted in common organic solvents such as dichloromethane, toluene, or tetrahydrofuran at ambient or slightly elevated temperatures. This shift towards milder conditions translates directly into enhanced process safety and reduced environmental impact, aligning with modern green chemistry principles. For procurement managers, this means a streamlined supply chain with fewer unit operations, lower solvent usage, and simplified waste treatment protocols, ultimately driving down the total cost of ownership for these critical chemical building blocks.
Mechanistic Insights into NHC-Metal Complex Catalyzed Hydrophosphination
The efficacy of this synthetic method is rooted in the sophisticated coordination chemistry of the N-heterocyclic carbene metal complexes. These catalysts function by stabilizing the transition metal center through strong sigma-donation from the carbene carbon, which enhances the metal's ability to activate the phosphine-hydrogen bond. 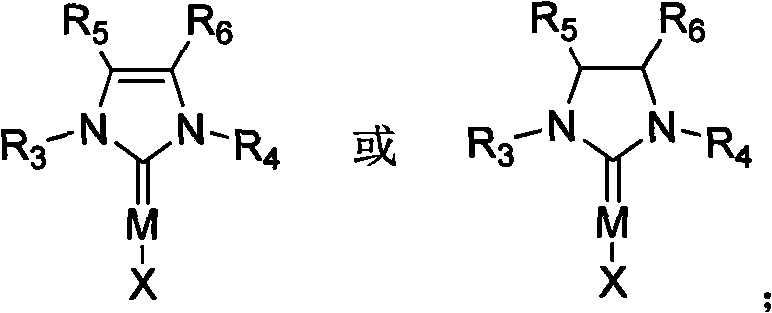 As illustrated in the general catalyst structure, the NHC ligand framework provides a tunable steric environment that can be modified by varying the substituents on the nitrogen atoms (R3, R4) and the backbone (R5, R6). This tunability allows chemists to optimize the catalyst for specific substrate classes, ensuring high turnover numbers and selectivity. The mechanism likely involves the oxidative addition of the P-H bond to the metal center, followed by the insertion of the electron-deficient olefin into the metal-phosphorus bond, and finally reductive elimination to release the product and regenerate the active catalyst species. This catalytic cycle avoids the high-energy intermediates associated with uncatalyzed radical pathways, thereby minimizing side reactions and polymerization of the olefin substrate.
As illustrated in the general catalyst structure, the NHC ligand framework provides a tunable steric environment that can be modified by varying the substituents on the nitrogen atoms (R3, R4) and the backbone (R5, R6). This tunability allows chemists to optimize the catalyst for specific substrate classes, ensuring high turnover numbers and selectivity. The mechanism likely involves the oxidative addition of the P-H bond to the metal center, followed by the insertion of the electron-deficient olefin into the metal-phosphorus bond, and finally reductive elimination to release the product and regenerate the active catalyst species. This catalytic cycle avoids the high-energy intermediates associated with uncatalyzed radical pathways, thereby minimizing side reactions and polymerization of the olefin substrate.
From an impurity control perspective, the mild nature of the NHC-catalyzed process significantly reduces the formation of by-products such as phosphine oxides or oligomerized olefins. In traditional base-mediated reactions, competing deprotonation events can lead to complex mixtures that are difficult to separate. However, the transition metal catalysis ensures a more defined reaction trajectory. The patent highlights that the resulting phosphine compounds can be isolated as free trivalent phosphines or converted in situ into air-stable derivatives. For instance, the addition of borane solutions or hydrogen peroxide post-reaction allows for the generation of borane-phosphine complexes or phosphine oxides, respectively. This flexibility is crucial for supply chain management, as it permits the storage and transport of stable intermediates which can be deprotected or utilized directly in downstream coupling reactions. The ability to control the oxidation state of the phosphorus atom post-synthesis provides a strategic advantage in managing the stability and shelf-life of inventory, reducing losses due to degradation during warehousing.
How to Synthesize Phosphine Compounds Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to both laboratory optimization and industrial scale-up. The process begins with the preparation of the reaction vessel under strictly anhydrous and oxygen-free conditions, typically utilizing Schlenk techniques or glovebox environments to preserve catalyst integrity. A representative procedure involves dissolving the NHC-metal catalyst, such as an IPr-CuCl complex, in a dry organic solvent like dichloromethane. The substituted phosphine hydride is then introduced, followed by the electron-deficient olefin substrate. The reaction mixture is stirred at room temperature or mildly heated, depending on the specific reactivity of the olefin. 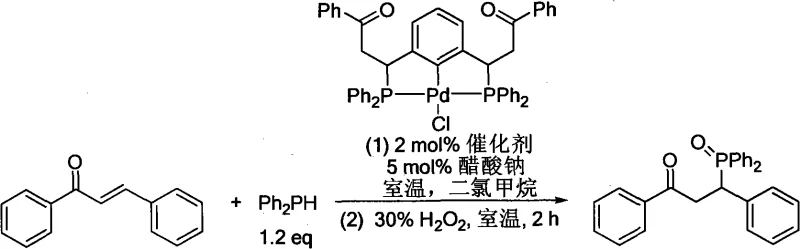 As demonstrated in the application example involving chalcone, the reaction proceeds smoothly to afford the corresponding phosphine oxide after an oxidative workup with hydrogen peroxide. This specific transformation highlights the robustness of the method, achieving high yields (e.g., 87%) with minimal catalyst loading (2 mol%). The detailed standardized synthesis steps for various substrates are provided in the guide below, ensuring reproducibility across different batches and facilities.
As demonstrated in the application example involving chalcone, the reaction proceeds smoothly to afford the corresponding phosphine oxide after an oxidative workup with hydrogen peroxide. This specific transformation highlights the robustness of the method, achieving high yields (e.g., 87%) with minimal catalyst loading (2 mol%). The detailed standardized synthesis steps for various substrates are provided in the guide below, ensuring reproducibility across different batches and facilities.
- Prepare the reaction system under anhydrous and oxygen-free conditions using a Schlenk tube, adding the NHC-metal catalyst and organic solvent such as dichloromethane.
- Introduce the substituted phosphine hydrogen compound and stir at room temperature, followed by the addition of the electron-deficient olefin substrate under nitrogen atmosphere.
- Allow the reaction to proceed at mild temperatures ranging from -20°C to 40°C, then concentrate and purify the resulting phosphine product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed synthesis route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the manufacturing process. By eliminating the need for borane protection groups and strong bases, the number of synthetic steps is reduced, which directly correlates to lower labor costs, reduced equipment occupancy time, and decreased consumption of auxiliary reagents. This streamlining of the process flow enhances the overall throughput of the manufacturing facility, allowing for faster turnaround times on custom synthesis orders. Furthermore, the use of mild reaction conditions reduces the energy burden associated with heating or cooling large reactors, contributing to a lower carbon footprint and alignment with corporate sustainability goals. The compatibility of the method with common, inexpensive solvents further aids in cost reduction in fine chemical manufacturing, as there is no need for specialized or exotic solvent systems that are difficult to source or recycle.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps inherently lowers the material costs associated with reagents like borane complexes and quenching agents. Additionally, the high catalytic efficiency means that precious metal usage is minimized, reducing the exposure to volatile metal prices. The simplified purification process, often requiring only standard column chromatography or recrystallization, reduces the demand for expensive silica gel or specialized resins. These factors combine to create a significantly more cost-effective production model compared to traditional methods, allowing for competitive pricing strategies in the global market without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against moisture and air (post-workup) improves the stability of the final products, reducing the risk of spoilage during transit and storage. The wide substrate scope ensures that the same catalytic platform can be used to produce a diverse portfolio of phosphine ligands, reducing the need for multiple specialized production lines. This flexibility enhances supply chain resilience, enabling rapid switching between products to meet fluctuating market demands. Moreover, the availability of simple starting materials like phosphine hydrides and common olefins ensures a stable upstream supply, mitigating risks associated with raw material shortages that often plague more complex synthetic routes.
- Scalability and Environmental Compliance: The mild conditions and lack of hazardous reagents make this process highly scalable from gram to tonne quantities with minimal engineering modifications. The reduction in hazardous waste generation simplifies compliance with increasingly stringent environmental regulations regarding waste disposal and emissions. The ability to produce air-stable derivatives directly in the reactor minimizes the handling of pyrophoric or highly sensitive intermediates, enhancing workplace safety. This ease of scale-up ensures that commercial production can meet the growing demand for high-purity phosphine compounds in the semiconductor and pharmaceutical sectors without the lengthy lead times typically associated with process development for sensitive chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners and clients. Understanding these nuances is critical for integrating this technology into existing manufacturing workflows and ensuring that quality expectations are met consistently.
Q: What are the advantages of using NHC-metal complexes over traditional strong base methods for phosphine synthesis?
A: Traditional methods often require harsh conditions like strong bases or borane protection groups which complicate purification. The NHC-metal complex method described in patent CN101824052A operates under mild conditions (-20°C to 40°C) without the need for initial borane protection, significantly simplifying the workflow and reducing waste.
Q: Can this catalytic method tolerate sensitive functional groups on the olefin substrate?
A: Yes, the mild nature of the NHC-catalyzed hydrophosphination allows for excellent tolerance of various functional groups. The patent demonstrates successful reactions with electron-deficient olefins containing ketones, esters, and halides without degradation, making it suitable for complex pharmaceutical intermediate synthesis.
Q: How is the air sensitivity of the trivalent phosphine products managed in this process?
A: While trivalent phosphines are air-sensitive, the process allows for direct isolation or in-situ conversion. The patent describes options to oxidize the product to stable pentavalent phosphine oxides using hydrogen peroxide or form air-stable borane complexes post-reaction, ensuring safe handling and storage for supply chain logistics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the NHC-metal complex system described in patent CN101824052A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild and efficient synthesis route are fully realized in large-volume supply contracts. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of phosphine intermediate meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality assurance ensures that impurity profiles are tightly controlled, providing our clients with the confidence needed for their own downstream processing.
We invite industry leaders to collaborate with us to leverage this technology for their specific supply chain needs. Whether you require custom synthesis of novel phosphine ligands or reliable sourcing of established intermediates, our technical procurement team is ready to assist. We encourage you to contact us for a Customized Cost-Saving Analysis that evaluates how adopting this catalytic route can optimize your specific production costs. Reach out today to request specific COA data and route feasibility assessments tailored to your project requirements, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
