Advanced Biocatalytic Synthesis of Levo-Praziquantel Intermediates for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift in the manufacturing of anti-parasitic agents, driven by the urgent need for greener, more efficient synthetic routes. Patent CN102925528B, published in July 2014, introduces a groundbreaking methodology for the production of levo-praziquantel intermediates and the final active pharmaceutical ingredient (API). This technology leverages the exquisite stereoselectivity of nitrilase enzymes to overcome the longstanding limitations of traditional chemical resolution. Unlike conventional methods that rely on stoichiometric chiral resolving agents and harsh reaction conditions, this biocatalytic approach utilizes a kinetic resolution strategy where the enzyme selectively hydrolyzes the unwanted S-isomer, leaving the desired R-configuration nitrile intact. This innovation not only simplifies the purification process but also aligns with modern green chemistry principles, making it a highly attractive option for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
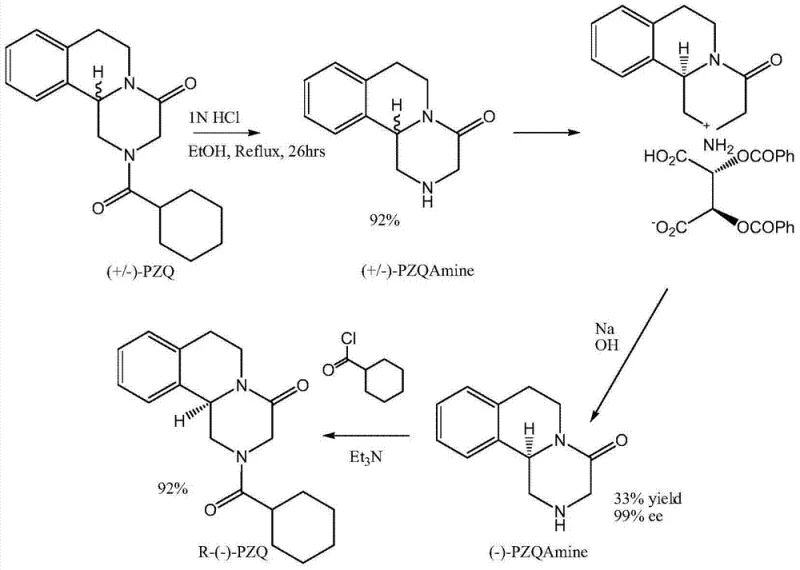
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of praziquantel has been plagued by significant technical and economic inefficiencies inherent to classical chemical synthesis. The traditional route, as illustrated in the comparative reaction schemes, typically involves the use of hazardous reagents such as potassium cyanide and hexamethylene acyl chloride under rigorous conditions. Furthermore, obtaining the pharmacologically active levo-isomer traditionally requires a chemical resolution step, often utilizing tartaric acid derivatives. This classical resolution is fundamentally limited by a maximum theoretical yield of 50% for the desired enantiomer, necessitating the recycling or disposal of the unwanted isomer, which drastically inflates production costs. The process also generates substantial amounts of chemical waste and requires energy-intensive recrystallization cycles to achieve acceptable enantiomeric purity, creating bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a biocatalytic kinetic resolution that fundamentally alters the economic landscape of levo-praziquantel manufacturing. By utilizing nitrilases derived from sources such as Arabidopsis thaliana or Aspergillus niger, the process achieves high regioselectivity and stereoselectivity at mild temperatures ranging from 0°C to 80°C. The enzyme specifically targets the S-configured nitrile group for hydrolysis into a carboxylic acid, which is easily separated from the unreacted R-configured nitrile intermediate. This R-nitrile serves as the direct precursor for the final drug substance. This approach effectively bypasses the 50% yield ceiling of traditional resolution by preventing the formation of the unwanted isomer in the downstream process, thereby streamlining the workflow and significantly reducing the raw material intensity required for cost reduction in pharmaceutical intermediate manufacturing.
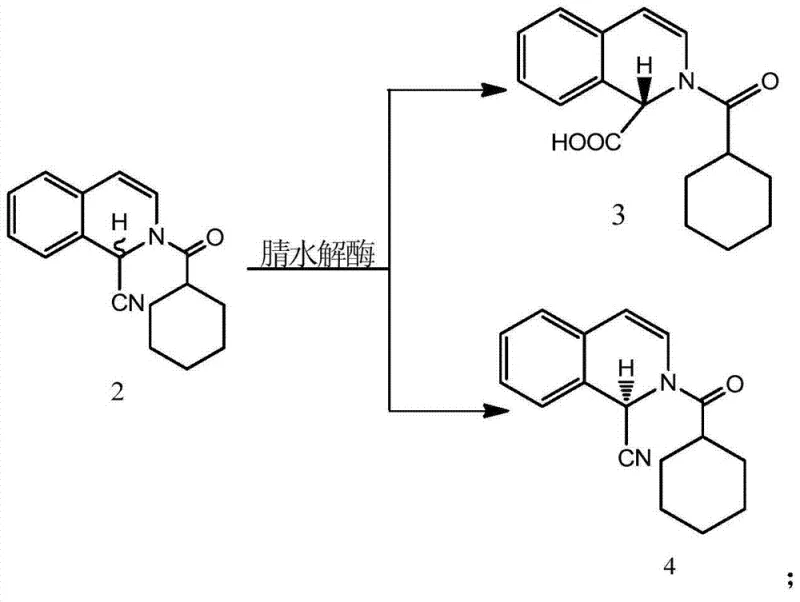
Mechanistic Insights into Nitrilase-Catalyzed Kinetic Resolution
The core of this technological breakthrough lies in the precise mechanistic action of the nitrilase enzyme on the racemic substrate. Nitrilases are hydrolytic enzymes that catalyze the conversion of nitriles directly to carboxylic acids and ammonia without forming amide intermediates. In this specific application, the enzyme exhibits strict enantioselectivity, recognizing and binding only the S-enantiomer of the racemic nitrile intermediate (Formula 2). The catalytic cycle involves the nucleophilic attack of a cysteine residue within the enzyme's active site on the nitrile carbon, leading to the formation of a covalent thioimidate intermediate which is subsequently hydrolyzed. This biological specificity ensures that the R-enantiomer remains chemically untouched in the reaction mixture. Consequently, the reaction mixture becomes a binary system of the water-soluble S-acid byproduct and the organic-soluble R-nitrile product, facilitating a straightforward liquid-liquid extraction that yields the intermediate with an enantiomeric excess (ee) value exceeding 98%.
From an impurity control perspective, this enzymatic route offers distinct advantages over transition metal-catalyzed alternatives. Traditional chemical synthesis often leaves traces of heavy metals or requires complex scavenging steps to meet regulatory limits for residual catalysts in APIs. The biocatalytic step operates in aqueous phosphate buffers at near-neutral pH, minimizing the formation of side products associated with acidic or basic hydrolysis. Furthermore, the subsequent steps—catalytic hydrogenation and cyclization—are performed on a highly purified intermediate, which reduces the burden on downstream purification units. The high stereochemical integrity of the R-nitrile intermediate ensures that the final levo-praziquantel product maintains high optical purity without the need for chiral chromatography, a technique that is often prohibitively expensive and difficult to scale for commercial production.
How to Synthesize Levo-Praziquantel Efficiently
The synthesis of levo-praziquantel via this patented route is designed for operational simplicity and robustness, making it ideal for transfer to large-scale production facilities. The process begins with the preparation of the racemic nitrile precursor, followed by the critical enzymatic resolution step which sets the stereochemistry. Once the R-nitrile intermediate is isolated, it undergoes catalytic hydrogenation to reduce the nitrile group to a primary amine, followed by a cyclization reaction with chloroacetyl chloride to close the piperazine ring. The detailed standardized synthetic steps, including specific solvent systems, catalyst loadings, and temperature profiles optimized for maximum yield and purity, are outlined in the guide below.
- Perform stereo-selective hydrolysis of racemic nitrile intermediate (Formula 2) using nitrilase at 0-80°C to isolate the R-configuration nitrile (Formula 4).
- Subject the isolated R-nitrile intermediate to catalytic hydrogenation in organic solvents (e.g., methanol) using Pd-C or Ru-C catalysts to form the amine intermediate (Formula 5).
- React the amine intermediate with chloroacetyl chloride in the presence of a base to effect cyclization, yielding the final levo-praziquantel product (Formula 6).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrilase-based technology translates into tangible strategic benefits beyond mere technical elegance. The shift from chemical resolution to biocatalysis eliminates the need for expensive chiral resolving agents, which are often sourced from limited suppliers and subject to price volatility. Additionally, the mild reaction conditions reduce the energy consumption associated with heating and cooling large reactors, contributing to a lower overall carbon footprint. The simplified workup procedures, relying on basic extraction rather than complex crystallizations, shorten the batch cycle time, allowing for increased throughput and better responsiveness to market demand fluctuations. These factors collectively drive a substantial cost reduction in pharmaceutical intermediate manufacturing while enhancing the resilience of the supply chain against raw material shortages.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the improvement in overall yield significantly lower the cost of goods sold (COGS). By avoiding the 50% yield loss inherent in classical resolution, the effective utilization of raw materials is nearly doubled. Furthermore, the use of commercially available nitrilases removes the dependency on proprietary chiral ligands or complex metal catalysts, simplifying the bill of materials and reducing procurement overheads associated with specialized reagents.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process ensures consistent quality across batches, reducing the risk of production delays caused by failed purity specifications. The starting materials, including isoquinoline and cyclohexanecarbonyl chloride, are commodity chemicals with stable global supply chains. This accessibility ensures that production schedules are not disrupted by the scarcity of exotic reagents, providing a reliable source of high-purity pharmaceutical intermediates for downstream formulation partners.
- Scalability and Environmental Compliance: Biocatalytic processes are inherently scalable, as enzyme activity can be maintained or enhanced through fed-batch strategies in large fermenters or stirred-tank reactors. The aqueous nature of the primary reaction step minimizes the use of volatile organic compounds (VOCs), aligning with increasingly stringent environmental regulations. This green chemistry profile facilitates easier permitting for new production lines and reduces the costs associated with waste treatment and disposal, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrilase-mediated synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their supply chains.
Q: What is the primary advantage of using nitrilase over chemical resolution for praziquantel?
A: The nitrilase-catalyzed route offers superior stereoselectivity, specifically hydrolyzing the S-isomer to leave the desired R-nitrile intermediate with high enantiomeric excess (>98% ee), avoiding the 50% yield loss typical of classical resolution.
Q: Which nitrilase sources are effective for this synthesis?
A: The patent specifies effective enzymes from Arabidopsis thaliana, Aspergillus niger, and Bacillus foecalis alkaligenes, providing flexibility in sourcing biocatalysts for industrial production.
Q: How does this method impact environmental compliance?
A: By replacing harsh chemical reagents like potassium cyanide and hexamethylene acyl chloride used in traditional routes with aqueous enzymatic processes, the method significantly reduces toxic waste and improves safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levo-Praziquantel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the nitrilase-catalyzed route described in CN102925528B and possesses the technical capability to implement this process at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed international pharmacopoeia standards.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cost-effective and environmentally friendly technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in biocatalysis can optimize your supply chain for levo-praziquantel and other complex chiral intermediates.
