Advancing Liquid Crystal Intermediates: A Green TEMPO-Catalyzed Oxidation Strategy for Commercial Scale-Up
Advancing Liquid Crystal Intermediates: A Green TEMPO-Catalyzed Oxidation Strategy for Commercial Scale-Up
The rapid evolution of the display industry demands increasingly sophisticated liquid crystal materials that offer superior stability and electro-optical performance. At the heart of this technological progression lies the efficient synthesis of key intermediates, specifically 4-(4'-alkylcyclohexyl)cyclohexanone, a critical building block for high-performance liquid crystal monomers. Recent intellectual property developments, notably patent CN107501059B, have introduced a transformative green synthesis method that addresses long-standing environmental and economic challenges in this sector. This innovation replaces hazardous heavy metal oxidants with a catalytic TEMPO (2,2,6,6-tetramethylpiperidine-N-oxyl) system, utilizing inexpensive hypochlorite as the terminal oxidant. For R&D directors and procurement strategists, this shift represents more than just a chemical modification; it signifies a fundamental restructuring of the supply chain towards sustainability, cost-efficiency, and regulatory compliance. By leveraging mild reaction conditions and recyclable non-polar solvents, this technology paves the way for reliable liquid crystal intermediate supplier networks that can meet the rigorous purity standards of the electronics sector without compromising on environmental stewardship.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4-(4'-alkylcyclohexyl)cyclohexanone relied heavily on stoichiometric oxidation using reagents such as Jones reagent, pyridinium chlorochromate (PCC), or various dichromate salts. While chemically effective, these traditional pathways are plagued by severe ecological and operational drawbacks that render them increasingly obsolete in modern manufacturing environments. The primary concern is the generation of substantial quantities of heavy metal waste, particularly hexavalent chromium salts, which are classified as carcinogenic and pose significant disposal challenges under strict international environmental regulations. Furthermore, alternative methods utilizing hydrogen peroxide with tungsten-based catalysts often necessitate high-temperature conditions ranging from 70°C to 90°C, introducing inherent safety risks related to thermal runaway and potential explosion hazards. Additionally, many of these legacy processes employ polar aprotic solvents like acetonitrile or N-methylpyrrolidone (NMP), which are difficult to recover due to their high boiling points and water miscibility, leading to excessive solvent consumption and the generation of large volumes of organic-contaminated wastewater that requires expensive treatment protocols.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in patent CN107501059B utilizes a biomimetic catalytic cycle driven by TEMPO and sodium hypochlorite. This methodology operates under remarkably mild conditions, typically between 0°C and 40°C, thereby eliminating the thermal safety risks associated with high-temperature peroxide oxidations. The reaction employs non-polar organic solvents such as toluene or dichloromethane, which form distinct biphasic systems with the aqueous hypochlorite solution, facilitating straightforward phase separation and enabling high-efficiency solvent recycling. As illustrated in the reaction scheme below, the transformation proceeds cleanly from the corresponding cyclohexanol precursor to the target ketone with exceptional selectivity.
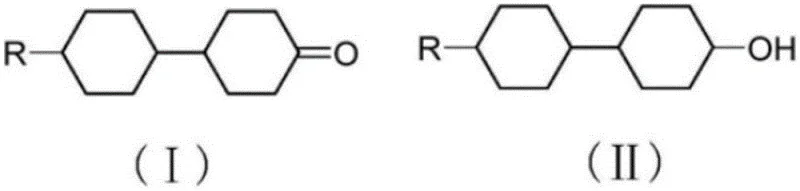
This biphasic nature ensures that the organic product remains in the solvent phase while inorganic salt byproducts remain in the aqueous phase, simplifying the workup procedure to mere washing and concentration. The result is a process that not only achieves high yields exceeding 98% but also drastically reduces the environmental footprint by minimizing waste emission and avoiding the use of toxic heavy metals, making it an ideal candidate for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into TEMPO-Catalyzed Oxidation
The core of this technological breakthrough lies in the elegant catalytic cycle mediated by the stable nitroxyl radical, TEMPO. In this mechanism, the TEMPO radical acts as the active oxidizing species, abstracting a hydrogen atom from the hydroxyl group of the 4-(4'-alkylcyclohexyl)cyclohexanol substrate to form an intermediate alkoxyamine species. This species subsequently undergoes elimination to release the desired ketone product and the reduced hydroxylamine form of the catalyst. Crucially, the catalytic cycle is closed by the stoichiometric oxidant, hypochlorite (bleach), which rapidly re-oxidizes the hydroxylamine back to the active nitroxyl radical, allowing the catalyst to turnover continuously. This regeneration step is highly efficient and occurs rapidly at ambient temperatures, ensuring that only catalytic amounts of the relatively expensive TEMPO are required while the bulk of the oxidation potential is supplied by the extremely cheap hypochlorite. The selectivity of this system is paramount; unlike harsh chromic acid oxidations which can lead to over-oxidation or ring cleavage, the TEMPO-mediated pathway is highly chemoselective for primary and secondary alcohols, preserving the sensitive cyclohexane ring structure essential for liquid crystal properties.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material and liquid crystal precursors. The mild reaction conditions prevent the formation of thermal degradation byproducts that are common in high-temperature processes. Furthermore, the biphasic reaction environment inherently separates the organic product from inorganic salts and water-soluble impurities. Any residual hypochlorite, which could potentially chlorinate the alkyl side chains, is effectively quenched during the workup using reducing agents like sodium bisulfite or sodium thiosulfate, as confirmed by starch-iodide testing. This rigorous control over the reaction environment ensures that the final product meets stringent purity specifications without the need for complex chromatographic purification, thereby streamlining the path to commercial scale-up of complex polymer additives and electronic chemicals.
How to Synthesize 4-(4'-alkylcyclohexyl)cyclohexanone Efficiently
Implementing this green synthesis route requires precise control over reaction parameters to maximize yield and solvent recovery. The process begins by dissolving the trans-4-(trans-4'-alkylcyclohexyl)cyclohexanol starting material in a recycled non-polar solvent such as toluene, followed by the addition of the TEMPO catalyst. The oxidation is initiated by the controlled dropwise addition of an aqueous hypochlorite solution while maintaining the temperature within the optimal 0-40°C range to manage exothermicity. Following the reaction period, the organic phase is separated and washed with a reducing agent solution to neutralize any remaining oxidant, ensuring product stability. For a comprehensive guide on the specific molar ratios, solvent volumes, and quenching procedures validated by patent data, please refer to the standardized protocol below.
- Dissolve the raw material 4-(4'-alkylcyclohexyl)cyclohexanol in a non-polar organic solvent such as toluene or dichloromethane.
- Add the TEMPO catalyst and dropwise add an aqueous hypochlorite solution while maintaining the temperature between 0-40°C.
- After reaction completion, remove excess hypochlorite using a reducing agent wash, separate the organic phase, and concentrate to obtain the pure ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TEMPO-catalyzed synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The transition away from heavy metal oxidants eliminates the substantial costs associated with hazardous waste disposal and environmental compliance reporting, which have become increasingly burdensome in the global chemical landscape. Moreover, the ability to utilize and recycle common non-polar solvents like toluene, with recovery rates demonstrated to be exceptionally high in pilot examples, significantly lowers the recurring raw material expenditure. This efficiency is compounded by the simplified post-treatment workflow, which removes the need for energy-intensive distillation steps required to recover high-boiling polar solvents used in older methods.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic chromium-based reagents with commodity-grade sodium hypochlorite results in a drastic reduction in direct material costs. Since the catalyst loading is minimal and the oxidant is inexpensive, the variable cost per kilogram of product is significantly optimized. Furthermore, the elimination of heavy metal waste streams removes the need for specialized waste treatment facilities, leading to substantial operational savings that enhance overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity chemicals such as bleach and toluene ensures a robust and resilient supply chain that is less susceptible to the shortages often seen with specialized reagents. The mild reaction conditions allow for production in standard glass-lined or stainless steel reactors without the need for exotic high-pressure or high-temperature equipment, increasing the number of qualified contract manufacturing organizations capable of executing the synthesis. This flexibility reduces lead time for high-purity liquid crystal intermediates and ensures consistent supply continuity even during market fluctuations.
- Scalability and Environmental Compliance: The inherent safety of the low-temperature process facilitates seamless scale-up from laboratory grams to multi-ton commercial production without the exponential increase in risk profile associated with peroxide oxidations. The process aligns perfectly with green chemistry principles by generating minimal waste and avoiding persistent organic pollutants, making it easier to secure regulatory approvals in strict jurisdictions. This environmental compatibility future-proofs the supply chain against tightening global regulations on chemical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding safety, purity, and scalability, derived directly from the technical disclosures and experimental data provided in the patent literature. These insights are intended to clarify the operational advantages and mitigate potential concerns regarding the transition to this greener catalytic system.
Q: Why is the TEMPO/hypochlorite system preferred over traditional Jones reagent for this intermediate?
A: Traditional Jones reagent utilizes hexavalent chromium, which generates highly toxic heavy metal waste requiring complex disposal procedures. The TEMPO system uses inexpensive hypochlorite and produces benign salt byproducts, aligning with modern green chemistry standards.
Q: What are the solvent recovery capabilities of this new process?
A: The process utilizes non-polar solvents like toluene which do not mix with the aqueous reaction phase. Patent data indicates solvent recovery rates exceeding 93%, significantly reducing raw material consumption and waste generation compared to water-miscible solvents.
Q: Is this oxidation method safe for large-scale industrial production?
A: Yes, the reaction operates at mild temperatures (0-40°C) avoiding the high-temperature explosion risks associated with hydrogen peroxide oxidations. The exotherm is easily controlled via dropwise addition, ensuring operational safety at the metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4'-alkylcyclohexyl)cyclohexanone Supplier
As the demand for high-performance display materials continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the TEMPO-oxidation route to deliver superior liquid crystal intermediates. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-(4'-alkylcyclohexyl)cyclohexanone meets the exacting standards required for next-generation LCD and OLED applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through innovative chemistry. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our green synthesis methods can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you build a more sustainable and efficient future for your electronic chemical sourcing.
