Scalable Copper-Catalyzed Synthesis of 2-Indole-3-Oxoindoline Compounds for Pharma
Introduction to Advanced Indole Synthesis Technologies
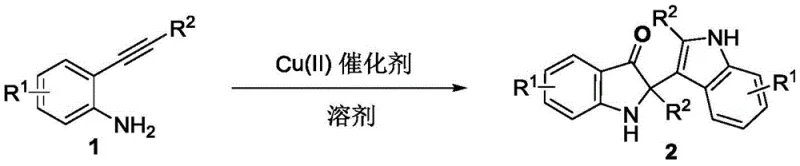
The landscape of organic synthesis for bioactive heterocycles is continuously evolving, driven by the demand for more efficient and sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN111574427A, which discloses a novel method for synthesizing 2-indole-3-oxoindoline compounds. These structures serve as critical scaffolds in medicinal chemistry, particularly for developing antiviral agents and fluorescent materials. The disclosed technology leverages an intermolecular dimerization strategy of 2-alkynylaniline derivatives, utilizing a divalent copper catalyst under mild oxidative conditions. This approach represents a paradigm shift from traditional multi-step syntheses, offering a direct route to complex bis-indole architectures that are otherwise difficult to access. For R&D teams focused on pipeline acceleration, understanding the nuances of this copper-catalyzed oxidative cyclization is essential for integrating these high-value intermediates into drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2,2-disubstituted indol-3-one frameworks has been plagued by synthetic inefficiencies that hinder large-scale production. Conventional routes often rely on harsh reaction conditions, including strong acids or bases and elevated temperatures that can degrade sensitive functional groups. Furthermore, many existing protocols suffer from poor atom economy, generating substantial waste streams due to the requirement for stoichiometric oxidants or protecting group manipulations. The availability of starting materials has also been a bottleneck, with many precursors being expensive or requiring tedious preparation steps. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for producing high-purity pharmaceutical intermediates, creating friction in the supply chain for contract development and manufacturing organizations (CDMOs).
The Novel Approach
In stark contrast, the methodology described in the patent introduces a streamlined, one-pot synthesis that directly couples two molecules of 2-alkynylaniline. By employing a catalytic amount of inexpensive copper salts, such as copper(II) acetate, the reaction proceeds efficiently in hexafluoroisopropanol (HFIP) under an oxygen atmosphere. This eliminates the need for precious metal catalysts like palladium or rhodium, drastically reducing raw material costs. The reaction conditions are remarkably mild, typically operating between 60°C and 100°C, which enhances safety profiles and reduces energy consumption. Moreover, the use of molecular oxygen as the terminal oxidant ensures that water is the primary byproduct, aligning perfectly with green chemistry principles. This novel approach not only simplifies the operational workflow but also expands the accessible chemical space by tolerating a wide range of substituents on the aromatic rings.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this transformation lies in the unique reactivity of the divalent copper species within the HFIP solvent system. The mechanism likely initiates with the coordination of the copper(II) center to the alkyne moiety of the 2-alkynylaniline, activating it towards nucleophilic attack. Subsequent intramolecular cyclization forms the initial indole ring, followed by an oxidative coupling event that links two indole units at the C3 and C2 positions respectively. The presence of oxygen is critical, as it regenerates the active copper(II) species from the reduced copper(I) state formed during the redox cycle, ensuring catalytic turnover without the accumulation of inactive metal species. This oxidative dimerization is highly selective, favoring the formation of the 2-indole-3-oxoindoline core over potential polymerization side products. The choice of HFIP as a solvent is not merely for solubility; its unique hydrogen-bonding donor capability stabilizes cationic intermediates and enhances the electrophilicity of the copper-alkyne complex, thereby accelerating the reaction rate.
From an impurity control perspective, the robustness of this catalytic system is evident in its tolerance to various electronic environments. The patent data explicitly notes that the addition of external peroxides, such as m-CPBA or TBHP, is detrimental to the yield, suggesting that the controlled generation of reactive oxygen species by the copper-oxygen complex is key to selectivity. Uncontrolled oxidation can lead to over-oxidation of the indole nitrogen or degradation of the sensitive alkene linkages. By strictly adhering to the oxygen atmosphere protocol without exogenous peroxides, manufacturers can achieve a cleaner crude reaction profile, simplifying downstream purification. This mechanistic understanding allows process chemists to fine-tune parameters such as oxygen pressure and catalyst loading to minimize trace impurities, ensuring that the final API intermediate meets stringent regulatory specifications for heavy metals and organic volatiles.
How to Synthesize 2-Indole-3-Oxoindoline Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and reproducibility. The standard protocol involves charging a reaction vessel with the 2-alkynylaniline substrate, a catalytic load of copper(II) acetate monohydrate, and HFIP solvent. The mixture is then sealed and heated under an oxygen atmosphere, typically at 80°C for around 20 hours. Workup procedures are straightforward, involving filtration and silica gel chromatography to isolate the yellow solid product. While the general procedure is robust, slight modifications may be necessary for substrates with extreme steric hindrance or electronic deactivation. For detailed operational specifics regarding stoichiometry, temperature ramps, and purification gradients, please refer to the standardized synthesis guide below.
- Mix 2-alkynylaniline compound, divalent copper salt catalyst (e.g., Cu(OAc)2·H2O), and HFIP solvent in a reaction vessel.
- Seal the vessel and establish an oxygen-containing atmosphere (air or pure O2) to facilitate oxidative cyclization.
- Heat the mixture to 60-100°C (optimally 80°C) for approximately 20 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed dimerization technology offers tangible strategic benefits beyond mere chemical elegance. The shift from precious metal catalysis to base metal copper represents a significant cost reduction in pharmaceutical intermediate manufacturing, as copper salts are orders of magnitude cheaper than palladium or platinum counterparts. Additionally, the elimination of stoichiometric oxidants and complex protecting group strategies simplifies the bill of materials, reducing the dependency on volatile specialty chemical markets. The mild reaction conditions also translate to lower energy costs and reduced wear on reactor equipment, contributing to a more sustainable and economically viable production model. These factors combined enhance the overall margin potential for high-volume campaigns.
- Cost Reduction in Manufacturing: The utilization of abundant and inexpensive copper catalysts instead of scarce precious metals fundamentally alters the cost structure of the synthesis. By avoiding expensive ligands and stoichiometric oxidants, the direct material costs are substantially lowered. Furthermore, the high atom economy of the dimerization reaction means that a greater proportion of the starting mass is incorporated into the final product, minimizing waste disposal fees and maximizing resource efficiency. This economic efficiency is crucial for maintaining competitiveness in the generic pharmaceutical market where price pressure is intense.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-alkynylanilines, are readily accessible from established chemical suppliers, mitigating the risk of raw material shortages. The robustness of the reaction conditions allows for flexibility in sourcing, as the process is not overly sensitive to minor variations in reagent quality. This reliability ensures consistent production schedules and reduces the likelihood of batch failures that could disrupt the supply of critical API intermediates to downstream customers. The simplified workflow also shortens the manufacturing cycle time, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the use of standard equipment and safe operating temperatures. The absence of hazardous reagents like peroxides reduces the safety risks associated with scale-up, lowering insurance and compliance costs. Moreover, the generation of water as the primary byproduct aligns with increasingly strict environmental regulations regarding solvent waste and emissions. This green profile makes the technology attractive for facilities aiming to reduce their carbon footprint and meet corporate sustainability goals without sacrificing output quality.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and scope studies presented in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances helps in troubleshooting potential issues during technology transfer and ensures that the full potential of the methodology is realized in a production environment.
Q: What is the optimal catalyst system for this dimerization?
A: The patent data indicates that Copper(II) acetate monohydrate (Cu(OAc)2·H2O) in hexafluoroisopropanol (HFIP) under an oxygen atmosphere provides the highest yields, significantly outperforming anhydrous copper salts or other solvents like DCE or THF.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the methodology demonstrates broad substrate scope, successfully accommodating electron-donating groups (methyl, methoxy) and electron-withdrawing groups (chloro, bromo, fluoro) at various positions on the phenyl rings without compromising the structural integrity of the indole core.
Q: Why are peroxides avoided in this reaction protocol?
A: Experimental results show that adding external peroxides like m-CPBA or TBHP adversely affects the reaction yield. The molecular oxygen serves as the sufficient terminal oxidant, making the process greener and more atom-economical by avoiding unnecessary oxidant additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Indole-3-Oxoindoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed dimerization technology for the pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-indole-3-oxoindoline intermediate meets the highest international standards. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific application needs.
We invite you to engage with our technical procurement team to discuss how this innovative method can drive value for your projects. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering high-quality chemical solutions efficiently.
