Advanced Carboprost Manufacturing: High-Yield NHS Esterification Strategy for Commercial Scale-up
Advanced Carboprost Manufacturing: High-Yield NHS Esterification Strategy for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for critical hormonal intermediates, particularly for uterotonic agents like Carboprost. A groundbreaking approach detailed in patent CN113548993B introduces a novel preparation method that fundamentally shifts the purification paradigm from chromatographic separation to crystallization-driven isolation. This technology addresses the longstanding challenges of low combined yields and complex post-processing associated with conventional Carboprost synthesis. By leveraging N-hydroxysuccinimide (NHS) activation, the process achieves exceptional purity levels exceeding 99.9% while drastically simplifying the operational workflow. For global supply chain leaders, this represents a pivotal advancement in securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials with consistent batch-to-batch reproducibility.
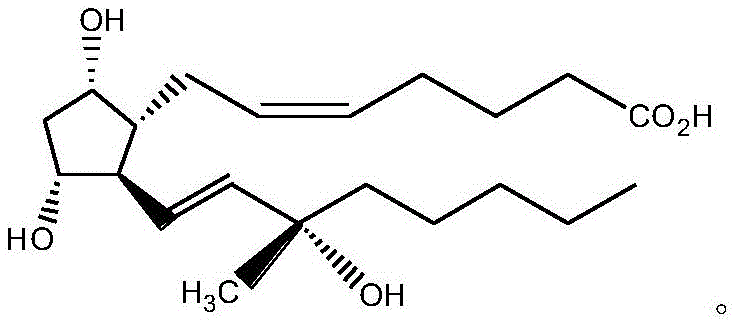
The structural complexity of Carboprost, characterized by multiple chiral centers and sensitive functional groups, traditionally necessitates rigorous purification to remove specific impurities such as the 15-position chiral isomer and the 5,6-position trans-isomer. The new methodology described in the patent circumvents these issues by converting the crude acid into an active NHS ester, which exhibits superior crystallization properties compared to the free acid or methyl ester forms. This strategic modification not only enhances the physical separation of impurities but also stabilizes the molecule during processing. Consequently, manufacturers can achieve high-purity Carboprost suitable for downstream salt formation (e.g., Carboprost Tromethamine) without the prohibitive costs associated with silica gel chromatography.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Carboprost has been hindered by the reliance on column chromatography for purification, a technique that is notoriously difficult to scale and economically inefficient for large-volume manufacturing. Conventional routes often result in synthetic products contaminated with stereoisomers that possess different pharmacological profiles and toxicity risks, necessitating expensive and time-consuming separation processes. Prior art, such as the methods reported by Earl L. Copper, utilized ordinary silica gel chromatography which failed to deliver satisfactory purity and yield simultaneously. Furthermore, existing patents like CN201110153194.5, while improving purity, still depended on silica gel columns for the ester intermediate, leading to significant solvent consumption and low overall recovery rates. These bottlenecks create substantial supply chain vulnerabilities, increasing lead times and limiting the ability to meet surging global demand for oxytocic drugs.
The Novel Approach
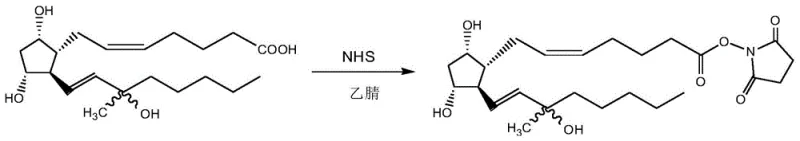
In stark contrast, the innovative route disclosed in CN113548993B employs a chemical activation strategy that transforms the purification challenge into a physical crystallization opportunity. By reacting crude Carboprost with N,N'-carbonyldiimidazole (CDI) and subsequently with NHS in a polar aprotic solvent, the process generates a Carboprost-NHS ester that can be readily purified through simple stirring and cooling in a specific solvent system. This eliminates the need for column chromatography entirely, replacing it with a scalable filtration and washing protocol. The subsequent hydrolysis of this purified ester yields the final Carboprost acid with remarkable efficiency. This shift from chromatographic to crystallization-based purification is a game-changer for cost reduction in API manufacturing, as it significantly reduces solvent usage, waste generation, and processing time while boosting the combined yield to nearly quantitative levels.
Mechanistic Insights into NHS-Mediated Activation and Crystallization
The core of this technological breakthrough lies in the selective formation and stabilization of the N-hydroxysuccinimide ester intermediate. The reaction initiates with the activation of the carboxylic acid group on the Carboprost side chain using CDI, forming a reactive acyl imidazole species. This intermediate rapidly reacts with NHS to form the stable NHS ester, a transformation that is crucial for the subsequent purification step. The NHS moiety introduces specific intermolecular interactions that facilitate the formation of a well-defined crystal lattice, allowing the desired isomer to precipitate while leaving impurities such as the 15-epimer and trans-isomers in the mother liquor. This mechanism effectively leverages thermodynamic stability to drive purity, a principle that is far more robust and scalable than kinetic separation methods like chromatography.
Furthermore, the hydrolysis step is meticulously optimized to ensure the integrity of the sensitive prostaglandin backbone is maintained while cleaving the NHS group. Conducted in a polar aprotic solvent with an alkaline solution, the hydrolysis proceeds under mild conditions that prevent epimerization or degradation of the double bonds. The use of specific solvent pairs, such as acetonitrile for reaction and methyl tert-butyl ether for extraction, ensures high recovery rates. This mechanistic understanding allows for precise control over the impurity profile, ensuring that the final product meets stringent regulatory standards for chiral drugs. The ability to convert potential impurities back to the active isomer or separate them efficiently during the crystallization phase underscores the sophistication of this commercial scale-up of complex prostaglandins.
How to Synthesize Carboprost Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage pathway that is amenable to industrial implementation. The process begins with the activation of the crude starting material, followed by a critical crystallization step that defines the purity of the intermediate, and concludes with a straightforward hydrolysis to release the final active pharmaceutical ingredient. Detailed operational parameters, including solvent ratios, temperatures, and reaction times, are optimized to maximize yield and minimize impurity carryover. For technical teams evaluating this route, the following guide summarizes the standardized synthesis steps derived from the patent examples.
- Activate crude Carboprost using CDI in a polar aprotic solvent, followed by reaction with N-hydroxysuccinimide (NHS) to form the active ester intermediate.
- Purify the Carboprost-NHS ester through controlled crystallization using a solvent system such as methyl tert-butyl ether and cyclohexane, eliminating chiral and trans-isomer impurities.
- Hydrolyze the purified ester intermediate using an alkaline solution (e.g., NaOH) in a polar solvent, followed by acidification and extraction to isolate high-purity Carboprost.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHS-based synthesis route offers compelling economic and logistical benefits that extend beyond mere technical superiority. The elimination of column chromatography translates directly into a drastic reduction in operating expenses, as silica gel and the vast quantities of elution solvents required for traditional purification are no longer needed. This simplification of the downstream processing workflow reduces the burden on waste treatment facilities and lowers the overall environmental footprint of the manufacturing site. Moreover, the high combined yield reported in the patent examples implies that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS) and enhancing margin potential for the final drug product.
- Cost Reduction in Manufacturing: The transition from chromatographic purification to crystallization represents a significant leap in process efficiency. By removing the need for expensive silica gel columns and reducing solvent consumption by a substantial margin, the overall production cost is significantly lowered. The simplified equipment requirements mean that existing reactor infrastructure can be utilized more effectively without the need for specialized chromatography skids, leading to immediate capital expenditure savings and reduced maintenance overheads.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent product quality, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers. The high yield and purity reduce the risk of batch failures and reprocessing, thereby stabilizing production schedules. This reliability is essential for meeting the rigorous demands of the pharmaceutical market, where delays in intermediate supply can impact the availability of life-saving medications. Partnering with a supplier utilizing this technology ensures a steady flow of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling a crystallization process is inherently more predictable and manageable than scaling chromatography, which often suffers from band broadening and resolution loss at larger volumes. This method facilitates a smooth transition from pilot plant to full commercial production, ensuring that supply can grow in tandem with market demand. Additionally, the reduction in hazardous solvent waste aligns with increasingly strict environmental regulations, mitigating compliance risks and supporting sustainable manufacturing initiatives within the organization.
Frequently Asked Questions (FAQ)
To assist technical and commercial stakeholders in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and purification of Carboprost using this novel method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the process capabilities and limitations.
Q: How does the NHS esterification method improve Carboprost purity compared to traditional chromatography?
A: The NHS esterification method allows for purification via crystallization rather than column chromatography. This physical separation effectively removes the 15-chiral isomer and 5,6-trans isomer impurities, achieving purity levels up to 99.97% as demonstrated in patent CN113548993B.
Q: What are the key solvents used in this novel Carboprost synthesis route?
A: The process utilizes polar aprotic solvents such as acetonitrile, DMF, or THF for the activation and hydrolysis steps. For the critical crystallization purification step, a mixture of methyl tert-butyl ether (MTBE) and cyclohexane or methylcyclohexane is employed to precipitate the pure ester intermediate.
Q: Is this method suitable for large-scale commercial production of prostaglandin intermediates?
A: Yes, the method is highly scalable because it replaces labor-intensive and solvent-heavy column chromatography with efficient crystallization and hydrolysis steps. This simplifies post-processing, reduces equipment requirements, and significantly lowers production costs, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboprost Supplier
The technological advancements presented in patent CN113548993B highlight the immense potential for optimizing the production of critical hormonal intermediates. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such innovations, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex chemistries like NHS activation and controlled crystallization, ensuring that we can deliver Carboprost and related intermediates with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify every batch, guaranteeing that our clients receive materials that meet the highest global standards for safety and efficacy.
We invite pharmaceutical companies and contract manufacturers to explore how this advanced synthesis route can enhance their supply chain resilience and cost efficiency. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our commitment to innovation can support your long-term strategic goals in the competitive landscape of reproductive health therapeutics.
