Scalable Green Synthesis of Aurone Intermediates Using Carbon Dioxide and Palladium Catalysis
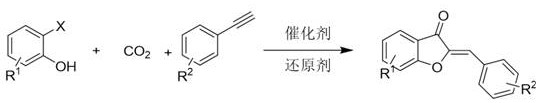
The chemical industry is currently witnessing a paradigm shift towards sustainable C1 chemistry, driven by the urgent need to utilize greenhouse gases as valuable feedstocks. Patent CN116003360A introduces a groundbreaking methodology for the synthesis of aurone compounds, also known as orange ketones, which are pivotal scaffolds in medicinal chemistry and agrochemical development. This innovation leverages carbon dioxide (CO2) as a renewable carbonyl source, reacting it with alkynes and 2-halophenols under palladium catalysis to construct the benzofuranone core efficiently. Unlike traditional extraction methods from natural sources which suffer from seasonal variability and low yields, or conventional synthetic routes relying on hazardous carbon monoxide, this patented process offers a robust, green alternative. The technology addresses critical pain points in the supply chain of high-purity pharmaceutical intermediates by providing a route that is not only environmentally benign but also economically superior due to the abundance of CO2. For R&D directors and procurement managers seeking reliable aurone intermediates supplier partnerships, understanding the mechanistic depth and commercial viability of this CO2-fixation strategy is essential for future-proofing their manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of aurone derivatives has been fraught with significant technical and economic challenges that hinder large-scale commercialization. The most traditional approach involves direct extraction from plant materials such as flowers and fruits, a method that is inherently limited by agricultural cycles, geographical constraints, and extremely low isolation yields, making it unsuitable for consistent industrial supply. Alternatively, synthetic chemists have relied on the cyclization of chalcones, a process that often requires harsh acidic or basic conditions, leading to poor atom economy and the generation of substantial chemical waste. Furthermore, transition metal-catalyzed carbonylation using carbon monoxide (CO) or formic acid has been explored; however, these methods introduce severe safety hazards due to the extreme toxicity of CO gas and the corrosive nature of formic acid. These legacy processes necessitate specialized, expensive containment infrastructure and rigorous safety protocols, which drastically inflate the capital expenditure and operational costs for manufacturing facilities. Consequently, the industry has long sought a safer, more efficient carbonyl source that does not compromise on reaction selectivity or product purity.
The Novel Approach
The methodology disclosed in patent CN116003360A represents a transformative leap forward by substituting toxic carbonyl sources with inert and abundant carbon dioxide. This novel approach utilizes a synergistic palladium catalyst system combined with phosphine ligands and a reducing agent to activate the thermodynamically stable CO2 molecule. By reacting CO2 directly with 2-halophenols and terminal or internal alkynes, the process achieves high regioselectivity and stereoselectivity in a single step, bypassing the need for pre-functionalized chalcone intermediates. The reaction conditions are remarkably mild, operating effectively at temperatures between 25°C and 200°C and CO2 pressures ranging from 1 to 30 bar, which are easily manageable in standard high-pressure reactors. This shift not only mitigates the environmental footprint by sequestering CO2 but also simplifies the engineering requirements for production plants. The result is a streamlined synthesis of complex aurone structures with excellent yields, providing a scalable solution for cost reduction in pharmaceutical intermediates manufacturing while adhering to increasingly stringent global environmental regulations.
Mechanistic Insights into Pd-Catalyzed CO2 Carbonylation and Cyclization
At the heart of this innovative synthesis lies a sophisticated palladium-catalyzed cycle that orchestrates the activation of CO2 and the subsequent construction of the heterocyclic ring. The mechanism initiates with the oxidative addition of the 2-halophenol substrate to the active Pd(0) species, generating an aryl-palladium(II) intermediate. This organometallic species then undergoes a crucial migratory insertion of carbon dioxide into the palladium-carbon bond, forming a carboxylate-palladium complex. This step is energetically demanding due to the kinetic stability of CO2, but the specific choice of ligands, such as bidentate phosphines like DPPF or XantPhos, stabilizes the transition state and facilitates this insertion. Following CO2 incorporation, the alkyne component coordinates to the metal center and inserts into the Pd-O or Pd-C bond, depending on the specific pathway, setting the stage for ring closure. The final step involves an intramolecular cyclization where the phenolic oxygen attacks the activated carbonyl or alkyne carbon, followed by reductive elimination to release the aurone product and regenerate the Pd(0) catalyst. The presence of a reducing agent, such as polymethylhydrosiloxane (PMHS) or hydrogen gas, is critical for maintaining the catalytic cycle by reducing any Pd(II) species back to the active Pd(0) state, ensuring high turnover numbers and sustained reaction efficiency throughout the process.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over random cyclization methods. The directed nature of the palladium coordination ensures that the carbonyl group is inserted specifically at the ortho-position relative to the hydroxyl group, minimizing the formation of regioisomers that are common in acid-catalyzed condensations. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups often present on the alkyne or phenol rings, such as nitro, cyano, or halogen substituents, which might otherwise decompose under harsher thermal or acidic conditions. The high chemoselectivity of the catalyst system means that side reactions like homocoupling of alkynes or dehalogenation of the phenol are significantly suppressed. This intrinsic selectivity translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification processes like chromatography or recrystallization. For quality control teams, this implies a more consistent impurity profile and higher batch-to-batch reproducibility, which are critical parameters for regulatory compliance in the production of high-purity OLED material or API precursors.
How to Synthesize Aurone Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must carefully manage the interplay between catalyst loading, gas pressure, and reducing agent stoichiometry. The patent outlines a robust protocol where the palladium source, ligand, base, and substrates are combined in an organic solvent such as THF, toluene, or DMF under an inert nitrogen atmosphere. The key operational parameter is the introduction of CO2 gas, which must be pressurized to the optimal range of 1 to 30 bar to drive the equilibrium towards carbonylation without requiring excessive energy input. The reaction mixture is then heated, typically between 80°C and 120°C for most substrates, and stirred for a duration of 1 to 36 hours depending on the steric bulk of the reactants. Upon completion, the system is cooled, and the pressure is safely vented before workup. The detailed standardized synthesis steps see the guide below.
- Load the reactor with palladium catalyst, ligand, base, 2-halophenol, alkyne, reducing agent, and organic solvent under inert atmosphere.
- Pressurize the sealed vessel with carbon dioxide gas to 1-30 bar and heat the mixture to 25-200°C with stirring.
- After reaction completion (1-36 hours), cool to room temperature, release pressure, and isolate the aurone product via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CO2-based synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of raw material sourcing and hazard management. By replacing toxic carbon monoxide cylinders or corrosive formic acid with compressed CO2, facilities can eliminate the need for specialized gas detection systems and expensive corrosion-resistant reactors, leading to significant capital expenditure savings. Moreover, CO2 is an industrial byproduct available in vast quantities at a fraction of the cost of specialty carbonyl reagents, insulating the supply chain from price volatility associated with petrochemical-derived feedstocks. This stability in raw material costs allows for more accurate long-term budgeting and pricing strategies for downstream clients. Additionally, the use of commercially available palladium catalysts and ligands ensures that the supply chain remains resilient, as these reagents are produced by multiple global vendors, reducing the risk of single-source bottlenecks that often plague niche synthetic routes.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive and hazardous reagents. Traditional carbonylation methods often require costly safety infrastructure and waste disposal protocols for toxic gases, which add hidden overheads to the final product cost. In contrast, the CO2 fixation method utilizes a benign gas that requires minimal containment investment. Furthermore, the high selectivity of the reaction reduces the consumption of solvents and silica gel during purification, as fewer byproducts are generated. The ability to use simple silanes or hydrogen as reductants, rather than complex hydride sources, further lowers the reagent bill of materials. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality or purity of the final aurone intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical and agrochemical manufacturers, and this process enhances reliability by decoupling production from agricultural harvests or volatile petrochemical markets. Since the key carbon source is CO2, which can be captured from various industrial streams, the risk of raw material shortage is virtually non-existent. The robustness of the catalytic system also means that the process is less sensitive to minor fluctuations in reaction conditions, leading to higher success rates in production batches. This consistency ensures that delivery schedules can be met with greater confidence, reducing the lead time for high-purity pharmaceutical intermediates. For global buyers, this translates to a more dependable partner capable of scaling production up or down in response to market demand without the logistical nightmares associated with hazardous material transport.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on carbon emissions and chemical waste, this synthesis method positions manufacturers as leaders in green chemistry. The process inherently consumes CO2, contributing to carbon neutrality goals, and generates minimal hazardous waste compared to stoichiometric oxidation or chlorination routes. The mild reaction conditions allow for easier scale-up from gram to ton scale without the exponential increase in safety risks seen with high-energy reactions. This scalability ensures that the technology can meet the demands of commercial scale-up of complex polymer additives or drug substances. Furthermore, the reduced environmental footprint simplifies the permitting process for new manufacturing lines, accelerating time-to-market for new products and enhancing the corporate sustainability profile of the supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2-mediated aurone synthesis. These insights are derived directly from the experimental data and claims within patent CN116003360A, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The answers reflect the current state of the art as described in the intellectual property documentation.
Q: What are the advantages of using CO2 over CO for aurone synthesis?
A: Using CO2 eliminates the need for highly toxic carbon monoxide gas, significantly improving operational safety and reducing equipment corrosion risks associated with traditional carbonylation methods.
Q: What is the substrate scope for this palladium-catalyzed reaction?
A: The method demonstrates excellent functional group tolerance, accommodating halogens, trifluoromethyl groups, nitro groups, methoxy groups, and various alkyl substituents on both the phenol and alkyne components.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and readily available raw materials like CO2 and operates under relatively mild conditions with high selectivity, making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aurone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the CO2-fixation technology described in CN116003360A for the next generation of fine chemical synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle high-pressure gas reactions safely and efficiently, adhering to stringent purity specifications required by top-tier pharmaceutical and agrochemical clients. With our rigorous QC labs and dedicated process development teams, we guarantee that every batch of aurone intermediates meets the highest standards of quality and consistency, ready for immediate integration into your drug discovery or crop protection pipelines.
We invite you to collaborate with us to leverage this green synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this CO2-based route can optimize your budget. Please contact us today to request specific COA data for our available aurone derivatives and to discuss route feasibility assessments for your target molecules. Let us help you build a more sustainable and cost-effective supply chain for your critical chemical intermediates.
