Scalable Manufacturing of HI-6 Dimesylate via Novel O-Protection Strategy for Global Pharma Supply Chains
Scalable Manufacturing of HI-6 Dimesylate via Novel O-Protection Strategy for Global Pharma Supply Chains
The development of effective antidotes for organophosphorus nerve agents remains a critical priority in global pharmaceutical defense and healthcare sectors. Central to this effort is the efficient and safe production of HI-6, a potent bispyridinium oxime. Patent CN101495453B discloses a groundbreaking process for the manufacture of HI-6 dimethanesulfonate (HI-6 DMS), addressing long-standing challenges associated with toxicity and low yield in traditional synthesis routes. This technical insight report analyzes the proprietary O-protection methodology that transforms the production landscape for this vital active pharmaceutical ingredient (API) intermediate. By shifting from hazardous chlorinating agents to a controlled quaternization of protected aldoximes, the technology offers a robust pathway for reliable pharmaceutical intermediate suppliers aiming to enhance supply chain security.
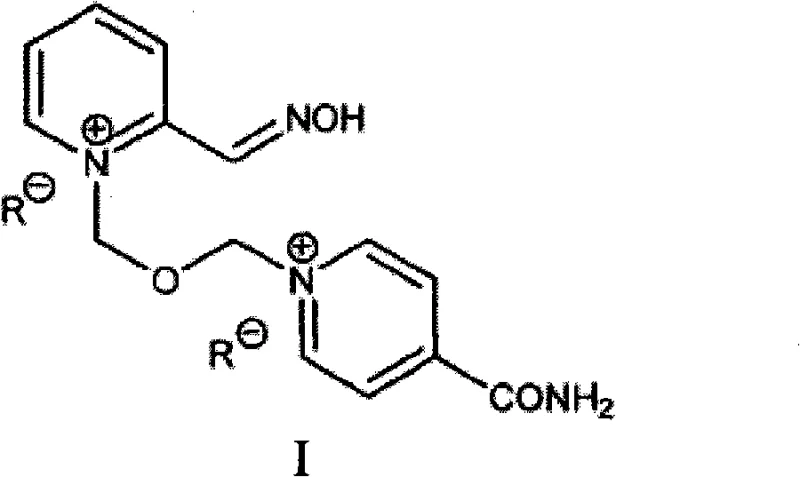
The structural integrity of HI-6, characterized by its bispyridinium oxime salt configuration, is paramount for its efficacy as a cholinesterase reactivator. As illustrated in the general formula, the molecule features two pyridine rings linked by an ether bridge, with oxime functionality essential for nucleophilic attack on phosphorylated enzymes. While the dichloride salt has been historically utilized, the dimethanesulfonate (mesylate) salt offers superior physicochemical properties, including improved stability and solubility profiles. However, accessing this specific salt form with high purity has traditionally been fraught with chemical engineering hurdles, necessitating the innovative approach detailed in the patent data to meet stringent regulatory standards for medical countermeasures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of HI-6 derivatives has relied heavily on the use of bis(chloromethyl)ether as a quaternizing agent to link the pyridine moieties. This reagent is notoriously carcinogenic and highly toxic, posing severe occupational health risks and complicating regulatory compliance for manufacturing facilities. Even when alternative non-chlorinated routes were attempted, such as the direct reaction of pyridine-2-aldoxime (P2A) with bis(methylsulfonyloxymethyl)ether (BSME), the outcomes were commercially unsatisfactory. Literature and prior art indicate that these unprotected routes suffer from extremely low yields, often reported around 11% after multiple recrystallizations. The fundamental chemical issue lies in the formation of stubborn byproducts, specifically isonicotinamide dimers, which exhibit solubility characteristics nearly identical to the desired HI-6 product, making purification via standard crystallization techniques nearly impossible.
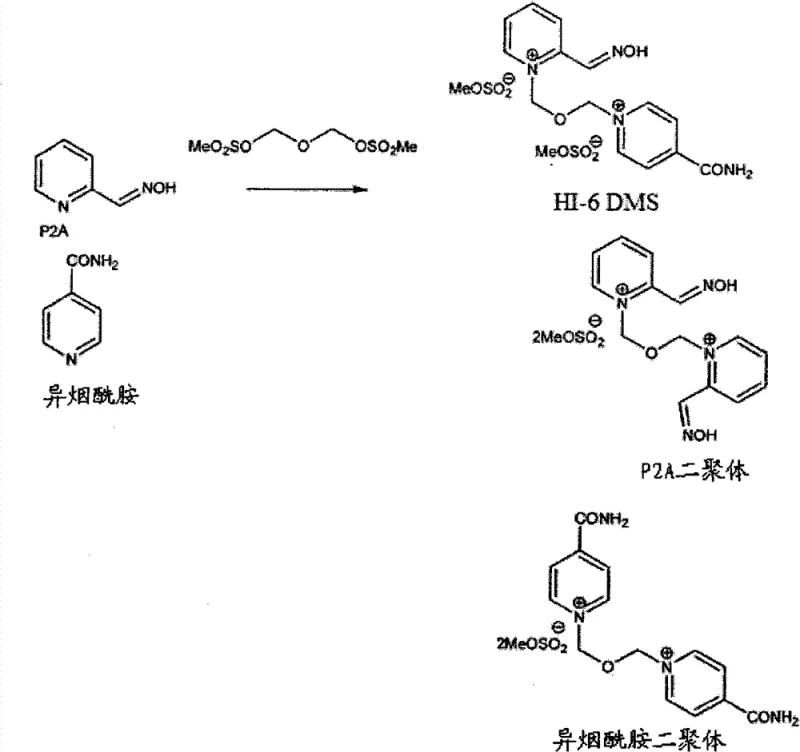
Furthermore, the physical handling of reaction mixtures in conventional unprotected syntheses presents significant operational bottlenecks. The reaction between unprotected P2A and BSME frequently generates tar-like pastes and heterogeneous slurries that require complex decantation steps. On an industrial scale, managing these viscous, semi-solid masses is inefficient and prone to product loss. The inability to effectively separate the desired mono-quaternized intermediate from di-quaternized impurities or unreacted starting materials results in a final product with unacceptable impurity profiles. For procurement managers and supply chain heads, these inefficiencies translate into volatile pricing, inconsistent batch quality, and potential supply disruptions due to the difficulty in scaling such erratic chemistry.
The Novel Approach
The patented process introduces a paradigm shift by employing an O-protected pyridine-2-aldoxime as the starting nucleophile. By masking the oxime hydroxyl group with a protecting group such as an acetyl, ethyl, or silyl moiety, the chemical reactivity and physical properties of the intermediate are fundamentally altered. This strategic modification prevents the formation of the problematic tar-like substances observed in unprotected routes, resulting in homogeneous reaction mixtures that are far easier to stir, pump, and process in large-scale reactors. The O-protection serves a dual purpose: it enhances the nucleophilicity of the pyridine nitrogen for more efficient quaternization and, crucially, modifies the solubility profile of the resulting intermediate species.
This alteration in solubility is the key to breaking the purification deadlock. In the protected state, the unwanted isonicotinamide dimer byproducts can be selectively precipitated or crystallized out of the solution, leaving the desired O-protected HI-6 precursor in the filtrate. This separation step, which is ineffective in the unprotected route, allows for the removal of impurities prior to the final deprotection stage. Consequently, the process achieves reproducible crude yields of 50% to 60%, with final isolated yields of pure HI-6 DMS reaching 35% to 42%. This represents a three-to-four-fold improvement over prior art, providing a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing without compromising on safety or quality.
Mechanistic Insights into O-Protection and Quaternization Chemistry
The success of this synthesis hinges on the careful selection of the protecting group to avoid side reactions while enabling efficient quaternization. Early attempts to protect the aldoxime functionality with groups like benzoyl resulted in beta-elimination reactions during the quaternization step, leading to the destruction of the oxime group and the formation of undesired 2-cyanopyridine derivatives. The patent data highlights that specific O-alkyl or O-acyl groups, such as acetyl or triethylsilyl, are stable under the quaternization conditions employed. The mechanism involves the nucleophilic attack of the pyridine nitrogen on the methylene carbon of the bis(methylsulfonyloxymethyl)ether (BSME), displacing the mesylate leaving group. The presence of the O-protecting group stabilizes the transition state and prevents the electron withdrawal that typically facilitates beta-elimination in other protected variants.
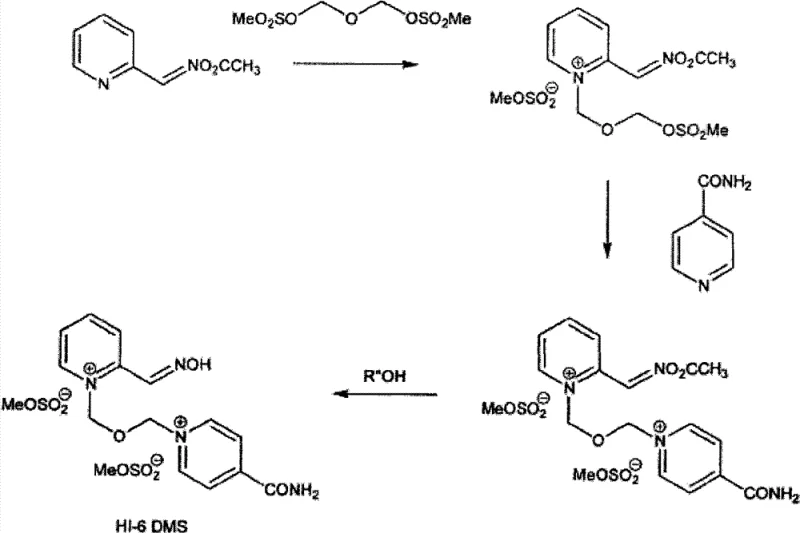
Following the initial quaternization, the second pyridine ring (isonicotinamide) is introduced. The O-protected intermediate reacts with isonicotinamide to form the bis-quaternary salt. At this stage, the molecule possesses the full carbon skeleton of HI-6 but retains the protecting group on the oxime. The critical mechanistic advantage here is that the protecting group imparts sufficient lipophilicity or specific polarity to the molecule, differentiating it from the polar dimeric impurities. When the reaction mixture is treated with specific solvent systems, such as aqueous alcohols, the impurities crystallize out while the protected product remains in solution. The final step involves a mild deprotection, typically using protic solvents like ethanol or water, sometimes catalyzed by bases, to reveal the free oxime hydroxyl group. This final deprotection is clean and high-yielding because the bulk of the impurities have already been removed, ensuring the final API intermediate meets rigorous purity specifications.
How to Synthesize HI-6 Dimesylate Efficiently
The implementation of this synthesis route requires precise control over reaction stoichiometry and solvent selection to maximize the benefits of the O-protection strategy. The process begins with the preparation of the O-protected aldoxime, followed by a sequential quaternization protocol that avoids the pitfalls of one-pot unprotected reactions. Detailed operational parameters, including temperature controls and specific solvent ratios, are critical to preventing the formation of tars and ensuring the solubility differential required for purification is maintained. For R&D teams looking to adopt this technology, understanding the interplay between the protecting group stability and the quaternization kinetics is essential for successful technology transfer.
- Quaternization of O-protected pyridine-2-aldoxime with bis(methylsulfonyloxymethyl)ether (BSME) in a suitable solvent to form an intermediate.
- Reaction of the intermediate with isonicotinamide to effect quaternization and form the O-protected HI-6 product precursor.
- Separation of impurities followed by deprotection of the precursor to yield pure HI-6 dimethanesulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this O-protected synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary driver for adoption is the substantial enhancement in process reliability and yield efficiency. By eliminating the need for hazardous bis(chloromethyl)ether, manufacturers remove a significant regulatory burden and liability risk associated with the storage and handling of carcinogenic materials. This simplification of the safety protocol reduces the overhead costs related to specialized containment equipment and hazardous waste disposal, contributing to a leaner cost structure. Furthermore, the dramatic improvement in yield—from roughly 11% in legacy processes to over 35% in this new method—directly correlates to a reduction in raw material consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The economic impact of the yield improvement cannot be overstated. Tripling the output from the same amount of starting materials effectively lowers the variable cost of goods sold (COGS). Additionally, the ability to purify the intermediate before deprotection reduces the number of recrystallization cycles required, saving on solvent usage and energy consumption. The elimination of expensive and difficult-to-remove impurities means less product is lost to mother liquors during final purification. This efficiency gain allows for more competitive pricing strategies in the global market for nerve agent antidotes, making the supply of this critical medicine more sustainable.
- Enhanced Supply Chain Reliability: Conventional methods often suffer from batch-to-batch variability due to the unpredictable nature of tar formation and difficult filtrations. The new process produces consistent, free-flowing solids that are amenable to standard industrial filtration and drying equipment. This predictability ensures that production schedules can be met with high confidence, reducing the risk of stockouts for emergency medical stockpiles. The use of readily available reagents like BSME and common protecting group precursors further secures the supply chain against raw material shortages, ensuring continuity of supply for downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior. The avoidance of carcinogens aligns with modern green chemistry principles and strict environmental regulations. The process generates less hazardous waste, and the solvents used (such as acetonitrile and ethanol) are easier to recover and recycle compared to the complex waste streams generated by chlorinating agents. This environmental compatibility facilitates easier permitting for manufacturing sites and supports the corporate sustainability goals of major pharmaceutical buyers who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of HI-6 dimethanesulfonate using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this route represents the current state-of-the-art in antidote manufacturing.
Q: Why is the O-protection strategy superior to conventional HI-6 synthesis?
A: Conventional methods often utilize highly toxic bis(chloromethyl)ether or result in low yields (around 11%) due to difficult purification. The O-protection strategy alters the solubility profile of intermediates, allowing for the effective removal of isonicotinamide dimer impurities before deprotection, thereby significantly increasing yield to over 35% and ensuring higher purity.
Q: What are the safety advantages of this new manufacturing process?
A: The primary safety advantage is the complete elimination of bis(chloromethyl)ether, a known carcinogen, from the synthesis route. Additionally, the process avoids the formation of tarry byproducts common in unprotected routes, reducing operational hazards and simplifying waste management.
Q: Can this process be scaled for industrial production?
A: Yes, the process is specifically designed for industrial scalability. Unlike unprotected routes that form difficult-to-handle slurries and tars requiring decantation, the O-protected route maintains better solubility characteristics, facilitating standard filtration and crystallization steps suitable for large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable HI-6 Dimesylate Supplier
As the global demand for effective medical countermeasures continues to evolve, securing a supply of high-purity HI-6 Dimesylate is paramount for national defense and public health initiatives. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the O-protection route to deliver superior quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both pilot-scale research needs and full-scale deployment requirements. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of HI-6 DMS meets the exacting standards required for pharmaceutical applications.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes safety, efficiency, and regulatory compliance. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and reduce total acquisition costs.
