Advanced Synthesis of Biphenyl Triarylamine Compounds for Next-Generation OLED Displays
Advanced Synthesis of Biphenyl Triarylamine Compounds for Next-Generation OLED Displays
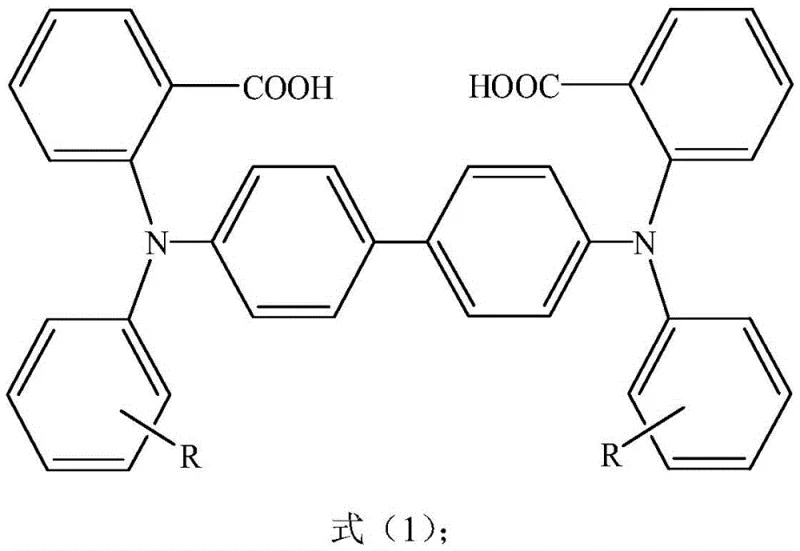
The rapid evolution of the organic light-emitting diode (OLED) industry demands hole transport materials that combine exceptional thermal stability with high charge carrier mobility. Patent CN108276300B introduces a groundbreaking methodology for preparing biphenyl triarylamine compounds by utilizing a carboxyl group as a transient directing group. This technical breakthrough addresses the longstanding challenges of high production costs and harsh reaction conditions associated with traditional synthesis routes. By leveraging a copper-catalyzed Ullmann coupling strategy followed by a controlled decarboxylation step, this process enables the scalable manufacturing of high-purity electronic chemicals. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a pivotal shift towards more economically viable and environmentally sustainable production protocols for advanced display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triarylamine-based hole transport materials has relied heavily on the Buchwald-Hartwig amination or standard Ullmann condensation reactions. The Buchwald-Hartwig protocol, while effective, necessitates the use of expensive palladium catalysts and sophisticated phosphine ligands, which drastically inflates the raw material costs and complicates the removal of trace metal residues—a critical factor for electronic grade purity. Conversely, traditional Ullmann reactions often require excessively high temperatures and prolonged reaction times, leading to lower yields and the formation of complex impurity profiles that are difficult to separate. Furthermore, existing literature methods frequently utilize costly starting materials like N,N'-diphenyl benzidine, which creates a significant bottleneck for industrial scale-up and limits the commercial competitiveness of the final hole transport layer in OLED devices.
The Novel Approach
The innovative strategy disclosed in the patent circumvents these obstacles by employing o-chlorobenzoic acid as a key building block, where the carboxyl moiety serves as an intramolecular guide for the copper catalyst. This directing effect facilitates the formation of the C-N bond under significantly milder conditions compared to non-directed couplings. As illustrated in the reaction scheme below, the process involves a sequential coupling where the carboxyl group ensures high regioselectivity and conversion efficiency before being cleanly removed in a final decarboxylation step. This approach not only utilizes inexpensive copper catalysts but also achieves product purities exceeding 99.5%, demonstrating a superior balance between cost efficiency and performance metrics required for high-end optoelectronic applications.
Mechanistic Insights into Carboxyl-Directed Copper Catalysis
The core mechanistic advantage of this synthesis lies in the coordination chemistry between the ortho-carboxyl group and the copper species during the Ullmann coupling phase. The carboxyl group acts as a bidentate or monodentate ligand that stabilizes the copper center, effectively lowering the energy barrier for the oxidative addition of the aryl halide. This stabilization allows the reaction to proceed efficiently at temperatures ranging from 150°C to 200°C, which is notably lower than the extreme conditions often required for unactivated substrates. The presence of metal carbonates, such as potassium carbonate or cesium carbonate, serves a dual purpose: acting as a base to neutralize the generated acid and potentially participating in the transmetallation step of the catalytic cycle. This synergistic interaction ensures that the substitution pattern on the biphenyl core is established with high fidelity, minimizing the formation of homocoupling byproducts that typically plague copper-mediated aminations.
Following the construction of the triarylamine backbone, the removal of the guiding group is achieved through a thermal decarboxylation mechanism facilitated by cuprous oxide and nitrogen-based ligands like phenanthroline or TMEDA. This step is crucial for restoring the electronic properties of the triarylamine system, as the electron-withdrawing carboxyl group would otherwise detrimentally affect the hole injection capability of the material. The decarboxylation proceeds smoothly in polar aprotic solvents like N-methyl pyrrolidone (NMP) at temperatures between 140°C and 200°C. The rigorous control of this step ensures that the final molecular architecture retains the desired alkyl substituents, which are essential for enhancing solubility in resin matrices and preventing crystallization in the solid state, thereby improving the morphological stability of the hole transport layer in operational devices.
How to Synthesize N,N'-bis(3-methyl)-N,N'-diphenyl-1,1'-biphenyl-4,4'-diamine Efficiently
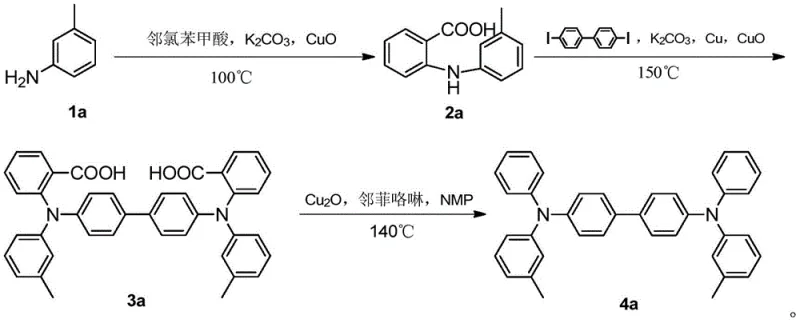
The synthesis of this specific biphenyl triarylamine derivative exemplifies the robustness of the carboxyl-guided methodology, transitioning from simple aniline derivatives to complex hole transport materials through a streamlined three-stage process. The initial stage involves the coupling of 3-methylaniline with o-chlorobenzoic acid in an aqueous medium, highlighting the process's ability to utilize green solvents for the first transformation. The subsequent coupling with 4,4'-diiodobiphenyl builds the central core, and the final decarboxylation yields the target molecule with exceptional purity. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Synthesize substituted diphenylamine by reacting substituted aniline with o-chlorobenzoic acid using a copper catalyst and metal carbonate base at 100-120°C.
- Perform a second Ullmann coupling between the substituted diphenylamine and 4,4'-dihalobiphenyl at 150-200°C to form the carboxyl-containing intermediate.
- Execute a decarboxylation reaction on the intermediate using cuprous oxide and a ligand in NMP solvent at 140-200°C to obtain the final biphenyl triarylamine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed, carboxyl-directed synthesis offers profound economic and logistical benefits over legacy palladium-based routes. The elimination of precious metal catalysts removes a major source of price volatility and supply risk, as copper is abundant and globally available compared to palladium. Furthermore, the ability to use water as a solvent in the initial step significantly reduces the volume of hazardous organic waste generated, simplifying environmental compliance and waste disposal costs. The high yields reported in the patent examples, often exceeding 90% for the intermediate and final steps, translate directly into improved material throughput and reduced raw material consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and specialized ligands with commodity copper powder and oxides results in a drastic reduction in direct material costs. Additionally, the high atom economy of the Ullmann coupling and the efficient recovery of solvents like nitrobenzene and NMP contribute to substantial overall cost savings in electronic chemical manufacturing. The simplified purification process, relying primarily on recrystallization rather than complex chromatography, further lowers the operational expenditure associated with downstream processing.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted anilines and o-chlorobenzoic acid, manufacturers can mitigate the risks associated with specialized reagent shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and bases, ensures consistent production output even with minor variations in raw material quality. This reliability is critical for maintaining continuous supply lines to panel manufacturers who require uninterrupted delivery of high-purity hole transport materials for mass production.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples which describe reactions on a multi-kilogram scale without loss of efficiency. The use of water in the first step and the potential for solvent recycling align with increasingly stringent global environmental regulations regarding volatile organic compound (VOC) emissions. This eco-friendly profile not only reduces regulatory burdens but also enhances the sustainability credentials of the final OLED materials, a growing priority for end-user electronics brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carboxyl-guided synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters and partners in the fine chemical sector.
Q: Why is the carboxyl group used as a guiding group in this synthesis?
A: The ortho-carboxyl group acts as a directing group that coordinates with the copper catalyst, significantly lowering the activation energy for the C-N bond formation. This allows the Ullmann coupling to proceed under milder conditions with higher yields compared to traditional non-directed methods, and it is subsequently removed via decarboxylation.
Q: What are the purity levels achievable with this copper-catalyzed method?
A: The patent data indicates that the final biphenyl triarylamine compounds can achieve purity levels exceeding 99.5% after recrystallization, with reaction yields consistently above 82%, making it highly suitable for electronic grade applications.
Q: How does this method reduce manufacturing costs compared to Buchwald-Hartwig reactions?
A: This method replaces expensive palladium catalysts and complex phosphine ligands with inexpensive and abundant copper powder or copper oxide. Additionally, the use of water as a solvent in the initial step and the recyclability of organic solvents like nitrobenzene further drive down operational expenditures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Triarylamine Supplier
As the demand for high-performance OLED materials continues to surge, partnering with an experienced CDMO capable of executing complex multi-step syntheses is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of biphenyl triarylamine compound meets the exacting standards required for next-generation display technologies.
We invite you to collaborate with our technical team to explore how this innovative carboxyl-directed synthesis can optimize your material costs and supply security. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless transition to this advanced and cost-effective production methodology.
