Advanced Chiral Phosphate Catalysis for Scalable Binaphtholamine and NOBIN Derivative Production
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks essential for modern drug discovery. Patent CN109535018B introduces a groundbreaking methodology for the catalytic synthesis of binaphtholamine derivatives, a critical class of axially chiral molecules serving as precursors to the renowned NOBIN ligands. This technology represents a paradigm shift from traditional oxidative cross-coupling strategies, utilizing a redox-neutral aryl-aryl cross-coupling approach mediated by chiral phosphoric acid or chiral phosphate salts. By leveraging the unique electrophilic properties of azo-aryl substrates, this invention achieves exceptional stereocontrol and atom economy, addressing long-standing challenges in the production of non-C2 symmetric chiral scaffolds. For R&D teams and procurement specialists alike, this patent offers a robust pathway to access complex biaryl architectures with high efficiency and reduced environmental impact.
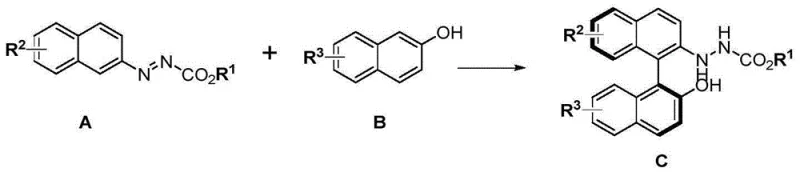
The limitations of conventional methods for synthesizing 1,1'-binaphthyl skeletons have long hindered the widespread adoption of NOBIN-type ligands in industrial applications. Traditional approaches often rely on oxidative cross-coupling, which is plagued by poor enantiospecificity, narrow substrate scope, and the formation of difficult-to-separate homocoupling byproducts. Furthermore, obtaining optically pure NOBIN from BINOL typically requires lengthy synthetic routes with low atom economy, driving up costs and complicating supply chains. In contrast, the novel approach disclosed in this patent circumvents these issues by converting nucleophilic aryl groups into electrophilic species via azo functionalization. This strategic inversion of aromatic polarity allows for a direct, catalytic C-H/C-H cross-coupling that is both redox-neutral and highly enantioselective. The method effectively suppresses side reactions and enables the construction of the chiral axis with precision, laying a solid foundation for the scalable manufacturing of high-value chiral intermediates.
Mechanistic Insights into Chiral Phosphate Catalyzed Cross-Coupling
The core of this technological breakthrough lies in the sophisticated interplay between the chiral phosphate catalyst and Lewis acid additives. The reaction mechanism involves the activation of the azo group on the naphthalene ring, which enhances the electrophilicity of the aromatic system while the chiral phosphate imposes steric hindrance to direct the incoming nucleophile. Extensive screening revealed that simple chiral phosphoric acids (CPA) alone were insufficient, often yielding low conversion and moderate enantioselectivity due to competing background reactions. However, the introduction of specific metal salts, particularly calcium-based chiral phosphates (CPA-Ca), dramatically improved performance. The synergy is further amplified by the addition of soluble Lewis acids like Zn(BarF)2(CH3CN)6, which likely coordinates with the catalyst to create a more rigid and defined chiral pocket. This dual-catalyst system ensures that the transition state is tightly controlled, resulting in enantiomeric excess (ee) values reaching up to 95% for various substrates.
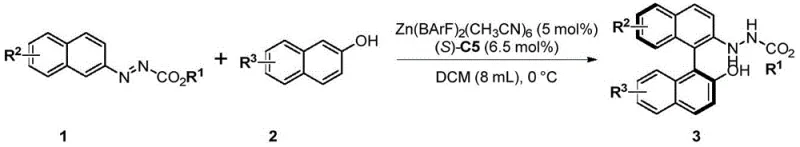
Impurity control is another critical aspect where this mechanism excels. The redox-neutral nature of the reaction eliminates the need for external oxidants, thereby reducing the formation of over-oxidized byproducts common in traditional methods. The specific choice of substituents on both the azo-naphthalene and the 2-naphthol partners plays a pivotal role in the outcome. Electron-withdrawing groups such as cyano, aldehyde, and ester on the 2-naphthol ring were found to maintain high ee values, while bulky groups at specific positions could hinder chiral induction. The process demonstrates remarkable tolerance to a wide range of functional groups, including halogens, alkyls, and alkoxy groups, allowing for the late-stage diversification of the molecular scaffold. This mechanistic robustness ensures that the final product profile is clean, simplifying downstream purification and enhancing the overall viability of the process for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Binaphtholamine Derivatives Efficiently
To implement this advanced synthesis route effectively, precise control over reaction conditions and catalyst preparation is paramount. The patent outlines a standardized protocol that begins with the pretreatment of the catalyst system to ensure maximum activity and stereoselectivity. The detailed operational steps involve specific solvent choices, temperature gradients, and stoichiometric ratios that have been optimized through rigorous experimentation. Following these guidelines allows chemists to reproduce the high yields and enantioselectivity reported in the patent examples consistently. For a comprehensive understanding of the exact procedural nuances required for successful execution, please refer to the standardized synthesis guide below.
- Prepare the catalyst system by mixing Zn(BarF)2(CH3CN)6 and chiral calcium phosphate (CPA-Ca) in DCE at 60°C, followed by solvent removal to activate the species.
- Dissolve the activated catalyst in DCM and cool the solution to 0°C under an inert atmosphere to ensure optimal stereocontrol.
- Add the azo-naphthalene substrate and 2-naphthol derivative sequentially, stirring at 0°C until completion, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this catalytic method offers substantial benefits that directly address the pain points of sourcing high-quality chiral intermediates. The shift from multi-step oxidative resolutions to a direct, one-pot cross-coupling reaction significantly streamlines the manufacturing process. This simplification translates into tangible operational efficiencies, as fewer unit operations mean reduced labor, lower energy consumption, and minimized equipment footprint. For procurement managers, the ability to source precursors that enable such efficient downstream processing is a key factor in reducing the total cost of ownership for chiral ligand projects. The high atom economy of the reaction ensures that raw materials are utilized effectively, minimizing waste generation and aligning with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the reduction of synthetic steps lead to a drastically simplified production workflow. By avoiding the need for optical resolution of racemic mixtures, which typically discards half of the material, this asymmetric catalytic approach maximizes material throughput. The use of commercially available starting materials and the potential for catalyst recycling further contribute to significant cost savings, making the production of high-purity binaphtholamine derivatives economically viable for large-scale applications.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent means that supply chain disruptions for specific substituted naphthols can be mitigated by switching to alternative analogs without re-optimizing the entire process. The robustness of the catalyst system against various functional groups ensures consistent quality even with slight variations in raw material grades. This flexibility enhances supply continuity, allowing manufacturers to maintain steady production schedules and meet the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild, typically operating at or near room temperature or slightly cooled, which reduces the energy load associated with heating or cryogenic cooling. The use of common organic solvents like dichloromethane facilitates easy solvent recovery and recycling. Furthermore, the high selectivity reduces the burden on waste treatment facilities by minimizing the volume of hazardous byproducts. These factors collectively support the commercial scale-up of complex chiral intermediates while adhering to stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this chiral phosphate catalysis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction parameters and product outcomes. Understanding these details is crucial for evaluating the feasibility of this method for specific project requirements.
Q: What are the advantages of this redox-neutral method over traditional oxidative coupling?
A: Unlike traditional oxidative coupling which suffers from poor enantiospecificity and homocoupling byproducts, this redox-neutral strategy utilizes azo-aryl substrates to achieve high atom economy and superior enantiomeric excess (ee) values up to 95%.
Q: Which catalyst system provides the highest enantioselectivity for NOBIN synthesis?
A: The patent data indicates that a combination of chiral calcium phosphate (CPA-Ca) as the primary catalyst and Zn(BarF)2(CH3CN)6 as a Lewis acid additive yields the best results, significantly outperforming simple chiral phosphoric acids or other metal salts.
Q: Can the intermediate hydrazine products be converted to final NOBIN ligands?
A: Yes, the synthesized binaphtholamine intermediates can be efficiently converted to the final axially chiral NOBIN ligands via hydrogenation using Raney-Ni under basic or neutral conditions without loss of optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphtholamine Supplier
The technological potential of chiral phosphate-catalyzed cross-coupling represents a significant opportunity for advancing the synthesis of axially chiral ligands. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of binaphtholamine or NOBIN derivative meets the exacting standards required for asymmetric catalysis in drug manufacturing.
We invite you to explore how our expertise can optimize your supply chain for chiral intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your development goals and reduce lead time for high-purity pharmaceutical intermediates.
