Revolutionizing Benzofuran Production: A One-Step Iron-Catalyzed Strategy for Commercial Scale-Up
Revolutionizing Benzofuran Production: A One-Step Iron-Catalyzed Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex heterocyclic scaffolds, particularly the ubiquitous benzofuran motif found in countless bioactive molecules. A groundbreaking approach detailed in patent CN101747302A introduces a transformative method for synthesizing polysubstituted benzofuran compounds through a direct oxidative coupling strategy. This innovation leverages an inexpensive iron catalyst system to activate industrial-grade phenols, bypassing the need for pre-functionalized substrates that have historically plagued synthetic routes. By shifting the paradigm from multi-step, harsh condition protocols to a streamlined one-step process, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios. The ability to directly utilize simple phenols and beta-diketones not only simplifies the synthetic logic but also significantly reduces the environmental footprint associated with traditional organic synthesis, marking a pivotal advancement in green chemistry applications for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzofuran core has relied on classical cyclization strategies that are often fraught with inefficiencies and operational complexities. As illustrated in the traditional reaction pathways, earlier methods typically required the preparation of specialized precursors, such as ortho-halo phenols or complex enol ethers, which necessitated multiple synthetic steps prior to the final ring closure. 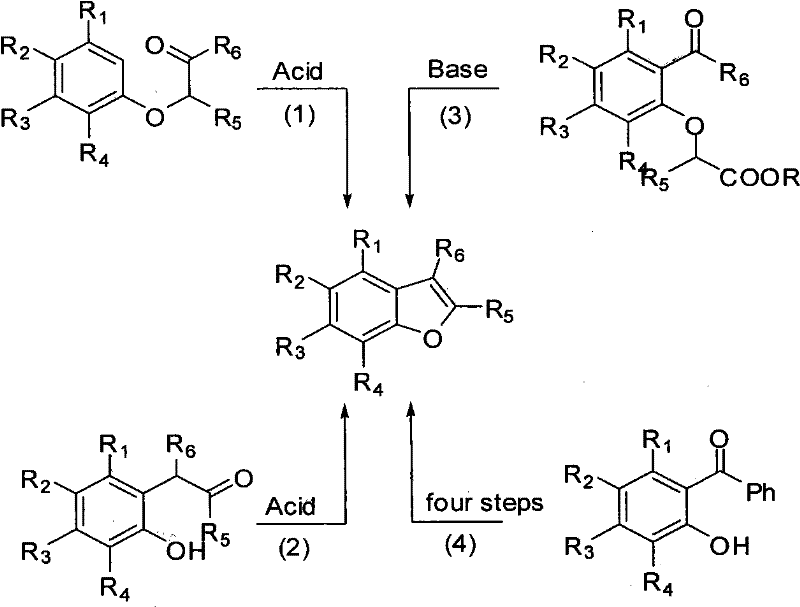 . These conventional approaches frequently demanded harsh reaction conditions, including strong acids or bases, elevated temperatures, and stoichiometric amounts of expensive reagents, leading to poor atom economy and significant waste generation. Furthermore, the reliance on precious metal catalysts in some modern variations introduced severe supply chain vulnerabilities and stringent regulatory hurdles regarding residual metal limits in final drug substances. The cumulative effect of these factors resulted in prolonged lead times, inflated production costs, and a limited scope for structural diversification, creating a bottleneck for R&D teams attempting to access diverse benzofuran libraries for structure-activity relationship studies.
. These conventional approaches frequently demanded harsh reaction conditions, including strong acids or bases, elevated temperatures, and stoichiometric amounts of expensive reagents, leading to poor atom economy and significant waste generation. Furthermore, the reliance on precious metal catalysts in some modern variations introduced severe supply chain vulnerabilities and stringent regulatory hurdles regarding residual metal limits in final drug substances. The cumulative effect of these factors resulted in prolonged lead times, inflated production costs, and a limited scope for structural diversification, creating a bottleneck for R&D teams attempting to access diverse benzofuran libraries for structure-activity relationship studies.
The Novel Approach
In stark contrast to these legacy methods, the novel methodology disclosed in the patent utilizes a direct C-H activation strategy that fundamentally simplifies the synthetic architecture. The core innovation lies in the use of a catalytic iron system combined with a peroxide oxidant to directly couple unactivated phenols with beta-diketones in a single operational step. 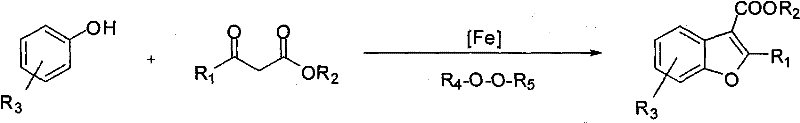 . This approach eliminates the necessity for pre-halogenation or other activating groups on the phenol ring, allowing manufacturers to start directly from commodity chemicals like phenol, cresol, or naphthol. The reaction proceeds under relatively mild reflux conditions in common solvents such as 1,2-dichloroethane or toluene, demonstrating remarkable tolerance for various functional groups including halogens, alkyl chains, and alkoxy substituents. By condensing what was previously a multi-stage sequence into a single pot operation, this technology drastically reduces the overall process mass intensity (PMI) and enhances the throughput capacity of production facilities, positioning it as an ideal solution for cost reduction in pharmaceutical intermediate manufacturing where efficiency is paramount.
. This approach eliminates the necessity for pre-halogenation or other activating groups on the phenol ring, allowing manufacturers to start directly from commodity chemicals like phenol, cresol, or naphthol. The reaction proceeds under relatively mild reflux conditions in common solvents such as 1,2-dichloroethane or toluene, demonstrating remarkable tolerance for various functional groups including halogens, alkyl chains, and alkoxy substituents. By condensing what was previously a multi-stage sequence into a single pot operation, this technology drastically reduces the overall process mass intensity (PMI) and enhances the throughput capacity of production facilities, positioning it as an ideal solution for cost reduction in pharmaceutical intermediate manufacturing where efficiency is paramount.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The mechanistic elegance of this transformation centers on the ability of the iron catalyst to mediate a radical oxidative coupling process that selectively functionalizes the ortho-position of the phenol ring. The proposed cycle involves the generation of reactive radical species from the beta-diketone substrate via hydrogen abstraction by the iron-peroxide complex, which then attacks the electron-rich aromatic ring of the phenol. This initial C-C bond formation is followed by an intramolecular cyclization and subsequent oxidative aromatization to yield the stable benzofuran skeleton. The use of iron salts, such as FeCl3·6H2O or FeCl2, is particularly advantageous due to their Lewis acidic nature which likely assists in coordinating the carbonyl oxygen of the diketone, thereby lowering the activation energy for the nucleophilic attack. This dual role of the iron species as both a redox mediator and a Lewis acid catalyst ensures high regioselectivity, minimizing the formation of unwanted by-products such as para-coupled isomers or polymerized tars. For R&D directors focused on purity profiles, this inherent selectivity translates to cleaner reaction mixtures that require less aggressive purification protocols, ultimately preserving yield and reducing solvent consumption during the isolation phase.
Furthermore, the robustness of this catalytic system is evidenced by its compatibility with a wide array of substrates, as demonstrated by the successful synthesis of various derivatives shown in the experimental examples. For instance, the reaction of phenol with ethyl benzoylacetate proceeds smoothly to afford the corresponding 2-phenyl-3-ethoxycarbonylbenzofuran in high yield, showcasing the method's efficacy with standard aromatic systems. 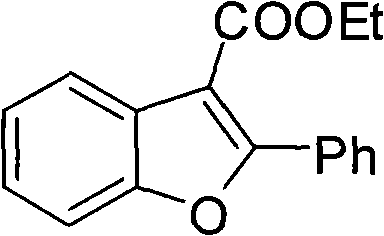 . The mechanism tolerates electron-donating groups like methyl and benzyl, as well as electron-withdrawing halogens, without significant erosion of efficiency, indicating a versatile electronic window for substrate engagement. Impurity control is further enhanced by the choice of oxidant; peroxides like tert-butyl peroxide decompose into volatile by-products that are easily removed during workup, avoiding the contamination issues associated with heavy metal oxidants. This level of mechanistic control provides a solid foundation for scaling the process, as the reaction kinetics are predictable and the catalyst loading can be optimized to balance cost against conversion rates, ensuring consistent quality across different batch sizes.
. The mechanism tolerates electron-donating groups like methyl and benzyl, as well as electron-withdrawing halogens, without significant erosion of efficiency, indicating a versatile electronic window for substrate engagement. Impurity control is further enhanced by the choice of oxidant; peroxides like tert-butyl peroxide decompose into volatile by-products that are easily removed during workup, avoiding the contamination issues associated with heavy metal oxidants. This level of mechanistic control provides a solid foundation for scaling the process, as the reaction kinetics are predictable and the catalyst loading can be optimized to balance cost against conversion rates, ensuring consistent quality across different batch sizes.
How to Synthesize Polysubstituted Benzofurans Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize the benefits of the iron-catalyzed system. The general protocol involves dissolving the phenol and beta-diketone starting materials in a chlorinated or aromatic solvent, followed by the sequential addition of the iron catalyst and the peroxide oxidant under an inert atmosphere or even open to air depending on the specific substrate sensitivity. The mixture is then heated to reflux, typically around 80°C to 100°C, for a duration ranging from 0.2 to 10 hours, with most examples reaching completion within 1 hour. Monitoring the reaction progress via TLC or HPLC is recommended to prevent over-oxidation, although the method is noted for its high conversion rates. Upon completion, the workup is straightforward, involving concentration and purification via standard silica gel chromatography or recrystallization, yielding the target benzofuran derivatives with high purity suitable for downstream applications. The detailed standardized synthesis steps for specific derivatives are outlined below to guide technical teams in replicating these results.
- Combine industrial phenol derivatives and beta-diketones in a suitable solvent such as 1,2-dichloroethane.
- Introduce a catalytic amount of iron salt (e.g., FeCl3·6H2O) and a peroxide oxidant to the reaction mixture.
- Heat the mixture under reflux conditions for approximately 1 hour, followed by standard silica gel purification to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this iron-catalyzed methodology offers substantial advantages that directly impact the bottom line and supply chain resilience. The primary driver for cost optimization is the substitution of scarce and volatile precious metal catalysts with abundant, low-cost iron salts, which effectively decouples production expenses from the fluctuating market prices of palladium or rhodium. Additionally, the ability to source starting materials directly from bulk industrial chemical suppliers, rather than relying on custom-synthesized fine chemical intermediates, significantly lowers the raw material bill of materials (BOM). This simplification of the supply base reduces the number of vendors required and mitigates the risk of supply disruptions caused by geopolitical or logistical issues affecting specialized reagent manufacturers. For supply chain heads, the reduced number of synthetic steps translates to shorter manufacturing cycles, enabling faster response times to market demand fluctuations and reducing the inventory holding costs associated with work-in-progress materials.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction of synthetic steps from multiple stages to a single pot operation result in drastic operational expenditure savings. By avoiding the need for specialized pre-functionalized substrates, manufacturers can leverage economies of scale associated with commodity phenols, while the simplified purification process reduces solvent usage and waste disposal fees. This holistic reduction in process complexity ensures that the cost of goods sold (COGS) is minimized, allowing for more competitive pricing strategies in the global marketplace without compromising margin integrity.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial raw materials such as phenol, naphthol, and simple beta-keto esters ensures a stable and continuous supply of inputs, as these chemicals are produced in massive volumes by the petrochemical industry. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, means that production can be executed in standard glass-lined or stainless steel reactors available in most multipurpose chemical plants. This flexibility allows for easier technology transfer between sites and reduces the dependency on specialized contract manufacturing organizations, thereby strengthening the overall security of supply for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of iron salts and organic by-products that are easier to treat than heavy metal residues, aligning with increasingly stringent environmental regulations. The high atom economy and the use of benign iron catalysts facilitate easier regulatory approval for new drug filings, as residual metal limits are less challenging to meet compared to processes using copper or palladium. Furthermore, the successful application of this method to complex substrates like naphthols, as seen in the synthesis of fused ring systems, demonstrates its scalability for producing high-value advanced intermediates without losing efficiency.
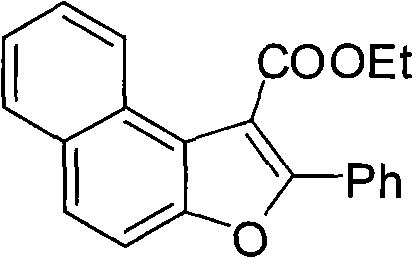 .
.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuran synthesis technology, derived from the specific capabilities and limitations observed in the patent data. These insights are intended to clarify the operational feasibility and strategic benefits for potential partners evaluating this route for their pipeline projects. Understanding these nuances is critical for making informed decisions about process adoption and long-term supply agreements.
Q: What are the primary cost advantages of this iron-catalyzed method?
A: The method replaces expensive noble metal catalysts with abundant iron salts and utilizes readily available industrial phenols, drastically reducing raw material costs and eliminating the need for costly heavy metal removal steps.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the process operates under standard reflux conditions with simple workup procedures, making it highly amenable to scale-up from kilogram to multi-ton annual production capacities without complex equipment requirements.
Q: What is the substrate scope for this benzofuran synthesis?
A: The technology demonstrates broad compatibility with various substituted phenols, naphthols, and beta-diketones, allowing for the creation of diverse polysubstituted benzofuran libraries essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis route and have integrated similar advanced methodologies into our CDMO service offerings to support global pharmaceutical innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted benzofuran meets the highest quality standards required for clinical and commercial applications. Our commitment to technical excellence allows us to navigate the complexities of C-H activation chemistry, delivering consistent results that empower our clients to accelerate their drug development timelines with confidence.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this cost-effective synthesis strategy can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability. Partnering with us means securing a supply of high-purity pharmaceutical intermediates that are produced sustainably and delivered on time, every time.
