Advanced C-H Activation for Bifuran Intermediates: Scalable & Green Manufacturing Solutions
The chemical industry is currently witnessing a paradigm shift towards sustainable synthesis, driven by the urgent need for greener processes in the production of high-value intermediates. Patent CN103880790A introduces a groundbreaking synthetic method for furan coupling compounds that addresses critical inefficiencies in traditional organic synthesis. This technology leverages direct C-H bond activation to achieve oxidative coupling of substituted furans, bypassing the cumbersome preparation of halogenated precursors. By utilizing palladium catalysts in conjunction with molecular oxygen, this innovation delivers a streamlined, one-step pathway to symmetrical bifuran structures. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity. The ability to construct biaryl ether linkages directly from simple furan feedstocks not only reduces the number of synthetic steps but also minimizes the generation of hazardous waste streams associated with stoichiometric metal reagents. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a robust platform for the reliable pharma intermediate supplier seeking to optimize their portfolio of heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oligomeric furan compounds, which are pivotal in the development of functional materials and pharmaceutical agents, has relied heavily on classical cross-coupling reactions such as the Stille or Suzuki-Miyaura protocols. These traditional methods necessitate the prior preparation of furan halides or organometallic reagents, introducing multiple additional steps that drastically increase production time and cost. Furthermore, organometallic reagents are notoriously sensitive to moisture and oxygen, requiring stringent anhydrous and anaerobic conditions that complicate reactor operations and increase energy consumption. The use of toxic tin reagents in Stille couplings poses severe environmental and safety challenges, necessitating complex purification procedures to remove trace metal contaminants to meet regulatory standards for API intermediates. Additionally, the atom economy of these traditional routes is often poor, as significant portions of the starting materials end up as waste byproducts rather than incorporated into the final product. These cumulative factors create substantial bottlenecks in cost reduction in pharmaceutical manufacturing, making the search for more efficient alternatives a top priority for forward-thinking chemical enterprises.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN103880790A utilizes a direct oxidative coupling strategy mediated by palladium catalysis. This novel approach employs substituted furans as the sole starting materials, reacting them in the presence of a palladium catalyst such as palladium trifluoroacetate or palladium acetate. Crucially, the reaction utilizes molecular oxygen as the terminal oxidant, which is not only inexpensive and abundant but also generates water as the only byproduct, aligning perfectly with green chemistry principles. The reaction proceeds efficiently in polar aprotic solvents like dimethyl sulfoxide (DMSO), often with the addition of trifluoroacetic acid to enhance reactivity. This method achieves high yields and excellent regioselectivity, specifically favoring the formation of C5-C5' linked bifuran products. The operational simplicity is remarkable, as the reaction can often be conducted at room temperature or with mild heating, eliminating the need for cryogenic conditions or high-pressure equipment. 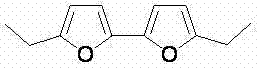
Mechanistic Insights into Palladium-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the mechanism of C-H bond activation, which allows for the direct functionalization of the furan ring without pre-activation. The catalytic cycle typically initiates with the coordination of the palladium(II) species to the electron-rich furan ring, followed by a concerted metalation-deprotonation (CMD) step that cleaves the C-H bond at the C5 position. This step is facilitated by the acidic additives and the specific electronic properties of the furan substrate, ensuring high regioselectivity even in the presence of other potential reactive sites. Once the organopalladium intermediate is formed, a second equivalent of the furan substrate undergoes a similar activation or transmetallation-like process, leading to a bis-furan palladium complex. The final step involves reductive elimination, which releases the desired bifuran product and regenerates the active palladium species, ready to enter the next catalytic cycle. The use of oxygen serves to re-oxidize any palladium(0) formed back to palladium(II), sustaining the catalytic turnover. This mechanistic elegance ensures that impurity profiles are cleaner compared to traditional methods, as there are no halogenated side products or organometallic residues to manage. 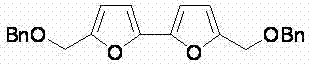
From an impurity control perspective, this method offers distinct advantages for the production of high-purity bifuran derivatives. Traditional cross-coupling often suffers from homocoupling of the organometallic reagent or incomplete conversion of the halide, leading to difficult-to-separate impurities. In this oxidative coupling system, the primary side reactions are minimized due to the high specificity of the C-H activation step. The patent data indicates that substrates with diverse functional groups, such as esters, ethers, and alkyl chains, are well-tolerated, yielding products with high structural integrity. For instance, the synthesis of bifurans bearing benzyloxy or ester groups proceeds smoothly, demonstrating the robustness of the catalytic system. This broad substrate scope is critical for R&D teams exploring structure-activity relationships, as it allows for the rapid generation of diverse libraries of furan-based scaffolds. The ability to control the reaction environment through simple parameters like temperature and oxygen flow further enhances the reproducibility of the process, ensuring consistent quality across different batches.
How to Synthesize Substituted Bifurans Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the reaction parameters outlined in the patent to maximize yield and purity. The process begins with the preparation of the reaction vessel, where the palladium catalyst is loaded and the system is purged with oxygen to ensure an oxidative atmosphere. The choice of solvent, typically DMSO, and the addition of trifluoroacetic acid are critical for solubilizing the substrates and promoting the C-H activation step. Detailed standardized synthesis steps see the guide below for precise operational protocols.
- Prepare the reaction vessel by adding palladium catalyst (Pd(OAc)2 or Pd(TFA)2) and flushing with oxygen to establish an oxidative atmosphere.
- Introduce the solvent system comprising DMSO and trifluoroacetic acid (TFA), followed by the addition of the substituted furan substrate.
- Stir the mixture at controlled temperatures (room temp to 50°C) for approximately 24 hours, then neutralize, extract, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this C-H activation technology translates into tangible strategic benefits that extend beyond mere technical novelty. The elimination of expensive and hazardous organometallic reagents significantly lowers the raw material costs associated with bifuran production. Furthermore, the simplified workflow reduces the demand for specialized equipment capable of handling air-sensitive materials, thereby lowering capital expenditure and maintenance costs. The use of oxygen as an oxidant removes the need for purchasing and disposing of stoichiometric oxidants, contributing to substantial cost savings in pharmaceutical manufacturing. From a supply chain perspective, the reliance on readily available commodity chemicals like furans, DMSO, and oxygen enhances supply security and reduces the risk of disruptions caused by the scarcity of specialized reagents. The robustness of the reaction conditions also implies a more forgiving process window, which facilitates smoother technology transfer from lab to plant and reduces the likelihood of batch failures.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the drastic simplification of the synthetic route. By removing the need for pre-functionalization steps such as halogenation or metalation, the overall process mass intensity is significantly reduced. This reduction in step count directly correlates to lower labor costs, reduced solvent consumption, and decreased energy usage per kilogram of product. Additionally, the avoidance of toxic tin or boron reagents eliminates the costly downstream processing required for metal scavenging and waste treatment. The high atom efficiency of the reaction ensures that a greater proportion of the input materials are converted into valuable product, minimizing waste disposal fees. These factors collectively drive down the cost of goods sold, allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable and abundant starting materials. Unlike organometallic reagents which have limited shelf lives and require cold chain logistics, the substituted furans and catalysts used in this process are stable and easy to store. This stability simplifies inventory management and reduces the risk of material degradation during transit. The ability to source key reagents from multiple suppliers further mitigates the risk of single-source dependency. Moreover, the scalability of the process means that production volumes can be ramped up quickly to meet surging demand without the need for extensive process re-engineering. This flexibility is crucial for maintaining continuity of supply in the fast-paced pharmaceutical and electronic materials sectors.
- Scalability and Environmental Compliance: Scaling this process to commercial levels is straightforward due to the benign nature of the reagents and conditions. The use of oxygen gas is inherently scalable, and the exothermicity of the reaction can be managed with standard cooling systems. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding waste discharge and solvent emissions. The generation of water as the primary byproduct and the potential for solvent recycling make this a highly sustainable manufacturing option. Companies adopting this technology can leverage its green credentials to meet corporate sustainability goals and satisfy the growing demand for eco-friendly supply chains. This compliance advantage not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier in the eyes of environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of this furan coupling technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their existing manufacturing frameworks.
Q: What are the primary advantages of this C-H activation method over traditional cross-coupling?
A: This method eliminates the need for pre-functionalized halides or toxic organometallic reagents, significantly reducing waste and operational complexity while utilizing molecular oxygen as a green oxidant.
Q: Does this process maintain regioselectivity for different furan substrates?
A: Yes, the patent demonstrates high regioselectivity for C5-C5' coupling across various substrates, including those with ester, alkyl, and benzyloxy substituents at the C2 or C3 positions.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Absolutely. The use of inexpensive solvents like DMSO, ambient or mild heating conditions, and oxygen gas makes this process highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bifuran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the landscape of intermediate synthesis. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. We are committed to delivering high-purity bifuran derivatives that meet the rigorous quality standards demanded by the pharmaceutical and electronic materials industries. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch we produce. By leveraging our deep technical expertise, we can help you navigate the complexities of scaling this green chemistry route, optimizing it for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to explore how this cutting-edge synthesis method can enhance your product portfolio and streamline your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us be your partner in driving innovation and efficiency in the production of high-value furan-based intermediates.
