Advanced Copper-Catalyzed Synthesis of 1,3-bis(3-aminophenoxy)benzene for High-Performance Polymer Applications
Introduction to High-Efficiency Polymer Monomer Synthesis
The development of heat-resistant polymers, such as polyimides and polyamides, relies heavily on the availability of high-purity aromatic diamine monomers. Patent CN101003487B introduces a groundbreaking preparation method for 1,3-bis(3-aminophenoxy)benzene, a critical building block that imparts exceptional thermal stability and mechanical strength to advanced polymer matrices. This technology represents a significant leap forward in fine chemical manufacturing, shifting away from hazardous solvents and inefficient catalytic systems towards a greener, more economical copper-catalyzed protocol. By utilizing 1,3-benzenediol (resorcinol) as the central scaffold and reacting it with m-chloroaniline under optimized basic conditions, the process achieves yields exceeding 85% with purity levels greater than 99%. 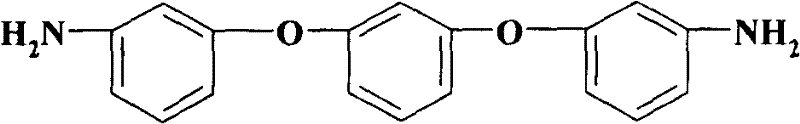
For R&D directors and procurement specialists in the polymer industry, understanding the nuances of this synthesis is vital for securing a reliable polymer monomer supplier capable of meeting stringent quality standards. The structural integrity of the resulting monomer, characterized by its specific melting point of 107-108°C and verified elemental composition, ensures consistent performance in downstream polymerization reactions. This report analyzes the technical merits of this patented route, highlighting how it resolves historical bottlenecks in supply chain continuity and production costs associated with high-performance material precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-bis(3-aminophenoxy)benzene has been plagued by inefficient methodologies that pose significant environmental and economic challenges. For instance, U.S. Patent US4222962 describes a route utilizing m-aminophenol sodium salt and m-dibromobenzene in pyridine solvent with a copper chloride catalyst. This legacy process suffers from severe drawbacks, including an excessively long reaction time of 24 hours and a disappointing yield of only 65%. Furthermore, the reliance on pyridine, a toxic and malodorous solvent, creates substantial workplace safety hazards and complicates waste disposal, making it unsuitable for modern green chemistry standards. Another approach, disclosed in U.S. Patent US4692554, involves a multi-step sequence using 1,3,5-trichlorobenzene and requires a costly palladium-on-carbon hydrogenation step followed by dehalogenation. This convoluted pathway not only inflates raw material costs due to the use of precious metal catalysts but also introduces complex purification steps like acidification and recrystallization that erode overall process efficiency.
The Novel Approach
In stark contrast, the methodology outlined in CN101003487B offers a streamlined, single-pot condensation strategy that dramatically simplifies the manufacturing landscape. By employing 1,3-benzenediol and m-chloroaniline in a mixed solvent system comprising N,N-dimethylformamide (DMF) and an azeotropic agent like toluene, the reaction kinetics are significantly accelerated. 
The novel approach utilizes inexpensive copper powder as a heterogeneous catalyst alongside organic amines such as triethylamine, eliminating the need for toxic pyridine or expensive palladium systems. The reaction proceeds efficiently at reflux temperatures for just 4 to 8 hours, achieving a remarkable yield improvement to the 85%-90% range. This shift not only reduces the operational footprint by shortening batch cycles but also enhances the economic viability of producing high-purity polymer synthesis additives. The simplicity of the workup—involving vacuum distillation of the solvent followed by direct crystallization—further underscores the industrial robustness of this technique, making it an ideal candidate for cost reduction in polymer synthesis additives manufacturing.
Mechanistic Insights into Copper-Catalyzed Etherification
The core of this technological advancement lies in the mechanistic efficiency of the copper-catalyzed nucleophilic aromatic substitution. In this system, the potassium salt (either potassium carbonate or potassium hydroxide) serves a dual purpose: it deprotonates the 1,3-benzenediol to form the reactive dipotassium phenolate species and acts as an acid scavenger for the hydrogen chloride byproduct generated during the coupling. The presence of the organic amine further stabilizes the reaction environment, ensuring that the phenolate anions remain sufficiently nucleophilic to attack the electron-deficient aromatic ring of the m-chloroaniline. The copper powder catalyst facilitates this coupling by likely forming a transient organocopper intermediate, which lowers the activation energy required for the carbon-oxygen bond formation. This mechanistic pathway is far superior to uncatalyzed thermal methods, allowing the reaction to proceed at moderate temperatures (80-90°C during addition and reflux thereafter) rather than requiring extreme thermal conditions that could degrade the sensitive amine functionalities.
From an impurity control perspective, the choice of a mixed solvent system plays a pivotal role in maintaining product integrity. The inclusion of an azeotropic solvent like toluene or xylene allows for the continuous removal of water generated during the initial salt formation and subsequent condensation. Water removal is critical because the presence of moisture can hydrolyze the aryl chloride starting material or deactivate the copper catalyst, leading to incomplete conversion and the formation of mono-substituted byproducts. By maintaining anhydrous conditions through azeotropic distillation, the process ensures that the reaction drives to completion, favoring the formation of the desired di-ether product. This precise control over reaction parameters results in a crude product with minimal impurities, which, after simple filtration and washing, yields a white crystalline solid with purity exceeding 99%, thereby meeting the rigorous specifications required for electronic grade or aerospace grade polymer applications.
How to Synthesize 1,3-bis(3-aminophenoxy)benzene Efficiently
The operational protocol for this synthesis is designed for maximum reproducibility and safety in a commercial setting. The process begins with the careful charging of 1,3-benzenediol, potassium salt, copper powder, and organic amine into a reactor equipped with a Dean-Stark trap for water removal. Following an initial stirring period at ambient temperature to ensure homogeneity, the mixture is heated to reflux to remove trace moisture, a critical step for preventing catalyst deactivation. Once the system is dry and cooled to the optimal addition temperature of 80-90°C, the m-chloroaniline is introduced dropwise to manage the exotherm and prevent local overheating.
- Prepare the reaction mixture by combining 1,3-benzenediol, potassium salt (carbonate or hydroxide), copper powder catalyst, and organic amine in a mixed solvent of DMF and toluene.
- Heat the mixture to reflux for azeotropic dehydration, then cool to 80-90°C before dropwise addition of m-chloroaniline.
- Reflux the reaction for 4-8 hours, followed by vacuum distillation, cooling, crystallization, filtration, and drying to isolate the white crystal product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere technical specifications. The transition from legacy methods to this copper-catalyzed process addresses several critical pain points in the global supply of specialty chemical intermediates. By replacing scarce or regulated reagents with commodity chemicals, manufacturers can secure a more resilient supply chain that is less susceptible to market volatility. The elimination of complex multi-step sequences found in older patents reduces the total processing time per batch, effectively increasing plant throughput without the need for capital-intensive infrastructure upgrades. Furthermore, the simplified purification workflow minimizes solvent consumption and waste generation, aligning with increasingly strict environmental regulations and reducing the total cost of ownership for the final product.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive catalysts and solvents with cost-effective alternatives. Unlike previous methods that relied on palladium on carbon for hydrogenation or pyridine as a solvent, this route utilizes cheap copper powder and standard industrial solvents like DMF and toluene. The dramatic increase in yield from roughly 65% to nearly 90% means that significantly less raw material is wasted per kilogram of finished product, directly lowering the variable cost of goods sold. Additionally, the shorter reaction cycle time allows for more batches to be produced within the same timeframe, optimizing asset utilization and labor costs. These factors combine to deliver substantial cost savings without compromising on the high purity required for downstream polymerization.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for continuous polymer production, and this method leverages widely available starting materials to mitigate supply risks. Both 1,3-benzenediol (resorcinol) and m-chloroaniline are bulk commodity chemicals produced on a massive global scale, ensuring that feedstock availability is rarely a bottleneck. In contrast, specialized halogenated intermediates or noble metal catalysts used in alternative routes can be subject to supply disruptions or geopolitical constraints. By anchoring the synthesis on stable, high-volume supply chains, manufacturers can offer more consistent lead times and guarantee long-term supply continuity for their customers. This stability is crucial for clients in the automotive and aerospace sectors who require validated sources for critical materials over multi-year contracts.
- Scalability and Environmental Compliance: The inherent simplicity of the reaction design makes it exceptionally well-suited for commercial scale-up of complex polymer additives. The absence of sensitive reagents that require cryogenic conditions or inert atmosphere gloveboxes simplifies the engineering requirements for large-scale reactors. Moreover, the workup procedure relies on standard unit operations such as distillation and crystallization, which are easily scaled from pilot plants to multi-ton production facilities. From an environmental standpoint, avoiding pyridine and minimizing heavy metal usage reduces the burden on wastewater treatment facilities and lowers the cost of hazardous waste disposal. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the sustainability profile of the final polymer products, a key selling point in modern markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-bis(3-aminophenoxy)benzene. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this monomer into their material formulations.
Q: What are the primary advantages of this copper-catalyzed method over prior art?
A: This method eliminates the use of toxic pyridine solvents and expensive palladium catalysts found in older patents, reducing reaction time from 24 hours to 4-8 hours while increasing yield from 65% to over 85%.
Q: What is the expected purity and yield of the final polymer monomer?
A: The optimized process consistently delivers 1,3-bis(3-aminophenoxy)benzene with a purity exceeding 99% and isolated yields ranging between 85% and 90%, suitable for high-performance polyimide synthesis.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the use of commodity raw materials like resorcinol and m-chloroaniline, combined with a simple workup procedure involving distillation and crystallization, makes this route highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-bis(3-aminophenoxy)benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polyimides depends on the consistent quality of your raw materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,3-bis(3-aminophenoxy)benzene meets the exacting standards required for high-performance applications. We understand the critical nature of your projects and are committed to delivering materials that enable your innovation.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can benefit your specific application. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and procurement strategies. Let us be your trusted partner in driving efficiency and quality in your polymer supply chain.
