Advanced Purification Technology for Methyl 3-Formyl-4-Hydroxyphenylacetate and Commercial Scale-Up
The global demand for potent natural antioxidants has placed hydroxytyrosol at the forefront of the nutraceutical and pharmaceutical industries, driving an urgent need for efficient synthetic routes to its key precursors. Patent CN114436905A introduces a transformative purification methodology for methyl 3-formyl-4-hydroxyphenylacetate, a critical intermediate that has historically posed significant challenges regarding purity and isolation efficiency. Traditional purification techniques often struggle to remove structurally similar impurities generated during the formylation or esterification steps, leading to bottlenecks in the downstream synthesis of high-value polyphenols. This patented approach leverages the specific chemical reactivity of the aldehyde functional group to achieve a dramatic enhancement in product quality, offering a robust solution for manufacturers seeking to optimize their supply chains for antioxidant ingredients. By shifting from physical separation methods to a chemically selective precipitation strategy, this technology addresses the core pain points of yield loss and inconsistent quality that have long plagued the production of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of aromatic aldehyde intermediates like methyl 3-formyl-4-hydroxyphenylacetate has relied heavily on column chromatography or repetitive recrystallization, both of which suffer from inherent inefficiencies at an industrial scale. Column chromatography, while effective for analytical purity, is prohibitively expensive and slow for multi-kilogram production due to the high consumption of silica gel and large volumes of organic solvents, creating significant waste disposal burdens. Alternatively, simple recrystallization often fails to adequately separate impurities that possess similar solubility profiles to the target molecule, resulting in products that may still contain substantial levels of contaminants, sometimes exceeding 10-15%. These residual impurities can interfere with subsequent reduction or coupling reactions in the synthesis of hydroxytyrosol, leading to lower overall yields and complicating the final purification of the active pharmaceutical ingredient. Furthermore, the thermal instability of some aldehyde intermediates can lead to degradation during prolonged heating required for traditional solvent removal processes.
The Novel Approach
The innovative strategy outlined in CN114436905A circumvents these limitations by utilizing a reversible chemical derivatization technique known as bisulfite adduct formation. Instead of attempting to physically separate the aldehyde from impurities based solely on solubility differences, this method chemically transforms the target aldehyde into a sodium bisulfite adduct, which exhibits drastically different solubility characteristics compared to the crude impurities. This adduct precipitates selectively out of the reaction medium, effectively leaving non-aldehydic impurities dissolved in the mother liquor. The process operates under mild conditions using inexpensive reagents like sodium bisulfite and common alcohol-water solvent systems, eliminating the need for hazardous or costly purification media. Once isolated, the adduct can be easily decomposed under acidic conditions to regenerate the parent aldehyde with exceptional purity, providing a streamlined workflow that is both economically viable and environmentally friendlier than traditional chromatographic methods.
Mechanistic Insights into Bisulfite Adduct Formation and Decomposition
The core of this purification technology lies in the nucleophilic addition of the bisulfite ion (HSO3-) to the electrophilic carbonyl carbon of the aldehyde group. In the first stage of the process, the crude methyl 3-formyl-4-hydroxyphenylacetate is dissolved in a heated aqueous alcohol solvent, typically methanol or ethanol, where it reacts with sodium bisulfite to form a stable alpha-hydroxy sulfonate salt. This reaction is highly specific to the aldehyde functionality, meaning that ketone impurities or other non-carbonyl byproducts generally do not form similar insoluble salts under these conditions. The resulting bisulfite adduct is an ionic species that possesses low solubility in the organic-rich solvent mixture, particularly upon cooling, which drives the equilibrium towards precipitation. This phase separation is the critical purification step, as the crystal lattice of the precipitating adduct tends to exclude impurities, thereby upgrading the chemical purity of the material trapped within the solid phase.
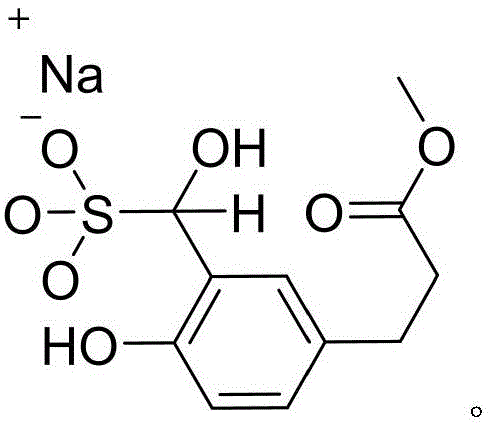
Following the isolation of the solid adduct via filtration, the second mechanistic phase involves the acid-catalyzed decomposition of the sulfonate bond. By suspending the purified adduct in a solvent and adjusting the pH to a strongly acidic range, typically between 1 and 4 using hydrochloric acid, the equilibrium is shifted back towards the free aldehyde. The protonation of the sulfonate group facilitates the elimination of sulfur dioxide and the regeneration of the carbonyl double bond. Because the impurities were largely removed in the previous filtration step, the regenerated aldehyde precipitates or remains in solution with a significantly cleaner impurity profile. This reversible chemical switch allows for a "catch-and-release" purification mechanism that is far more selective than physical methods, ensuring that the final product meets the stringent specifications required for pharmaceutical and high-end nutraceutical applications without the need for complex distillation or chromatography.
How to Synthesize Methyl 3-Formyl-4-Hydroxyphenylacetate Efficiently
To implement this purification protocol effectively, manufacturers must adhere to precise stoichiometric and thermal controls to maximize the recovery of the bisulfite adduct. The process begins with the dissolution of the crude material in a specific ratio of alcohol to water, followed by the addition of sodium bisulfite at elevated temperatures to ensure complete conversion to the adduct. Subsequent cooling induces crystallization, which must be managed carefully to prevent the occlusion of mother liquor impurities.
- React crude methyl 3-formyl-4-hydroxyphenylacetate with sodium bisulfite in an aqueous alcohol solvent system at 45-55°C to form an insoluble bisulfite adduct precipitate.
- Filter the reaction mixture while maintaining agitation and cooling to 15-25°C to isolate the solid bisulfite adduct, washing away soluble impurities.
- Decompose the purified adduct by suspending it in a solvent and adjusting the pH to 1-4 with acid, regenerating the pure aldehyde product via filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bisulfite-based purification route offers compelling economic and operational benefits that directly impact the bottom line. The primary advantage lies in the drastic simplification of the manufacturing process, which replaces expensive and time-consuming chromatographic steps with standard filtration and crystallization units that are ubiquitous in chemical plants. This transition significantly reduces the capital expenditure required for specialized equipment and lowers the operational costs associated with solvent consumption and waste treatment. Furthermore, the reagents involved, specifically sodium bisulfite and methanol, are commodity chemicals with stable pricing and abundant global availability, mitigating the risk of supply chain disruptions caused by the scarcity of exotic catalysts or proprietary resins. The robustness of the method also enhances production throughput, allowing facilities to turn over batches more rapidly and respond more agilely to market demand fluctuations for hydroxytyrosol precursors.
- Cost Reduction in Manufacturing: The elimination of silica gel chromatography and the reduction in solvent volume usage lead to substantial cost savings in raw materials and waste disposal fees. By utilizing a precipitation-based purification, the process avoids the high energy costs associated with large-scale solvent evaporation and recovery typical of traditional methods. Additionally, the high selectivity of the bisulfite reaction minimizes product loss, improving the overall mass balance and yield of the valuable intermediate, which directly translates to a lower cost per kilogram of finished goods.
- Enhanced Supply Chain Reliability: Relying on widely available inorganic salts and common organic solvents ensures that production is not held hostage by the supply constraints of niche reagents. The simplicity of the operation also reduces the dependency on highly specialized technical labor, as the process parameters are straightforward to monitor and control. This operational resilience ensures consistent delivery schedules and reduces the likelihood of batch failures due to reagent quality variability, providing a more predictable supply stream for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as the precipitation and filtration steps can be easily transferred from laboratory glassware to industrial reactors and filter presses without significant re-engineering. From an environmental perspective, the method generates less hazardous waste compared to chromatography, and the aqueous-alcoholic solvent system is easier to treat and recycle. This alignment with green chemistry principles helps manufacturers meet increasingly stringent environmental regulations and sustainability goals, enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What purity levels can be achieved with the bisulfite adduct purification method?
A: According to patent data CN114436905A, this method can elevate the purity of methyl 3-formyl-4-hydroxyphenylacetate to approximately 98.4%, significantly reducing impurity content from nearly 18% down to 0.3%.
Q: Is this purification process suitable for large-scale industrial manufacturing?
A: Yes, the process relies on standard unit operations such as dissolution, precipitation, and filtration using commodity solvents like methanol and ethanol, making it highly scalable for ton-level production without requiring exotic equipment.
Q: What are the critical control parameters for maximizing yield in this synthesis?
A: Critical parameters include maintaining a molar ratio of sodium bisulfite to aldehyde of approximately 1.3:1, controlling the solvent water content, and managing the temperature cycle between 45-55°C for dissolution and 15-25°C for crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 3-Formyl-4-Hydroxyphenylacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final therapeutic or nutraceutical product is inextricably linked to the purity of its starting materials. As a leading CDMO and supplier, we have integrated advanced purification technologies, such as the bisulfite adduct method described in CN114436905A, into our manufacturing repertoire to ensure the highest standards of quality. Our facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify that every batch of methyl 3-formyl-4-hydroxyphenylacetate meets the exacting impurity profiles necessary for sensitive downstream synthesis.
We invite you to collaborate with us to optimize your supply chain for hydroxytyrosol and related antioxidants. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient purification processes can reduce your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term business goals.
