Scalable Production of High-Purity 5-Nitro-2,4-Dihalogenated Phenols via Novel Nitration
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for critical intermediates that balance high purity with economic viability. Patent CN101381311A introduces a groundbreaking preparation method for 5-nitro-2,4-bihalogenated phenol compounds, which serve as pivotal building blocks in the synthesis of advanced active pharmaceutical ingredients. This technology addresses long-standing challenges in the nitration of halogenated phenols by utilizing a simplified protocol that relies exclusively on concentrated nitric acid, bypassing the need for costly activating agents or hazardous mixed acid systems. By leveraging abundant and low-cost raw materials, this innovation not only enhances the economic feasibility of producing these valuable intermediates but also aligns with modern green chemistry principles by reducing waste generation. For R&D directors and procurement specialists alike, understanding the nuances of this patent provides a strategic advantage in sourcing reliable pharmaceutical intermediate suppliers who can deliver consistent quality at optimized costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-nitro-2,4-bihalogenated phenols has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Traditional methodologies often necessitate the use of expensive and environmentally burdensome reagents, such as trifluoroacetic anhydride, to activate the nitration process, which drastically inflates production costs and complicates waste disposal protocols. Furthermore, existing literature describes routes employing mixed acid systems or low-temperature conditions ranging from 60°C down to -40°C, which demand specialized cooling equipment and energy-intensive operations that are difficult to sustain in an industrial setting. These conventional approaches frequently suffer from inconsistent yields and purity profiles, creating variability that is unacceptable for the stringent quality standards required in API manufacturing. The reliance on complex solvent systems and the generation of hazardous byproducts further exacerbate the operational risks, making these legacy processes less attractive for modern supply chains focused on sustainability and cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a remarkably straightforward strategy that employs concentrated nitric acid with a mass percentage greater than 90% as the sole nitrating agent. This method operates under mild temperature conditions, typically between -20°C and 30°C, which are easily achievable with standard industrial refrigeration units, thereby eliminating the need for extreme cryogenic setups. The process allows for the direct nitration of 2,4-dihalo phenol carboxylic esters or sulfonates, followed by a seamless hydrolysis step that can be performed either in a two-step sequence or a convenient one-pot system. This flexibility in operation significantly streamlines the workflow, reducing the number of unit operations and minimizing solvent consumption. By removing the dependency on exotic reagents and simplifying the reaction conditions, this technology offers a scalable and economically superior pathway for producing high-purity intermediates, positioning it as a preferred choice for commercial scale-up of complex pharmaceutical additives.
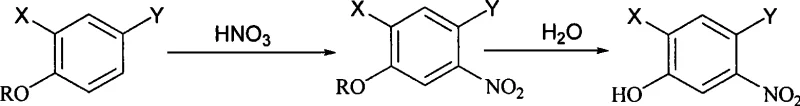
Mechanistic Insights into Concentrated Nitric Acid Nitration
The core of this innovative synthesis lies in the unique reactivity of concentrated nitric acid when interacting with protected halogenated phenol derivatives, specifically carboxylic esters or sulfonates. Unlike dilute acid systems that may require strong Lewis acid catalysts to generate the active nitronium ion, the high concentration of nitric acid in this protocol ensures a sufficient density of electrophilic species to drive the substitution reaction efficiently on the electron-deficient aromatic ring. The presence of the protecting group, such as an acetyl or tosyl moiety, plays a critical role in directing the regioselectivity of the nitration, ensuring that the nitro group is installed precisely at the 5-position relative to the oxygen substituent. This precise control over regiochemistry is vital for downstream applications, as it minimizes the formation of isomeric impurities that are notoriously difficult to separate. The subsequent hydrolysis step cleaves the protecting group under aqueous thermal conditions, regenerating the phenolic hydroxyl group without compromising the integrity of the newly installed nitro functionality or the halogen substituents.
From an impurity control perspective, the mechanism inherently favors high purity due to the specificity of the reaction conditions and the stability of the intermediates involved. The use of concentrated nitric acid minimizes side reactions such as oxidation or over-nitration, which are common pitfalls in less controlled environments. Additionally, the hydrolysis step serves as a purification opportunity, as many organic byproducts generated during the nitration phase remain soluble in the aqueous phase or are removed during the filtration of the final solid product. The patent data indicates that this mechanistic pathway consistently yields products with purity levels exceeding 98%, demonstrating the robustness of the chemical transformation. For quality assurance teams, this implies a reduced burden on downstream purification processes, allowing for more efficient resource allocation and faster time-to-market for the final drug substance.
How to Synthesize 5-Nitro-2,4-Dihalogenated Phenols Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize yield and safety. The process begins with the preparation of the reaction vessel, typically a three-necked flask equipped with a thermometer and agitator, into which concentrated nitric acid is charged and cooled to the specified range. The substrate, whether it be a propionate, butyrate, or sulfonate derivative, is then added dropwise to maintain thermal stability, followed by a stirring period to ensure complete conversion as monitored by HPLC. Following the nitration, the mixture undergoes hydrolysis either by direct water addition or after an extraction workup, depending on the specific variant of the process chosen. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- React 2,4-dihalo phenol carboxylic esters or sulfonates with concentrated nitric acid (>90%) at temperatures between -20°C and 30°C.
- Perform hydrolysis by adding water directly to the reaction mixture or after extraction, heating to 80-95°C for 3-8 hours.
- Cool the mixture to room temperature and filter to isolate the high-purity yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical transformation. By eliminating the requirement for expensive activators like trifluoroacetic anhydride, the raw material cost structure is significantly optimized, allowing for more competitive pricing models in the global marketplace. The simplification of the process flow, particularly the ability to perform hydrolysis in a one-pot manner, reduces the demand for extensive solvent inventory and minimizes the footprint of the manufacturing facility, leading to substantial overhead savings. Furthermore, the reliance on commercially available and abundant reagents ensures a stable supply chain that is less susceptible to the volatility often associated with specialty chemical markets. This resilience is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by international pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of high-cost reagents such as trifluoroacetic anhydride and the avoidance of complex mixed acid systems directly lowers the bill of materials for every batch produced. Additionally, the simplified workup procedure reduces solvent consumption and waste treatment costs, contributing to a leaner and more efficient manufacturing operation. The high yields reported in the patent embodiments further enhance cost efficiency by maximizing the output from each unit of raw material input. These factors combined create a compelling economic case for switching to this technology, offering significant margin improvements for manufacturers of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available concentrated nitric acid and common ester precursors mitigates the risk of supply disruptions that can occur with niche or imported specialty chemicals. The robustness of the reaction conditions means that production can be maintained across different geographic locations without the need for highly specialized infrastructure. This geographical flexibility allows for a more diversified supply base, reducing the risk of single-source dependency. Consequently, partners can rely on a steady flow of materials, ensuring that downstream API synthesis is never halted due to intermediate shortages, thereby securing the overall value chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale, operating at temperatures and pressures that are standard in industrial chemical plants, which facilitates a smooth transition from pilot scale to multi-ton production. The reduction in hazardous waste generation, particularly the avoidance of fluorinated byproducts, simplifies environmental compliance and lowers the cost of waste disposal. This alignment with green chemistry principles not only meets regulatory requirements but also enhances the corporate sustainability profile of the manufacturer. Such environmental stewardship is increasingly becoming a key criterion for selection by major pharmaceutical companies looking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific capabilities and limitations outlined in the intellectual property, ensuring accurate expectations for performance and output.
Q: What are the primary advantages of this nitration method over conventional routes?
A: This method eliminates the need for expensive reagents like trifluoroacetic anhydride or complex mixed acid systems, significantly reducing raw material costs and environmental impact while maintaining high yields.
Q: What purity levels can be achieved with this synthesis route?
A: According to patent embodiments, the process consistently achieves purity levels exceeding 98%, with specific examples reaching up to 99.6% as verified by HPLC analysis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes commercially available reagents and standard equipment without requiring special devices, making it highly adaptable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro-2,4-Dihalogenated Phenols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our expertise in handling complex halogenated compounds allows us to navigate the intricacies of this patented process effectively, guaranteeing a supply of material that supports your R&D and commercial goals without compromise.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this technology can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for your fine chemical and pharmaceutical intermediate needs.
