Advanced Room-Temperature Synthesis of S-Substituted Cysteine Derivatives for Commercial Scale-Up
Advanced Room-Temperature Synthesis of S-Substituted Cysteine Derivatives for Commercial Scale-Up
The landscape of fine chemical engineering is constantly evolving, driven by the urgent need for more sustainable and cost-effective synthetic methodologies, particularly in the realm of pharmaceutical intermediates. A groundbreaking development detailed in patent CN113735750B introduces a novel protocol for the preparation of S-substituted cysteine derivatives utilizing N-bromosuccinimide (NBS) as a key promoter at ambient temperatures. This innovation represents a significant paradigm shift away from traditional transition-metal catalyzed processes, offering a streamlined, one-pot synthetic route that operates under mild conditions. For R&D directors and process chemists, this method unlocks new possibilities for constructing asymmetric disulfide bonds, which are critical structural motifs found in a vast array of bioactive compounds and self-healing materials. By leveraging the unique reactivity of NBS, this technology not only simplifies the operational complexity but also aligns perfectly with modern green chemistry principles, positioning it as a highly attractive candidate for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
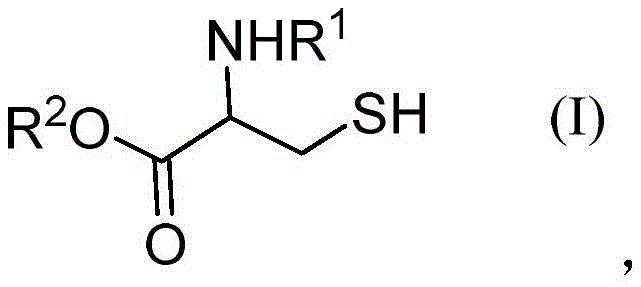
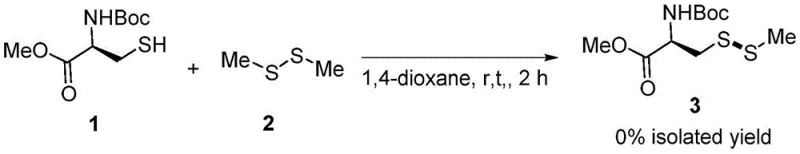
Historically, the direct exchange reaction between thiols and symmetric disulfides to generate asymmetric disulfides has been a challenging transformation requiring harsh conditions or expensive catalysts. Prior art, such as the method reported by researchers at Shanghai University, relied heavily on palladium chloride (PdCl2) as a catalyst and dimethyl sulfoxide (DMSO) as a solvent, necessitating elevated temperatures around 80°C to drive the reaction to completion. While effective, this legacy approach presents substantial drawbacks for large-scale manufacturing, primarily due to the prohibitive cost of palladium and the stringent regulatory requirements associated with removing trace heavy metals from final pharmaceutical products. Furthermore, the high thermal energy input increases the risk of racemization for chiral amino acid derivatives and complicates the safety profile of the process, making it less desirable for the production of sensitive biological intermediates. As demonstrated in comparative studies within the patent data, attempting this transformation without a suitable promoter or using alternative halogenating agents often results in complete reaction failure, yielding 0% of the desired product, which underscores the critical need for a more specialized catalytic system.
The Novel Approach
In stark contrast to these conventional limitations, the novel methodology disclosed in the patent utilizes N-bromosuccinimide (NBS) to facilitate the efficient construction of S-substituted cysteine derivatives at room temperature. This approach eliminates the dependency on precious metal catalysts entirely, replacing them with a readily available and cost-effective organic reagent that activates the symmetric disulfide for nucleophilic attack by the thiol group. The reaction proceeds smoothly in 1,4-dioxane, achieving impressive isolated yields of up to 85% within just two hours, as exemplified by the synthesis of N-(tert-butyloxycarbonyl)-S-methylthio-L-cysteine methyl ester. This mild, metal-free strategy not only drastically reduces the environmental footprint of the synthesis but also simplifies the downstream purification process, as there is no need for specialized scavengers to remove palladium residues. The versatility of this system is further highlighted by its compatibility with various symmetric disulfides, including diphenyl disulfide, allowing for the modular synthesis of diverse aryl and alkyl thio-derivatives with high fidelity.
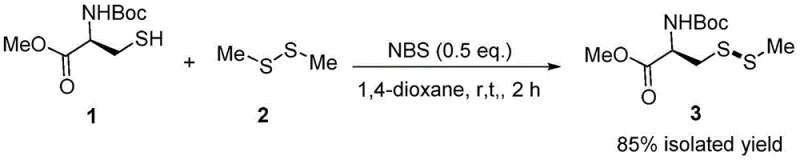
Mechanistic Insights into NBS-Mediated Disulfide Exchange
The core of this technological advancement lies in the unique ability of NBS to mediate the thiol-disulfide exchange reaction under neutral, mild conditions. Unlike traditional radical initiators or strong oxidants, NBS acts as a subtle activator that likely generates a reactive sulfenyl bromide intermediate in situ, which is then rapidly displaced by the thiolate anion of the cysteine derivative to form the new asymmetric disulfide bond. This mechanism avoids the formation of highly reactive radical species that could lead to polymerization or degradation of the sensitive amino acid backbone, thereby ensuring high chemoselectivity. The general reaction scheme illustrates how the symmetric disulfide (RSSR) interacts with the cysteine thiol (R'-SH) in the presence of catalytic amounts of NBS to yield the target asymmetric disulfide (R-S-S-R') with high efficiency. This mechanistic pathway is distinct from simple oxidative coupling, as it specifically leverages the electrophilic nature of the bromine atom in NBS to activate the sulfur-sulfur bond without over-oxidizing the substrate to sulfones or sulfoxides.
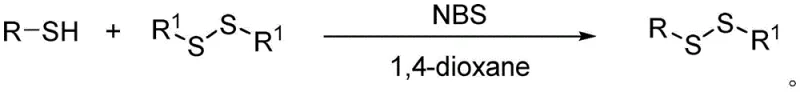
From an impurity control perspective, the room-temperature operation is paramount for maintaining the stereochemical integrity of the cysteine scaffold. High-temperature processes often induce epimerization at the alpha-carbon of amino acids, leading to difficult-to-separate diastereomeric impurities that can compromise the safety and efficacy of the final drug substance. By conducting the reaction at 20-30°C, this novel method effectively suppresses thermal racemization, resulting in a cleaner crude reaction profile that requires less aggressive purification. Additionally, the absence of transition metals removes a major class of genotoxic impurities from the process stream, significantly de-risking the regulatory filing for any API manufactured using this intermediate. The specificity of NBS is further confirmed by control experiments where N-chlorosuccinimide (NCS) failed to promote the reaction, indicating that the bromine atom plays a specific and irreplaceable role in the activation energy landscape of this transformation.
How to Synthesize S-Substituted Cysteine Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and requires standard equipment, making it highly accessible for process development teams. The procedure involves dissolving the cysteine derivative and the symmetric disulfide in 1,4-dioxane, followed by the addition of 0.5 equivalents of NBS, after which the mixture is stirred at ambient temperature. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for maximizing yield and reproducibility. For a comprehensive, step-by-step guide on executing this reaction with precise stoichiometry and purification techniques, please refer to the standardized protocol below.
- Combine the cysteine derivative (such as N-Boc-L-cysteine methyl ester), a symmetric disulfide (like dimethyl disulfide), and N-bromosuccinimide (NBS) in an organic solvent such as 1,4-dioxane.
- Stir the reaction mixture vigorously at room temperature (20-30°C) for approximately 2 hours to ensure complete conversion to the S-substituted product.
- Concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the high-purity target disulfide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this NBS-mediated synthesis offers profound strategic advantages that extend far beyond simple chemical yield improvements. By transitioning away from palladium-catalyzed routes, organizations can achieve significant cost reduction in API manufacturing through the elimination of expensive noble metals and the associated analytical costs for residual metal testing. The simplified workflow reduces the number of unit operations required, leading to shorter batch cycle times and increased throughput capacity without the need for capital-intensive high-temperature reactors. Furthermore, the reliance on commodity chemicals like NBS and 1,4-dioxane ensures a robust and resilient supply chain, mitigating the risks associated with the volatility of precious metal markets. This process intensification allows for a more agile response to market demands, ensuring consistent availability of high-quality intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of palladium catalysts, which are subject to extreme price fluctuations and require costly recovery or disposal protocols. By substituting this with NBS, a stable and inexpensive reagent, the direct material cost of the synthesis is drastically lowered. Additionally, the mild reaction conditions eliminate the energy costs associated with heating reactions to 80°C or higher, contributing to lower utility expenses per kilogram of product. The simplified purification process, devoid of heavy metal scavenging steps, further reduces the consumption of auxiliary materials and labor hours, compounding the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Dependence on single-source or geographically concentrated suppliers for precious metals poses a significant risk to production continuity. This new method diversifies the raw material base to include widely available organic reagents, thereby insulating the supply chain from geopolitical disruptions or mining shortages. The stability of the reagents allows for longer shelf-life and easier storage logistics, reducing waste from expired catalysts. Moreover, the robustness of the reaction across different batches ensures predictable output schedules, enabling supply chain planners to optimize inventory levels and reduce the need for safety stock buffers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this room-temperature protocol inherently minimizes thermal hazards and solvent emissions. The absence of heavy metals simplifies wastewater treatment and waste disposal, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The process is easily adaptable from gram-scale discovery to multi-ton commercial production using standard stainless steel reactors, facilitating rapid technology transfer. This scalability ensures that as demand for the final therapeutic grows, the supply of the intermediate can be ramped up seamlessly without requiring complex process re-engineering or new regulatory filings for hazardous conditions.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope and operational parameters. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, ensuring accuracy and relevance for process planning. Understanding these nuances is essential for integrating this method into existing production workflows effectively.
Q: Why is NBS preferred over palladium catalysts for this synthesis?
A: NBS allows the reaction to proceed efficiently at room temperature without the need for expensive transition metals like palladium, significantly reducing raw material costs and eliminating the complex downstream processing required to remove heavy metal residues.
Q: What is the optimal solvent system for this disulfide exchange reaction?
A: While various solvents like acetonitrile and THF were tested, 1,4-dioxane was identified as the superior medium, providing the highest isolated yields (up to 85%) for the formation of asymmetric disulfide bonds.
Q: Can this method be applied to unprotected amino acids?
A: Yes, the methodology is robust and versatile, successfully accommodating both protected derivatives (such as N-Boc esters) and free amino acid forms, making it suitable for diverse synthetic pathways in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Substituted Cysteine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this NBS-mediated synthesis for the next generation of pharmaceutical intermediates and fine chemicals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and compliant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of S-substituted cysteine derivative meets the highest industry standards for chirality and chemical purity. We are committed to delivering not just a product, but a comprehensive solution that optimizes your supply chain and accelerates your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free protocol for your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our reference standards and to schedule a consultation for detailed route feasibility assessments, ensuring your project moves forward with confidence and precision.
