Advanced Asymmetric Hydrogenation for High-Purity Fidarestat Intermediate Production
Advanced Asymmetric Hydrogenation for High-Purity Fidarestat Intermediate Production
The pharmaceutical industry's relentless pursuit of effective treatments for diabetic complications has placed Aldose Reductase Inhibitors (ARIs) at the forefront of metabolic disease research. Among these, Fidarestat stands out as a potent candidate due to its strong enzyme inhibitory activity and favorable safety profile. However, the commercial viability of such advanced therapeutics often hinges on the efficiency of synthesizing their chiral building blocks. A groundbreaking patent, CN113979983A, filed in early 2022, introduces a transformative asymmetric synthesis method for the key intermediate of Fidarestat, specifically (S)-6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid. This technology leverages a sophisticated rhodium-catalyzed asymmetric hydrogenation system that promises to redefine the manufacturing landscape for this critical pharmaceutical intermediate. By utilizing a novel chiral ferrocene diphosphine ligand metal complex, the process achieves exceptional enantioselectivity and conversion rates under mild conditions, addressing long-standing challenges in yield and purity that have plagued previous synthetic routes.
For R&D directors and process chemists, the implications of this patent are profound. It shifts the paradigm from stoichiometric resolution or expensive hydrosilation methods to a catalytic, atom-economical hydrogenation process. The ability to generate the chiral center directly from the achiral precursor, 6-fluoro-4-oxo-4H-chromene-2-carboxylic acid, without the need for cumbersome separation steps, represents a significant leap forward in process chemistry. This report delves deep into the technical nuances of this innovation, analyzing how it optimizes the supply chain for reliable pharmaceutical intermediates supplier networks and reduces the overall cost burden in API manufacturing. As we explore the mechanistic details and commercial advantages, it becomes clear that this technology is not just a laboratory curiosity but a robust platform ready for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the chiral intermediate required for Fidarestat has been fraught with inefficiencies that hinder large-scale production. The traditional approach primarily relied on the preparation of the racemic modification of 6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid followed by optical resolution. This classical resolution strategy is inherently flawed because it imposes a theoretical maximum yield of merely 50%, meaning half of the valuable starting material is discarded or requires energy-intensive recycling processes. Furthermore, resolution often involves the use of chiral resolving agents which add to the material cost and complicate the downstream purification, leading to a serious three-waste problem that conflicts with modern green chemistry principles. Another existing method involves copper-complex catalyzed hydrosilation, which, while capable of achieving high enantiomeric excess (up to 99% ee), suffers from poor atom economy. This method necessitates the use of two equivalents of expensive silane reagents and high catalyst loadings (up to 20 mmol%), making it economically unviable for ton-scale production. These limitations create bottlenecks in the supply chain, increasing lead times and driving up the cost of goods sold (COGS) for the final API.
The Novel Approach
In stark contrast, the method disclosed in patent CN113979983A offers a streamlined, direct asymmetric catalytic hydrogenation route that elegantly bypasses these historical hurdles. By employing a transition metal-catalyzed hydrogenation using molecular hydrogen, the process achieves high atom economy, as the only byproduct is essentially the saturated bond formation without the generation of stoichiometric waste associated with silanes or resolving agents. The core of this innovation lies in the use of a specific chiral bisphosphine ligand metal complex, formed by the in-situ complexation of a chiral diphosphine ligand and a metal precursor. This catalytic system operates efficiently under mild conditions, eliminating the need for harsh reagents or extreme temperatures. The reaction converts the readily available 6-fluoro-4-oxo-4H-chromene-2-carboxylic acid directly into the desired chiral intermediate with high fidelity.
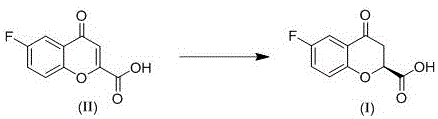
As illustrated in the reaction scheme above, the transformation is direct and clean. The novel approach allows for a catalyst-to-substrate molar ratio ranging from 1:100 to 1:10000, which is a drastic reduction in precious metal usage compared to the copper-catalyzed alternatives. This efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, as the expensive metal catalyst constitutes a smaller fraction of the total production cost. Moreover, the operational simplicity—using common solvents like methanol or ethanol and standard hydrogenation equipment—makes this method highly attractive for commercial scale-up of complex pharmaceutical intermediates, ensuring a stable and continuous supply for downstream drug formulation.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The success of this asymmetric synthesis hinges on the precise design of the chiral catalyst system. The patent specifies the use of a chiral diphosphine ligand featuring a ferrocene skeleton, which serves as the chiral inducer in the catalytic cycle. Ferrocene-based ligands are renowned in organometallic chemistry for their rigidity and the ability to create a well-defined chiral pocket around the metal center. In this specific application, the ligand structure (III) contains variable groups R1 and R2, where R1 can be tert-butyl or phenyl derivatives, and R2 can be naphthyl or phenyl groups. These bulky substituents are critical for steric differentiation, forcing the substrate to approach the metal-hydride species from a specific face, thereby controlling the stereochemistry of the hydrogen addition. The metal precursor is typically derived from Group 9 metals such as Ruthenium (Ru), Rhodium (Rh), Iridium (Ir), Palladium (Pd), or Platinum (Pt), with Rhodium complexes like [Rh(NBD)2]BF4 showing exemplary performance in the provided examples.
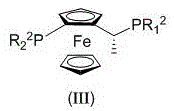
The catalytic cycle likely proceeds through the coordination of the olefinic double bond of the chromone substrate to the rhodium center, followed by oxidative addition of hydrogen and migratory insertion. The chiral environment provided by the ferrocene ligand ensures that the hydride transfer occurs selectively to generate the (S)-enantiomer. The patent data highlights that this mechanism is robust across a range of conditions, with hydrogen pressures from 1 to 100 atm and temperatures from -20°C to 100°C. Crucially, the system demonstrates remarkable tolerance to functional groups, preserving the carboxylic acid and the fluorine substituent on the aromatic ring without side reactions. This high level of chemoselectivity is vital for maintaining the integrity of the molecule, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. The ability to achieve >99% conversion and 99% ee in Example 1 underscores the potency of this ligand-metal combination in controlling the reaction pathway.
How to Synthesize (S)-6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid Efficiently
Implementing this synthesis in a pilot or production plant requires careful attention to the preparation of the active catalyst species and the control of reaction parameters. The process begins with the in-situ generation of the catalyst, where the metal precursor and the chiral ligand are mixed in a solvent such as methanol at room temperature. This pre-activation step ensures that the active catalytic species is fully formed before the introduction of the substrate, maximizing the initial reaction rate. The substrate, 6-fluoro-4-oxo-4H-chromene-2-carboxylic acid, is then introduced, and the mixture is subjected to a hydrogen atmosphere. The flexibility of the process allows for the use of various polar protic or aprotic solvents, giving process engineers the freedom to optimize for solubility and downstream processing. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the catalyst solution by complexing a metal precursor (e.g., [Rh(NBD)2]BF4) with a chiral diphosphine ligand in a solvent like methanol at room temperature.
- Mix the catalyst solution with the substrate 6-fluoro-4-oxo-4H-chromene-2-carboxylic acid in a reaction solvent, ensuring a molar ratio of catalyst to substrate between 1: 100 and 1:10000.
- Conduct the asymmetric hydrogenation reaction under hydrogen pressure (1-100 atm) at temperatures ranging from -20°C to 100°C for 1 to 48 hours.
- Upon completion, release hydrogen, remove the solvent, dissolve the residue in an organic solvent, wash with water and brine, dry, and evaporate to obtain the pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology offers tangible strategic benefits that extend beyond mere technical elegance. The shift from resolution or hydrosilation to direct hydrogenation fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the need for stoichiometric amounts of expensive silane reagents and reducing the catalyst loading by orders of magnitude, the raw material costs are significantly curtailed. Furthermore, the simplification of the workflow—removing the resolution and recycling steps—reduces the number of unit operations, which in turn lowers labor costs, energy consumption, and equipment occupancy time. This streamlining leads to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing of the final API in a crowded market.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of material efficiency. Traditional resolution methods inherently waste at least half of the starting material, effectively doubling the raw material cost for the chiral intermediate. In contrast, this catalytic hydrogenation approach approaches 100% theoretical yield, meaning every kilogram of starting material purchased contributes to the final product. Additionally, the elimination of silane reagents, which are not only costly but also require special handling and storage due to their reactivity, removes a significant line item from the bill of materials. The low catalyst loading further minimizes the expenditure on precious metals, and the potential for catalyst recovery and recycling adds another layer of financial optimization. These factors combine to drastically lower the variable cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that rely on niche reagents or multi-step sequences prone to failure. This new method enhances reliability by relying on commodity chemicals like molecular hydrogen and common alcohols, which are readily available globally. The robustness of the reaction conditions, which tolerate a wide range of temperatures and pressures, reduces the risk of batch failures due to minor process deviations. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the likelihood of batch rejection during quality control. This predictability allows supply chain planners to forecast production timelines with greater accuracy, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream API synthesis is never starved of critical inputs.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a process becomes a key decision factor. This hydrogenation method aligns perfectly with green chemistry principles by maximizing atom economy and minimizing waste generation. The absence of heavy metal waste streams associated with stoichiometric reagents and the use of greener solvents like ethanol simplify wastewater treatment and reduce disposal costs. From a scalability perspective, hydrogenation is a well-understood unit operation in the fine chemical industry, with established engineering protocols for scaling from liters to cubic meters. The ability to run the reaction at moderate pressures and temperatures means that standard stainless steel reactors can be used without the need for exotic metallurgy, facilitating rapid commercial scale-up to meet market demand without prohibitive capital expenditure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the process for potential partners and licensees. Understanding these details is crucial for making informed decisions about integrating this route into your existing manufacturing portfolio.
Q: What are the advantages of this asymmetric hydrogenation method over traditional resolution?
A: Unlike traditional resolution methods which suffer from a maximum theoretical yield of 50% and generate significant waste, this asymmetric catalytic hydrogenation offers high atom economy, potentially achieving near 100% yield with excellent enantiomeric excess (up to 99% ee), drastically reducing raw material costs and environmental impact.
Q: Why is the ferrocene-based ligand critical for this synthesis?
A: The specific chiral ferrocene diphosphine ligand described in the patent provides a rigid and well-defined chiral environment around the rhodium metal center. This structural feature is essential for differentiating the prochiral faces of the chromone substrate during hydrogenation, ensuring the high stereoselectivity required for the biologically active (S)-enantiomer.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes mild reaction conditions (room temperature to 100°C, moderate hydrogen pressure), avoids expensive and hazardous silane reagents used in copper-catalyzed alternatives, and employs common solvents like methanol or ethanol, making it highly practical for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fidarestat Intermediate Supplier
The technological breakthroughs detailed in patent CN113979983A represent a significant opportunity for the pharmaceutical industry to optimize the production of diabetes therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methods into robust, industrial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promise of high efficiency and low cost is realized in the final product. Our state-of-the-art facilities are equipped with advanced hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Fidarestat intermediate we produce adheres to the highest global standards.
We invite forward-thinking pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced synthesis route. By partnering with our technical team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical intermediate, driving your diabetic complication therapies forward with confidence and reliability.
