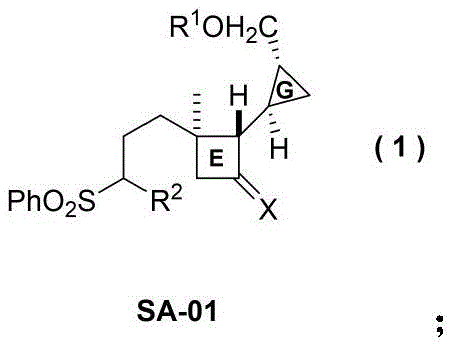
Scalable Synthesis of Solanoeclepin A Intermediate SA-01 for Advanced Agrochemical Development
The agricultural and pharmaceutical sectors are constantly seeking potent bioactive molecules to combat resistant pests and diseases, and the natural product Solanoeclepin A stands out as a prime candidate due to its unique structural complexity and biological efficacy. Patent CN111187186B discloses a groundbreaking preparation method for Intermediate SA-01, which represents the critical GE ring system of Solanoeclepin A. This intermediate is not merely a chemical curiosity; it is the functional core responsible for the molecule's extremely strong induction activity on the hatching process of potato cyst nematodes (PCN), specifically Globodera rostochiensis and G. pallida. The patent details a novel synthetic strategy that overcomes the historical limitations of sourcing this compound from natural extracts, which are notoriously scarce and economically unviable for mass application. By establishing a robust, linear synthesis starting from commercially abundant geraniol acetate, this technology paves the way for the reliable supply of high-purity agrochemical intermediates necessary for next-generation crop protection solutions.
The significance of this invention extends beyond simple molecule construction; it addresses the urgent global need for sustainable pest management tools that operate at ultra-low dosages, cited in the patent as effective at merely 0.3 g/hectare. For R&D directors and procurement specialists alike, the ability to access such a complex chiral scaffold through a predictable, non-natural synthesis route represents a paradigm shift in supply chain security. The methodology described eliminates the bottlenecks associated with botanical extraction, offering a consistent, quality-controlled alternative that aligns with the rigorous standards required for regulatory approval in both agrochemical and pharmaceutical domains. This report analyzes the technical depth and commercial viability of this synthesis, highlighting its potential to become a cornerstone in the manufacturing of advanced nematicides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Solanoeclepin A and its key fragments has been plagued by severe supply chain vulnerabilities inherent to natural product isolation. The compound was originally isolated from potato roots of the Solanaceae family, a source that yields minute quantities relative to the agricultural land area required for cultivation. Relying on extraction means facing fluctuating yields dependent on seasonal variations, soil conditions, and plant health, rendering the cost of goods sold (COGS) prohibitively high for widespread commercial deployment. Furthermore, the structural complexity of the DEFG ring system, featuring fused chiral cyclopropane and cyclobutane moieties, makes total synthesis via traditional routes exceptionally challenging. Conventional synthetic attempts often suffer from poor stereocontrol, requiring extensive chromatographic purification that drastically reduces overall throughput and generates significant chemical waste. These factors combined create a barrier to entry for developing formulations based on this active scaffold, limiting its potential impact on potato cyst nematode control despite its proven biological potency.
The Novel Approach
The methodology presented in patent CN111187186B offers a transformative solution by utilizing geraniol acetate, a cheap and abundant terpene derivative, as the foundational building block. This strategic choice immediately lowers the raw material cost baseline and ensures a stable supply chain independent of agricultural harvests. The synthetic route is designed with modularity and efficiency in mind, employing classic organic transformations such as nucleophilic substitution, Horner-Wadsworth-Emmons (HWE) olefination, and Wittig reactions that are well-understood and easily optimized in a pilot plant setting. Crucially, the route integrates a highly selective asymmetric cyclopropanation step induced by a Charette chiral ligand, which installs the critical stereocenters early in the sequence with high fidelity. This approach not only simplifies the purification burden but also ensures that the final intermediate SA-01 possesses the precise three-dimensional architecture required for biological activity. The entire process is streamlined to minimize unit operations, avoiding the use of exotic or toxic reagents wherever possible, thereby enhancing the environmental profile and operational safety of the manufacturing process.
Mechanistic Insights into Charette Ligand-Induced Asymmetric Cyclopropanation
A pivotal moment in this synthesis is the construction of the chiral cyclopropane ring, a structural motif essential for the bioactivity of the Solanoeclepin A core. The patent describes a chemoselective asymmetric cyclopropanation reaction performed on the diallyl alcohol compound SA-08. This transformation is mediated by a zinc carbenoid species generated in situ from diiodomethane and diethylzinc, but the true driver of stereochemical outcome is the Charette chiral ligand (Ligand A). Mechanistically, the ligand coordinates to the zinc center, creating a chiral environment that directs the approach of the carbenoid to the alkene substrate. This coordination lowers the activation energy for the formation of one specific enantiomer while sterically hindering the other, resulting in high enantiomeric excess without the need for resolution steps. The success of this step is critical because any erosion of chirality here would propagate through the subsequent Staudinger ketene cycloaddition, potentially rendering the final product biologically inactive. The careful control of temperature (-20 °C) and solvent composition (DME/DCM) further fine-tunes the reactivity, preventing non-selective background reactions and ensuring that the cyclopropanation occurs exclusively at the desired allylic position.
Following the cyclopropanation, the synthesis proceeds through a substrate-chirality-induced asymmetric Staudinger cycloaddition to construct the cyclobutanone ring. This cascade of stereocontrolled events demonstrates a sophisticated understanding of conformational analysis and transition state theory. The existing chirality from the cyclopropane ring influences the facial selectivity of the ketene addition, effectively transferring chiral information across the molecular framework. This "chiral relay" mechanism allows for the rapid assembly of the complex GE ring system with multiple contiguous stereocenters from relatively simple precursors. From an impurity control perspective, this high degree of intrinsic selectivity means that the crude reaction profiles are much cleaner than those obtained from non-catalyzed or poorly catalyzed routes. Fewer diastereomers are formed, which simplifies downstream processing and reduces the load on purification columns, directly translating to higher recovery rates of the target intermediate and lower solvent consumption per kilogram of product.
How to Synthesize Solanoeclepin A Intermediate SA-01 Efficiently
The synthesis of Intermediate SA-01 is a testament to modern organic process chemistry, balancing complexity with operational simplicity. The route begins with the conversion of a tosylate precursor (SA-03) into a phenyl sulfone (SA-04) via a straightforward SN2 displacement, followed by deprotection and chain elongation to set up the diene system required for cyclization. The subsequent steps involve precise stoichiometric control, particularly during the reduction of the ester to the alcohol using DIBAL-H, where temperature management is crucial to prevent over-reduction. The final stages involve the installation of the side chain functionality through alkylation of the sulfone-stabilized carbanion, a reaction that requires strict anhydrous conditions and low temperatures to maintain regioselectivity. While the laboratory procedures are detailed in the patent, scaling this sequence requires attention to heat transfer during exothermic steps like the zinc-copper couple preparation and the handling of pyrophoric reagents like n-butyllithium. Detailed standardized operating procedures (SOPs) are essential to replicate the high yields reported in the examples consistently.
- Convert geraniol acetate derivative SA-03 to phenyl sulfone SA-04 via nucleophilic substitution with sodium phenylsulfinate.
- Perform deprotection, oxidation, HWE olefination, and reduction to generate diallyl alcohol SA-08.
- Execute chemoselective asymmetric cyclopropanation using Charette ligand A, followed by Staudinger cycloaddition and final alkylation to yield SA-01.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic route are profound, primarily driven by the decoupling of production from natural sources. The reliance on geraniol acetate, a commodity chemical produced in massive volumes for the fragrance industry, ensures that raw material costs remain low and volatility is minimized. Unlike natural extraction, which is subject to the vagaries of climate and crop disease, this chemical synthesis offers a predictable, year-round production schedule that can be ramped up to meet surging demand without lead time penalties. The elimination of expensive noble metal catalysts (such as palladium or rhodium) in favor of base metals like zinc and copper further drives down the direct material costs. Additionally, the avoidance of transition metals simplifies the purification process, as there is no need for costly metal scavenging resins or extensive testing for residual heavy metals, which is a stringent requirement for agrochemical registration. This streamlined workflow results in a significantly reduced cost of goods sold (COGS), making the final nematicide product more competitive in the marketplace.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive, bulk-available starting materials and avoiding proprietary, high-cost catalysts. The synthetic steps are designed to be telescoped where possible, reducing the number of isolation and purification stages which are typically the most expensive parts of fine chemical manufacturing. By minimizing solvent exchanges and utilizing common reagents like sodium phenylsulfinate and manganese dioxide, the operational expenditure (OPEX) is kept to a minimum. Furthermore, the high atom economy of the key coupling reactions ensures that raw materials are efficiently converted into product rather than waste, maximizing the value derived from every kilogram of input.
- Enhanced Supply Chain Reliability: Adopting this synthetic route mitigates the risks associated with single-source natural extracts, providing a robust alternative that guarantees continuity of supply. The reagents used are standard industrial chemicals with multiple global suppliers, preventing bottlenecks caused by the monopoly of a single vendor. The robustness of the chemistry allows for flexible manufacturing; if one batch fails or a specific reagent is delayed, the process parameters are forgiving enough to allow for recovery or substitution without compromising the final quality. This resilience is critical for maintaining the production schedules of downstream formulation plants, ensuring that farmers receive their crop protection products exactly when needed during the planting season.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, utilizing reaction types that are routinely performed in multi-ton reactors, such as refluxing substitutions and cryogenic additions. The absence of highly toxic or persistent organic pollutants in the reagent list simplifies waste treatment and disposal, aligning with increasingly strict environmental regulations. The use of zinc/copper couples and ammonium chloride workups generates waste streams that are easier to treat compared to those containing heavy transition metals or halogenated solvents in large quantities. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, a key factor for modern procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Intermediate SA-01. These answers are derived directly from the experimental data and technical specifications outlined in patent CN111187186B, providing clarity on the feasibility and advantages of this specific synthetic pathway. Understanding these details is crucial for stakeholders evaluating the integration of this intermediate into their broader R&D pipelines or supply networks.
Q: What is the primary biological activity associated with Intermediate SA-01?
A: Intermediate SA-01 constitutes the GE ring system of Solanoeclepin A, a natural product exhibiting extremely strong induction activity (0.3 g/hectare) on the hatching process of potato cyst nematodes (PCN), making it vital for developing novel nematicides.
Q: How does this synthesis route ensure stereochemical control?
A: The route employs a Charette chiral ligand-induced asymmetric cyclopropanation reaction to establish the critical chiral cyclopropane moiety with high selectivity, ensuring the structural integrity required for biological activity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes readily available starting materials like geraniol acetate and avoids expensive transition metal catalysts, relying instead on robust reactions like SN2 substitution and Wittig olefination that are amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solanoeclepin A Intermediate SA-01 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of Intermediate SA-01 in the development of next-generation nematicides and pharmaceutical agents. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate the innovative chemistry described in CN111187186B from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical or field trials to full market launch. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify structural integrity and chiral purity at every stage of production.
We invite you to collaborate with us to leverage this advanced synthesis for your product pipeline. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can improve your margins. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in bringing this powerful bioactive scaffold to the global market efficiently and sustainably.
