Advanced Copper-Catalyzed Synthesis of Alpha-Fluorocarboxylic Acids for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex fluorinated scaffolds, which are pivotal for enhancing the metabolic stability and bioavailability of drug candidates. Patent CN112794782B introduces a groundbreaking approach for the synthesis of fluorine-containing carboxylic acid compounds, specifically targeting the challenging activation of inert carbon-fluorine bonds. This technology leverages a copper-catalyzed system to facilitate the selective carboxylation of alpha-trifluoromethyl styrenes using carbon dioxide as a sustainable C1 source. By operating under mild reaction conditions ranging from 50°C to 100°C, this process circumvents the extreme thermal requirements typically associated with C-F bond cleavage. For R&D directors and process chemists, this represents a significant leap forward in accessing alpha-fluorocarboxyl structures, which are prevalent in numerous bioactive molecules, without relying on expensive precious metal catalysts or hazardous reagents.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-fluorocarboxylic acids has been plagued by significant synthetic hurdles that hinder efficient commercial production. Traditional routes often involve multi-step sequences with poor atom economy, requiring harsh reaction conditions that can degrade sensitive functional groups present in complex drug intermediates. Many existing methods rely on stoichiometric amounts of toxic reagents or expensive noble metal catalysts like palladium, which not only inflate the cost of goods but also introduce stringent purification requirements to meet residual metal specifications for pharmaceutical applications. Furthermore, the inherent strength of the carbon-fluorine bond makes selective activation difficult, often leading to mixtures of products with low regioselectivity and diastereoselectivity. These limitations result in low overall yields and generate substantial chemical waste, posing challenges for both economic viability and environmental compliance in large-scale manufacturing settings.
The Novel Approach
In stark contrast, the methodology disclosed in CN112794782B offers a streamlined, single-pot solution that directly transforms readily available alpha-trifluoromethyl styrenes into valuable alpha-difluorobutenoate esters. This novel approach utilizes a cooperative catalytic system involving a copper catalyst, a bidentate phosphine ligand such as Xantphos, and a boron reagent to activate the C-F bond selectively. The reaction proceeds efficiently at moderate temperatures, typically around 80°C, in polar aprotic solvents like NMP. Crucially, this method employs carbon dioxide, an abundant and inexpensive greenhouse gas, as the carboxylating agent, thereby adding significant value to a waste product. The process exhibits exceptional functional group tolerance, accommodating substrates with halogens, ethers, and bulky aromatic systems without compromising yield or selectivity, making it highly suitable for the diverse needs of modern medicinal chemistry.
Mechanistic Insights into Copper-Catalyzed C-F Bond Carboxylation
The success of this transformation lies in the intricate interplay between the copper catalyst and the boron reagent, which facilitates a unique mechanistic pathway for C-F bond activation. The cycle initiates with the formation of a copper-boron species, which undergoes conjugate addition to the unsaturated bond of the trifluoromethyl styrene substrate. This addition is followed by a critical beta-fluorine elimination step, generating a vinyl boron intermediate and a copper-fluoride species. Subsequent transmetallation between the vinyl boron species and the copper center produces a reactive organocopper nucleophile. This nucleophile then attacks the electrophilic carbon of the carbon dioxide molecule, forming a carboxylate intermediate. Finally, protonation or alkylation yields the desired alpha-fluorocarboxylic acid derivative while regenerating the active copper catalyst. This mechanism ensures high chemoselectivity, breaking only one specific C-F bond while preserving others, which is essential for maintaining the structural integrity of polyfluorinated motifs.
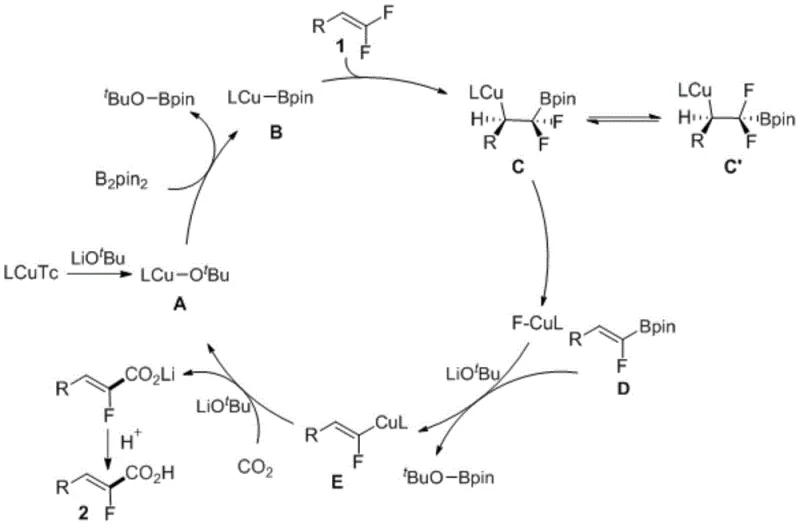
Understanding the impurity profile is vital for scaling this reaction to commercial levels, and the mechanistic clarity provided by this patent aids significantly in that regard. The high selectivity of the copper-boron addition minimizes the formation of side products such as hydrodefluorination byproducts or homocoupling dimers, which are common in other transition metal-catalyzed reactions. The use of specific ligands like Xantphos stabilizes the copper center, preventing catalyst decomposition and ensuring consistent turnover numbers throughout the reaction duration. Moreover, the mild basic conditions employed prevent the epimerization of chiral centers that might be present in more complex substrates, although the current scope focuses primarily on achiral styrenes. For quality control teams, this predictable reaction pathway means that impurity profiles are consistent and manageable, simplifying the downstream purification processes such as flash column chromatography or crystallization.
How to Synthesize 2,2-Difluoro-3-butenoic Acid Esters Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to atmospheric conditions and reagent quality to maximize yield and reproducibility. The protocol typically involves charging a dry reaction vessel with the substrate, boron reagent, copper catalyst, ligand, and base under an inert atmosphere, often utilizing a glovebox for precise handling of air-sensitive components. Following the initial mixing, the system is purged with carbon dioxide to establish the necessary reaction atmosphere before the addition of the anhydrous solvent. The reaction is then heated to the optimal temperature, usually 80°C, for an extended period to ensure complete conversion of the starting material. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Load a dry Schlenk tube with the alpha-trifluoromethyl styrene substrate, bis(2-methyl-2,4-pentanediol)borate, CuCl catalyst, Xantphos ligand, and potassium methoxide base under inert atmosphere.
- Purge the reaction vessel with CO2 gas three times to ensure a carbon dioxide atmosphere, then add anhydrous NMP solvent.
- Heat the mixture to 80°C for 24 hours, then add 1-iodohexane and continue heating for 4 hours before purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed carboxylation technology presents compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from precious metal catalysts to earth-abundant copper salts drastically reduces the raw material costs associated with catalytic loading, eliminating the volatility linked to the pricing of metals like palladium or rhodium. Additionally, the use of carbon dioxide as a feedstock not only lowers reagent costs but also aligns with corporate sustainability goals by utilizing a greenhouse gas, potentially qualifying the process for green manufacturing incentives. The mild reaction conditions reduce energy consumption compared to high-temperature alternatives, further contributing to operational cost reductions in utility expenses. These factors combined create a more resilient and cost-effective supply chain for producing high-value fluorinated intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive copper chloride significantly lowers the direct material costs per kilogram of product. Furthermore, the high atom economy of incorporating CO2 means less waste is generated, reducing the costs associated with waste disposal and treatment. The simplified workup procedure, which often requires only standard extraction and chromatography, minimizes the need for specialized purification equipment or consumables. This holistic reduction in input costs allows for a more competitive pricing structure for the final pharmaceutical intermediates without sacrificing quality or purity standards required by regulatory bodies.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including alpha-trifluoromethyl styrenes and common copper salts, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction against moisture and air, once initiated, allows for more flexible manufacturing schedules and reduces the likelihood of batch failures due to minor environmental fluctuations. By securing a reliable source of these key building blocks, manufacturers can ensure consistent production timelines and meet the demanding delivery schedules of downstream pharmaceutical clients. This reliability is crucial for maintaining long-term contracts and fostering trust with international partners.
- Scalability and Environmental Compliance: The mild thermal profile of this reaction makes it inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton commercial production. The absence of highly toxic reagents or extreme pressures simplifies the engineering controls required for large-scale reactors, facilitating a smoother technology transfer process. Moreover, the utilization of CO2 contributes to a lower carbon footprint for the manufacturing process, helping companies meet increasingly stringent environmental regulations and corporate social responsibility targets. This alignment with green chemistry principles enhances the marketability of the final products in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows. The responses cover catalyst selection, substrate compatibility, and the environmental impact of the process.
Q: What are the key advantages of using copper catalysts over precious metals for this transformation?
A: The use of copper catalysts, such as CuCl, significantly reduces raw material costs compared to palladium or rhodium systems. Furthermore, copper offers excellent chemoselectivity for C-F bond activation under mild conditions, avoiding the harsh temperatures often required by other metal systems.
Q: Does this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates broad substrate scope. Substituents such as halogens (Cl, Br), methoxy groups, esters, and even bulky groups like tert-butyl or naphthyl rings are well-tolerated, yielding the corresponding alpha-fluorocarboxylic acid derivatives with good efficiency.
Q: How does this process contribute to environmental sustainability?
A: This methodology utilizes carbon dioxide (CO2) as a C1 building block, effectively converting a greenhouse gas into value-added organic molecules. Additionally, the avoidance of toxic heavy metals and the use of mild reaction conditions align with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Difluoro-3-butenoic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the copper-mediated carboxylation described in CN112794782B for the future of fluorinated chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorinated intermediate delivered meets the highest global standards. We are committed to leveraging our technical expertise to optimize these copper-catalyzed processes for maximum efficiency and yield, providing our clients with a secure and high-quality supply of critical building blocks.
We invite you to collaborate with us to explore how this sustainable synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments for your next project. Let us help you navigate the complexities of fluorine chemistry with confidence and precision.
