Scalable Synthesis of 4-[4-(Methylsulfonyl)phenyl]-5-phenyl-2-(Trifluoromethyl)-1H-Imidazole for Pharma
Introduction to Advanced Imidazole Synthesis
The development of selective Cyclooxygenase-2 (COX-II) inhibitors represents a pivotal advancement in anti-inflammatory pharmacotherapy, aiming to mitigate gastrointestinal side effects associated with traditional NSAIDs. Central to this therapeutic class is the 4,5-diarylimidazole scaffold, specifically 4-[4-(methylsulfonyl)phenyl]-5-phenyl-2-(trifluoromethyl)-1H-imidazole, which exhibits potent inhibitory activity with an IC50 value below 0.2 μM. However, the commercial viability of such high-value pharmaceutical intermediates is often constrained by legacy synthetic routes that suffer from poor reproducibility and severe safety hazards. The patent CN102603646B discloses a robust, five-step synthetic methodology that addresses these critical bottlenecks by replacing toxic cyanide-based transformations and aggressive permanganate oxidations with safer, iron-catalyzed alternatives. This technical insight report analyzes the mechanistic advantages and supply chain implications of this novel route for global procurement and R&D stakeholders.
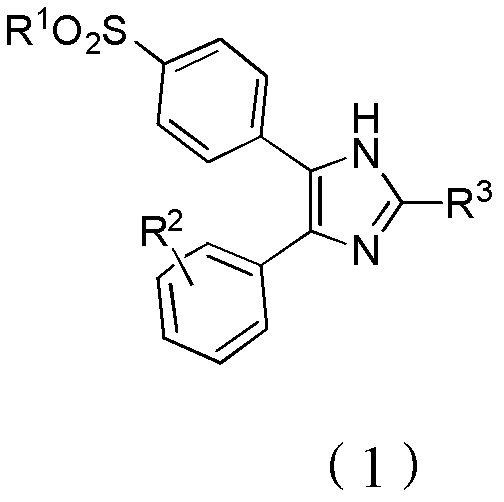
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4,5-diarylimidazole core has relied heavily on benzoin condensation strategies or Wittig olefination sequences, both of which present significant barriers to safe and efficient manufacturing. As illustrated in the prior art reaction schemes, early methods involved the condensation of aldehydes followed by oxidation and ring closure, a pathway notoriously plagued by the generation of hydrogen cyanide (HCN) during the hydrolysis of cyanohydrin intermediates. This not only necessitates expensive scrubbing systems and specialized containment infrastructure but also introduces severe regulatory hurdles regarding hazardous waste disposal. Furthermore, alternative routes utilizing Wittig reactions require subsequent oxidation steps using potassium permanganate (KMnO4), a strong oxidant known to indiscriminately cleave carbon-carbon double bonds, leading to unpredictable degradation of the molecular scaffold and difficult-to-remove impurities that compromise final API purity.
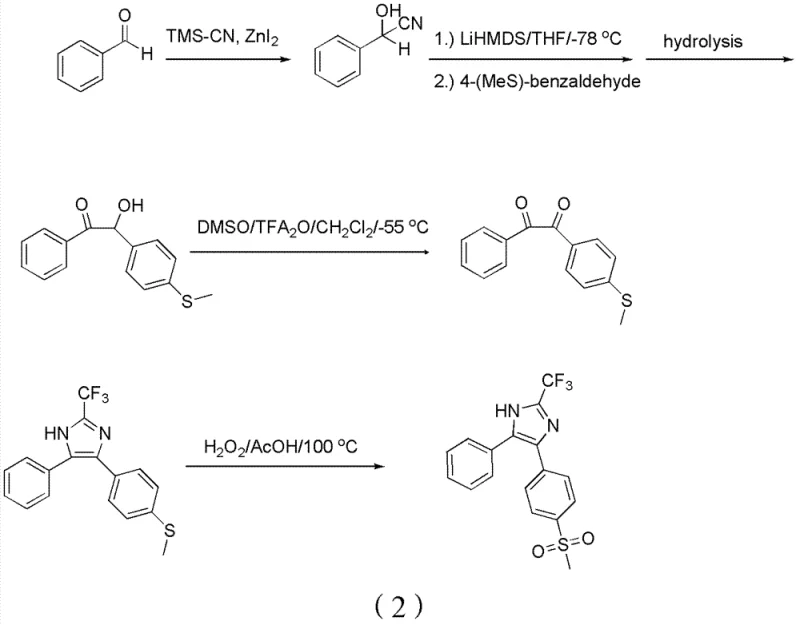
The Novel Approach
In stark contrast to these hazardous legacy processes, the methodology outlined in CN102603646B introduces a streamlined sequence that begins with a straightforward Friedel-Crafts acylation of thioanisole. This initial step establishes the carbon framework using abundant commodity chemicals, avoiding the need for complex aldehyde preparations. The core innovation lies in the strategic conversion of a 1,3-diketone intermediate directly into a 1,2-diketone using a catalytic amount of iron salts and tert-butyl nitrite. This transformation bypasses the need for stoichiometric heavy metal oxidants entirely. By deferring the introduction of the sulfone functionality until the final step, the synthesis protects the sensitive aromatic rings from over-oxidation. This logical disconnection of the synthetic pathway ensures that each intermediate is stable and easily purifiable, thereby enhancing the overall reliability of the manufacturing process for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Iron-Catalyzed Oxidative Transformation
The centerpiece of this synthetic strategy is the oxidative conversion of 1-(4-methylthiophenyl)-3-phenyl-1,3-dione to its corresponding 1,2-diketone congener. Mechanistically, this reaction leverages the Lewis acidity of ferric chloride (FeCl3) to activate the tert-butyl nitrite (TBN) oxidant. Unlike radical-based oxidations that can lead to non-selective degradation, this coordination complex facilitates a controlled electron transfer that specifically targets the active methylene position between the two carbonyl groups. The reaction proceeds under mild thermal conditions, typically between 20°C and 40°C, which minimizes thermal stress on the molecule and prevents the formation of polymeric byproducts often seen in high-temperature oxidations. This selectivity is paramount for maintaining the integrity of the trifluoromethyl group introduced in the subsequent cyclization step, ensuring that the final product meets the stringent purity specifications required for clinical applications.
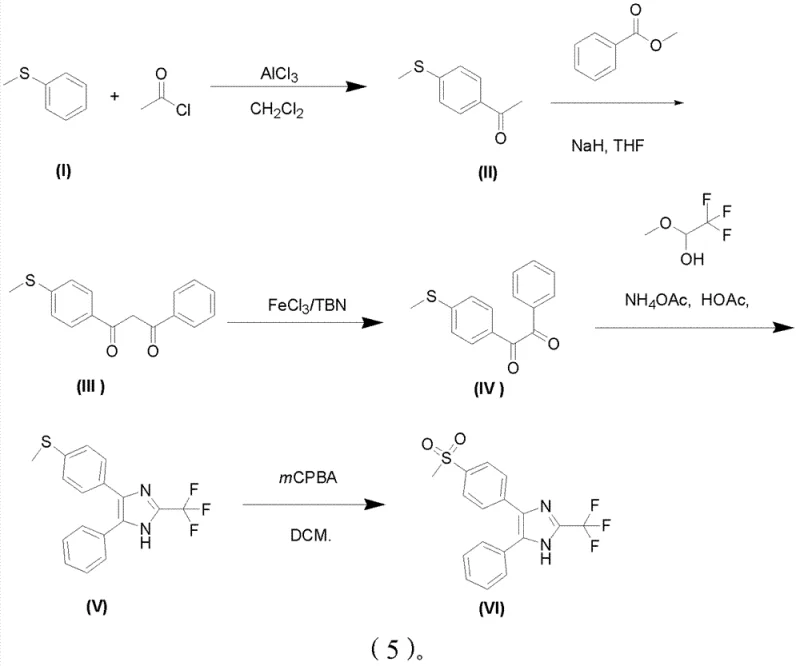
Furthermore, the final oxidation step converting the methylthio group to the methylsulfonyl moiety demonstrates exceptional chemoselectivity. In previous art, the presence of electron-rich aromatic rings often led to ring oxidation when using strong oxidants. However, by utilizing reagents such as m-chloroperoxybenzoic acid (mCPBA) or hydrogen peroxide in a controlled dichloromethane solvent system at 0°C, the process achieves exclusive oxidation of the sulfur atom. This precision eliminates the need for extensive chromatographic purification to remove ring-oxidized impurities, significantly reducing solvent consumption and processing time. The ability to perform this oxidation on the fully formed imidazole ring, rather than on a fragile alkene precursor as in Wittig routes, underscores the thermodynamic stability of this new pathway and its suitability for cost reduction in pharmaceutical intermediate manufacturing.
How to Synthesize 4-[4-(Methylsulfonyl)phenyl]-5-phenyl-2-(Trifluoromethyl)-1H-Imidazole Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control across five distinct operational units. The process initiates with the acylation of thioanisole, followed by a base-mediated Claisen condensation to build the diketone backbone. The critical iron-catalyzed oxidation is then performed, followed by cyclization with trifluoroacetaldehyde methyl hemiacetal and ammonium acetate in refluxing acetic acid. The detailed standardized operating procedures, including specific molar ratios such as the 1:0.1 to 0.5 ratio of diketone to iron catalyst and the precise quenching protocols for the exothermic oxidation steps, are essential for reproducing the high yields reported in the patent data. For a comprehensive breakdown of the reaction parameters and workup procedures, please refer to the technical guide below.
- Perform Friedel-Crafts acylation of thioanisole with acetyl chloride using AlCl3 or FeCl3 catalyst in DCM to form 4-methylthioacetophenone.
- Execute Claisen condensation between 4-methylthioacetophenone and methyl benzoate using NaH in THF to generate 1,3-diketone intermediate.
- Oxidize the 1,3-diketone to 1,2-diketone using tert-butyl nitrite and catalytic FeCl3, followed by cyclization with trifluoroacetaldehyde methyl hemiacetal.
- Finalize the synthesis by oxidizing the methylthio group to methylsulfonyl using mCPBA or hydrogen peroxide in dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers profound advantages by decoupling production from volatile supply chains associated with specialized reagents. The reliance on bulk commodity chemicals such as thioanisole, acetyl chloride, and methyl benzoate ensures that raw material availability remains stable even during market fluctuations. Moreover, the substitution of expensive and regulated cyanide sources with inexpensive iron salts drastically simplifies the environmental compliance profile of the manufacturing facility. This shift not only reduces the capital expenditure required for safety infrastructure but also accelerates the regulatory approval timeline for new drug master files (DMFs), providing a competitive edge in time-to-market for downstream API developers seeking a reliable pharmaceutical intermediate supplier.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic cyanide reagents results in substantial cost savings across the entire production lifecycle. By utilizing ferric chloride, a widely available industrial chemical, instead of specialized oxidants, the direct material costs are significantly lowered. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity of the reactions minimizes the loss of valuable starting materials to side products, thereby improving the overall mass balance and economic efficiency of the process.
- Enhanced Supply Chain Reliability: The robustness of this five-step sequence enhances supply continuity by reducing the number of critical path dependencies. Traditional routes often face bottlenecks due to the limited suppliers of hazardous reagents like TMSCN or the logistical challenges of transporting strong oxidants. In contrast, the reagents employed in this novel method are standard inventory items for most fine chemical manufacturers. This ubiquity ensures that production schedules are less susceptible to external disruptions, guaranteeing consistent delivery timelines for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing solvents like dichloromethane and acetic acid that are easily recovered and recycled in standard distillation units. The avoidance of heavy metal waste streams simplifies wastewater treatment protocols, aligning with increasingly stringent global environmental regulations. This green chemistry approach not only mitigates regulatory risk but also supports the sustainability goals of modern pharmaceutical companies, making it an ideal candidate for large-volume production of complex polymer additives or specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthetic pathway. These responses are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield optimization, impurity control, and safety protocols. Understanding these nuances is critical for process chemists evaluating technology transfer feasibility.
Q: How does this synthesis method improve safety compared to traditional benzoin condensation routes?
A: Traditional methods often rely on benzoin condensation followed by hydrolysis, which generates highly toxic hydrogen cyanide (HCN) as a byproduct. The patented method described in CN102603646B completely eliminates the use of cyanide sources, significantly reducing hazardous waste treatment costs and improving operator safety during commercial manufacturing.
Q: Why is the iron-catalyzed oxidation step critical for product purity?
A: Conventional routes utilizing potassium permanganate (KMnO4) for oxidation pose a risk of cleaving carbon-carbon double bonds in sensitive intermediates, leading to complex impurity profiles. The novel approach employs a mild iron-catalyzed system with tert-butyl nitrite to convert 1,3-diketones to 1,2-diketones selectively, preserving the structural integrity of the scaffold and ensuring a cleaner impurity spectrum for downstream API synthesis.
Q: Is this process suitable for large-scale production of COX-II inhibitor intermediates?
A: Yes, the process is designed for industrial scalability. It utilizes commodity chemicals such as thioanisole, acetyl chloride, and inexpensive iron salts rather than precious metal catalysts. The reaction conditions operate at moderate temperatures (ranging from 0°C to 110°C) and use standard organic solvents like dichloromethane and acetic acid, facilitating straightforward technology transfer from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-[4-(Methylsulfonyl)phenyl]-5-phenyl-2-(Trifluoromethyl)-1H-Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of reaction kinetics and thermodynamics described in this patent is maintained at every scale. We operate state-of-the-art rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of 4-[4-(methylsulfonyl)phenyl]-5-phenyl-2-(trifluoromethyl)-1H-imidazole meets the stringent purity specifications demanded by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this iron-catalyzed route can optimize your bill of materials. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical COX-II inhibitor programs.
