Advanced Silver-Promoted Cyclization for Scalable 5-Trifluoromethyl Imidazole Production
Advanced Silver-Promoted Cyclization for Scalable 5-Trifluoromethyl Imidazole Production
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As detailed in the groundbreaking patent CN113735778A, a novel preparation method for 5-trifluoromethyl substituted imidazole compounds has been developed, addressing critical bottlenecks in the synthesis of these high-value intermediates. This technology leverages a transition metal silver oxide-promoted [3+2] cycloaddition reaction, utilizing cheap and easily obtainable starting materials such as trifluoroethylimidoyl chloride and imidates. The significance of this chemical architecture cannot be overstated, as the imidazole motif is pervasive in blockbuster pharmaceuticals ranging from antifungal agents like econazole to antihistamines like alcaftadine, as well as serving as critical ligands in coordination catalysis. 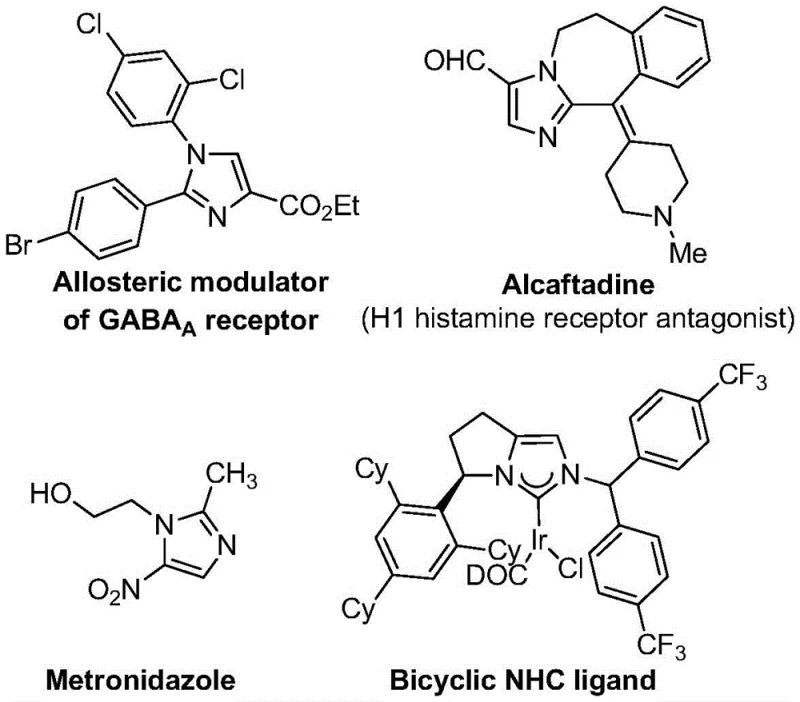
For R&D directors and process chemists, the ability to access diverse, fully substituted imidazole derivatives with high regioselectivity and purity is paramount. The disclosed method not only simplifies the synthetic route but also expands the substrate scope, allowing for the introduction of various aryl and alkyl substituents at the 1, 2, and 4 positions of the imidazole ring. This flexibility is crucial for structure-activity relationship (SAR) studies during lead optimization phases. Furthermore, the operational simplicity of the protocol, which avoids cryogenic conditions or exotic reagents, positions it as a highly attractive candidate for technology transfer from laboratory discovery to pilot plant operations, ensuring a reliable supply chain for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted imidazoles has been plagued by significant economic and technical hurdles that hinder efficient commercial manufacturing. Traditional methodologies often rely on the [3+2] cycloaddition of methylene amine ylides with trifluoromethyl-substituted imines, a pathway that necessitates the use of expensive and unstable precursors like trifluoro-acetaldehyde ethyl hemiacetal. The procurement of such specialized synthons not only inflates the cost of goods sold (COGS) but also introduces supply chain vulnerabilities due to their limited availability from bulk chemical vendors. Moreover, these conventional routes frequently suffer from moderate reaction efficiencies and narrow substrate tolerance, requiring extensive purification efforts to remove side products and unreacted starting materials. The reliance on harsh reaction conditions or sensitive catalysts further complicates the scale-up process, often resulting in inconsistent batch-to-batch quality and increased waste generation, which poses challenges for environmental compliance and overall process sustainability in large-scale API manufacturing.
The Novel Approach
In stark contrast to legacy techniques, the innovative strategy outlined in patent CN113735778A utilizes a robust silver oxide-promoted cyclization between trifluoroethylimidoyl chloride and imidates to construct the imidazole core with exceptional efficiency. This approach capitalizes on the high reactivity of the imidoyl chloride functionality, which serves as a versatile electrophile, coupling seamlessly with the nucleophilic imidate species under mild thermal conditions. 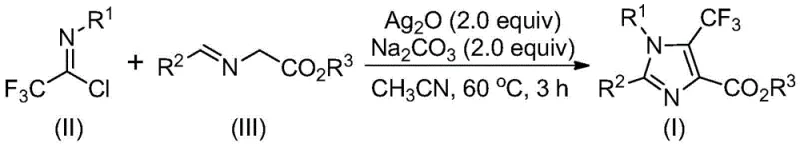
The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures around 60°C, eliminating the need for energy-intensive heating or cooling cycles. By employing sodium carbonate as a benign additive alongside the silver oxide promoter, the method ensures high conversion rates and minimizes the formation of undesirable byproducts. This streamlined process not only drastically reduces the number of unit operations required but also enhances the overall atom economy of the synthesis. The ability to achieve near-quantitative yields across a broad range of substrates, including those with electron-withdrawing and electron-donating groups, demonstrates the superior versatility of this novel pathway, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Silver-Promoted [3+2] Cycloaddition
The mechanistic pathway of this transformation involves a sophisticated sequence of bond-forming events orchestrated by the silver catalyst. Initially, the basic conditions facilitated by sodium carbonate promote an intermolecular carbon-carbon bond formation between the imidoyl chloride and the imidate, generating a bisimine intermediate. This key intermediate subsequently undergoes a silver-promoted intramolecular cyclization to form a 2-hydroimidazole species. The final and crucial step is the oxidative aromatization driven by silver oxide, which converts the dihydro-species into the fully aromatic 5-trifluoromethyl-substituted imidazole product. This oxidative step is particularly elegant as it utilizes the silver species itself as the terminal oxidant, avoiding the need for external stoichiometric oxidants that could generate hazardous waste streams. The precise control over oxidation states ensures that the sensitive trifluoromethyl group remains intact while the heterocyclic ring achieves full aromaticity, resulting in a thermodynamically stable and chemically robust final product.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining high product purity. The use of acetonitrile as the solvent provides an optimal polarity balance that solubilizes both organic substrates and inorganic salts, facilitating homogeneous reaction kinetics and preventing localized hot spots that could lead to decomposition. The tolerance of the system towards various functional groups, such as halogens (fluorine, bromine), alkyl chains (methyl, tert-butyl), and alkoxy groups, allows for the direct synthesis of complex analogues without the need for extensive protecting group strategies. 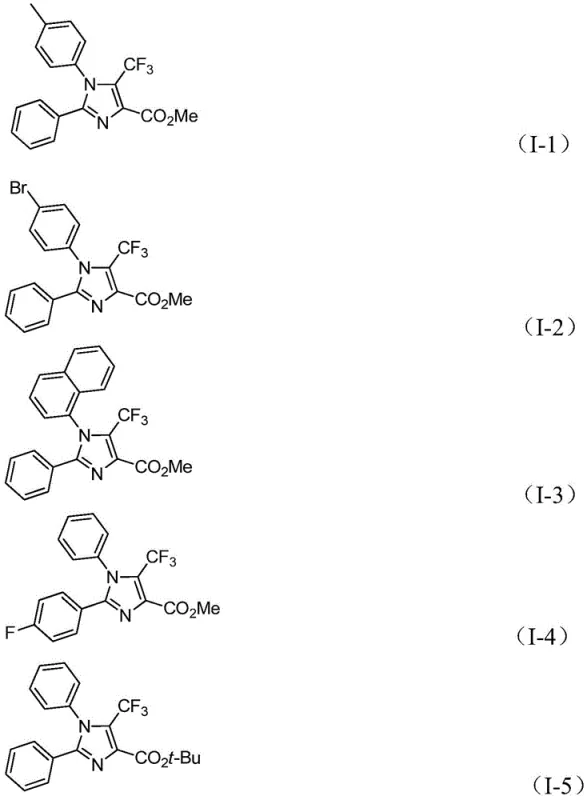
This broad functional group compatibility is essential for minimizing downstream purification burdens, as fewer side reactions mean a cleaner crude profile. For quality assurance teams, this translates to simpler analytical workflows and higher confidence in meeting stringent specification limits for residual metals and organic impurities, thereby accelerating the release of materials for clinical or commercial use.
How to Synthesize 5-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for maximum operational ease, requiring standard laboratory equipment and commercially available reagents. The protocol involves charging a reaction vessel with the trifluoroethylimidoyl chloride, the specific imidate derivative, silver oxide, and sodium carbonate in acetonitrile. The mixture is then heated to 60°C and stirred for approximately 3 hours, after which the reaction is deemed complete based on TLC or HPLC monitoring. Workup involves simple filtration to remove inorganic salts, followed by adsorption onto silica gel and purification via standard column chromatography.
- Combine trifluoroethylimidoyl chloride, imidate substrate, silver oxide promoter, and sodium carbonate additive in acetonitrile solvent.
- Heat the reaction mixture to 60°C and stir for 3 hours to facilitate the [3+2] cycloaddition and oxidative aromatization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from exotic, high-cost synthons to commodity chemicals like trifluoroethylimidoyl chloride and glycine-derived imidates represents a fundamental restructuring of the cost base. By leveraging widely available feedstocks, manufacturers can mitigate the risks associated with single-source suppliers and volatile pricing of niche reagents. This strategic sourcing advantage ensures a more predictable expenditure profile and protects against supply disruptions that could halt production lines. Furthermore, the simplified workflow reduces the demand for specialized reactor configurations, allowing existing multipurpose facilities to accommodate production without significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive trifluoro-acetaldehyde derivatives and the use of catalytic amounts of silver promoters relative to the value of the final product lead to substantial cost savings. The high reaction efficiency means less raw material is wasted, and the simplified purification process reduces solvent consumption and labor hours associated with chromatography or recrystallization. These factors collectively drive down the cost per kilogram of the active pharmaceutical ingredient (API) intermediate, enhancing profit margins in a competitive market landscape.
- Enhanced Supply Chain Reliability: Since the key starting materials are bulk chemicals with established global supply chains, lead times for raw material acquisition are significantly shortened. This reliability allows for more agile inventory management and just-in-time production strategies. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive parameters, ensuring consistent output volumes that meet delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from milligram to gram scales with consistent yields. The use of acetonitrile, a solvent with well-defined recovery and recycling protocols, aligns with green chemistry principles. Additionally, the avoidance of heavy metal catalysts that require complex removal steps simplifies waste treatment, reducing the environmental footprint and ensuring compliance with increasingly strict regulatory standards regarding metal residues in pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of using trifluoroethylimidoyl chloride over traditional synthons?
A: Unlike expensive trifluoro-acetaldehyde ethyl hemiacetal compounds often required in prior art, trifluoroethylimidoyl chloride is廉价 (cheap) and readily available, significantly lowering raw material costs while maintaining high reaction efficiency.
Q: How does the silver oxide promoter influence the reaction mechanism?
A: Silver oxide acts as both a base and an oxidant, facilitating the initial intermolecular carbon-carbon bond formation and driving the final oxidative aromatization step to yield the fully substituted imidazole ring without requiring harsh oxidizing agents.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes common organic solvents like acetonitrile, operates at moderate temperatures (60°C), and achieves near-quantitative yields, making it highly amenable to commercial scale-up from gram to multi-kilogram batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensively evaluated the silver-promoted cyclization route described in CN113735778A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl imidazole delivered meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable and cost-effective chemical solutions.
