Advanced Gold-Catalyzed Synthesis of 1,5-Benzodiazepine Derivatives for Commercial Scale-Up
Advanced Gold-Catalyzed Synthesis of 1,5-Benzodiazepine Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing complex heterocyclic scaffolds. Patent CN102690239A introduces a groundbreaking methodology for the synthesis of 1,5-benzodiazepine derivatives, a class of compounds renowned for their diverse pharmacological activities including anti-inflammatory, anticonvulsant, and anxiolytic properties. This innovation leverages a tandem intermolecular amination and cyclization process between o-phenylenediamine compounds and alkyne compounds, facilitated by a specialized gold catalyst system. Unlike traditional approaches that suffer from poor atom economy and significant waste generation, this novel route achieves 100% atom utilization, marking a significant leap forward in green chemistry principles for API intermediate manufacturing. The technology not only simplifies the synthetic workflow but also enhances the purity profile of the final products, addressing critical quality concerns for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-benzodiazepine derivatives has relied heavily on condensation reactions between o-phenylenediamines and activated carbonyl compounds such as α,β-unsaturated carbonyls, β-haloketones, or simple ketones. While these methods have served the industry for decades, they are inherently flawed due to their stoichiometric generation of by-products. For instance, condensation with ketones typically releases water molecules, while reactions involving haloketones produce hydrogen halides as waste. These副产物 (by-products) necessitate extensive downstream processing, including neutralization, extraction, and rigorous purification to meet pharmaceutical grade standards. Furthermore, the atom economy of these traditional routes is significantly compromised, leading to higher raw material consumption and increased environmental burden. The reliance on harsh conditions or unstable intermediates in some conventional protocols also poses challenges for process safety and scalability, often resulting in variable yields and inconsistent impurity profiles that complicate regulatory filings.
The Novel Approach
In stark contrast, the methodology disclosed in CN102690239A utilizes terminal alkynes as direct building blocks, reacting them with o-phenylenediamines in a catalytic cycle that incorporates every atom of the reactants into the final product structure. This addition-type reaction mechanism eliminates the formation of small molecule by-products entirely, achieving a theoretical atom economy of 100%. The process operates under remarkably mild conditions, typically between 0°C and 100°C, with optimal performance observed around 60°C. By bypassing the need for pre-functionalized ketone precursors, this approach shortens the synthetic sequence and reduces the overall carbon footprint of the manufacturing process. The versatility of the method is further demonstrated by its tolerance to a wide range of substituents on both the diamine and alkyne components, allowing for the rapid generation of diverse chemical libraries for drug discovery without the need for protecting group strategies or harsh reagents.
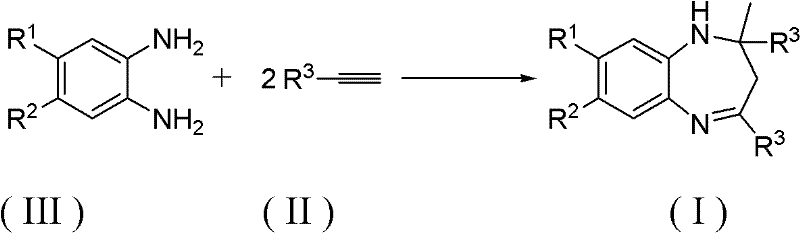
Mechanistic Insights into Gold-Catalyzed Hydroamination/Cyclization
The core of this technological advancement lies in the activation of the alkyne triple bond by a cationic gold species, which renders the carbon-carbon bond susceptible to nucleophilic attack by the amine groups of the o-phenylenediamine. The patent highlights a specific preference for gold(I) complexes ligated with bulky phosphines and paired with non-coordinating anions, such as [bis(trifluoromethanesulfonimide)][2-(dicyclohexylphosphino)]gold(I). This specific catalyst architecture stabilizes the reactive gold-alkyne π-complex while preventing catalyst deactivation through aggregation or poisoning by the amine substrate. The reaction proceeds through a sequential hydroamination followed by an intramolecular cyclization, effectively constructing the seven-membered diazepine ring in a single operational step. The choice of solvent plays a crucial role in stabilizing the ionic intermediates; chloroform is identified as the preferred medium, providing an ideal balance of solubility for organic substrates and compatibility with the gold catalyst system.
From an impurity control perspective, the absence of condensation by-products like water or acids fundamentally changes the purification landscape. In traditional syntheses, residual water can lead to hydrolysis of sensitive functional groups or promote oligomerization, while acidic by-products can cause salt formation or degradation of the basic nitrogen centers in the benzodiazepine ring. By eliminating these sources of contamination at the source, the new method ensures a cleaner crude reaction mixture. This inherent cleanliness reduces the load on purification columns and minimizes the risk of carry-over impurities that could affect the stability of the final API. Moreover, the high regioselectivity of the gold catalyst ensures that the addition occurs in a controlled manner, preventing the formation of isomeric by-products that are difficult to separate, thereby enhancing the overall optical and chemical purity of the synthesized derivatives.
How to Synthesize 1,5-Benzodiazepine Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize catalyst loading. The protocol involves dissolving the o-phenylenediamine and the substituted alkyne in a dry organic solvent, typically chloroform, under an inert atmosphere to prevent oxidation of the catalyst. The molar ratio of alkyne to diamine is optimized at approximately 2.5:1 to drive the reaction to completion without excessive waste of the alkyne starting material. Upon addition of the gold catalyst, the mixture is heated to a moderate temperature, generally around 60°C, and monitored via thin-layer chromatography (TLC) until the starting materials are fully consumed. The workup procedure is straightforward, involving the removal of solvent and direct purification via silica gel chromatography, leveraging the clean nature of the reaction profile.
- Prepare the reaction mixture by combining o-phenylenediamine compounds and substituted acetylene compounds in an organic solvent such as chloroform.
- Add the preferred gold catalyst, specifically [bis(trifluoromethanesulfonimide)][2-(dicyclohexylphosphino)]gold(I), to the mixture under inert atmosphere.
- Heat the reaction to 60°C and monitor progress via TLC until completion, then purify the product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this alkyne-based synthesis represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with complex intermediate manufacturing. The elimination of by-product formation translates directly into reduced waste disposal costs and lower solvent usage for washing and extraction steps, contributing to a leaner and more sustainable production model. Additionally, the use of alkynes as starting materials offers a significant logistical advantage; alkynes are often more readily available and stable than the corresponding α,β-unsaturated ketones or β-haloketones required in traditional routes, ensuring a more robust and continuous supply of raw materials. This reliability is crucial for maintaining uninterrupted production schedules for high-volume pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The 100% atom economy of this process means that virtually all purchased raw materials end up in the final product, drastically reducing the effective cost per kilogram of the active ingredient. By removing the need for stoichiometric activating agents or dehydrating agents, the bill of materials is simplified, and the cost of goods sold (COGS) is significantly lowered. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure decreases labor hours and equipment occupancy time, leading to substantial operational expenditure savings across the manufacturing lifecycle.
- Enhanced Supply Chain Reliability: Sourcing high-purity o-phenylenediamines and substituted alkynes is generally more straightforward than sourcing specialized enones or haloketones, which may have limited suppliers or long lead times. This broader supplier base for starting materials reduces the risk of single-source dependency and price volatility. The robustness of the gold-catalyzed method also allows for flexibility in raw material specifications, as the catalyst system can tolerate minor variations in substrate quality without compromising the final yield, thereby insulating the supply chain from minor upstream fluctuations.
- Scalability and Environmental Compliance: The absence of corrosive by-products like hydrogen halides eliminates the need for specialized corrosion-resistant reactors and extensive scrubbing systems for exhaust gas treatment. This simplifies the engineering requirements for scaling up from pilot plant to commercial tonnage production. Additionally, the reduced waste stream aligns perfectly with increasingly stringent environmental regulations regarding hazardous waste discharge, facilitating easier permitting and compliance auditing. The ability to run the reaction at moderate temperatures also enhances process safety, reducing the thermal hazards associated with large-scale exothermic condensation reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical benefits for industrial adoption. Understanding these nuances is essential for R&D teams evaluating process transfer and for procurement specialists assessing the long-term viability of this supply route.
Q: What are the primary advantages of using alkynes over ketones for synthesizing 1,5-benzodiazepines?
A: Using alkynes as starting materials offers 100% atom economy with no by-products like water or hydrogen halides, unlike traditional condensation methods with ketones which generate waste and require additional purification steps.
Q: Which catalyst system provides the highest yield and stability for this transformation?
A: The patent identifies [bis(trifluoromethanesulfonimide)][2-(dicyclohexylphosphino)]gold(I) as the preferred catalyst, offering high yields, excellent regioselectivity, and superior stability for storage and handling compared to other gold or transition metal salts.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method operates under mild conditions (40-80°C) using readily available starting materials and standard organic solvents like chloroform, making it highly suitable for commercial scale-up and consistent supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Benzodiazepine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this gold-catalyzed synthesis technology in modernizing the production of critical pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle homogeneous gold catalysis with the necessary containment and recovery systems to maximize efficiency and minimize metal residue, adhering to stringent purity specifications required by global regulatory bodies. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our 1,5-benzodiazepine derivatives meet the highest standards for downstream drug synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this innovative synthetic route for their specific pipeline needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, quantifying the potential economic benefits of switching to this atom-economical process. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects, ensuring your supply chain is built on the foundation of cutting-edge chemical innovation.
