Advanced Cinchona-Squaramide Hydrogen Bond Catalysts for Industrial Asymmetric Synthesis
Advanced Cinchona-Squaramide Hydrogen Bond Catalysts for Industrial Asymmetric Synthesis
The landscape of asymmetric catalysis has undergone a significant transformation with the introduction of novel organocatalytic systems that offer superior stereocontrol without relying on expensive transition metals. Patent CN102397793A details a groundbreaking class of cinchona base-squaramide hydrogen bond catalysts, representing a pivotal advancement in the field of chiral synthesis. These catalysts leverage the robust structural framework of cinchona alkaloids combined with the potent hydrogen-bonding capabilities of the squaramide moiety. For R&D directors and process chemists, this technology offers a compelling alternative to traditional thiourea-based systems, providing a pathway to high-purity pharmaceutical intermediates with exceptional enantioselectivity. The patent outlines a streamlined synthetic route that utilizes readily available starting materials, addressing critical supply chain concerns regarding raw material accessibility and cost stability.
The strategic integration of electron-withdrawing aryl groups on the squaramide nitrogen enhances the acidity of the hydrogen bond donors, thereby activating electrophiles more effectively than previous generations of catalysts. This design principle not only improves reaction rates but also tightens the chiral environment around the reactive center, leading to consistently high ee values across diverse substrate scopes. From a commercial perspective, the ability to achieve such high levels of stereochemical purity under mild conditions translates directly into reduced downstream processing costs and simplified purification protocols. As the pharmaceutical industry continues to demand greener and more efficient synthetic methodologies, the adoption of these cinchona-squaramide catalysts positions manufacturers at the forefront of sustainable chemical innovation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, chiral amine-(thio)urea bifunctional catalysts have served as the workhorses for many asymmetric transformations, yet they are not without significant drawbacks that hinder their widespread industrial adoption. Traditional thiourea catalysts often suffer from limited structural diversity and can exhibit weaker hydrogen-bonding interactions compared to their squaramide counterparts, which may result in lower enantioselectivity or the need for cryogenic reaction temperatures to achieve acceptable stereocontrol. Furthermore, the synthesis of complex thiourea derivatives can be cumbersome, frequently requiring multi-step sequences involving hazardous reagents like isothiocyanates, which pose safety risks and complicate waste management protocols. These factors collectively contribute to higher manufacturing costs and extended lead times, creating bottlenecks for procurement managers seeking reliable sources of high-performance chiral catalysts. Additionally, the flexibility of the thiourea backbone can sometimes lead to less defined transition states, compromising the consistency of chiral induction in large-scale batch processes.
The Novel Approach
In contrast, the novel approach described in the patent utilizes a squaramide scaffold that offers a rigid, planar structure capable of forming stronger and more directional hydrogen bonds. This structural rigidity is key to creating a well-defined chiral pocket that effectively differentiates between enantiotopic faces of the substrate. The synthesis strategy employs a straightforward two-step nucleophilic substitution reaction starting from dimethyl squarate, a commercially abundant and inexpensive reagent. 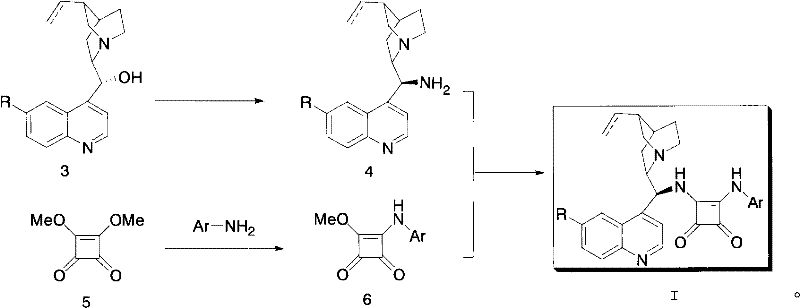 This method allows for the modular introduction of various functional groups on both the squaramide nitrogen and the cinchona backbone, enabling fine-tuning of the catalyst's electronic and steric properties to match specific reaction requirements. By operating under mild conditions, typically at room temperature in methanol, this novel approach drastically simplifies the manufacturing process, eliminating the need for specialized equipment or extreme thermal controls. The result is a robust catalytic system that delivers excellent yields and stereoselectivity while significantly lowering the barrier to entry for industrial scale-up.
This method allows for the modular introduction of various functional groups on both the squaramide nitrogen and the cinchona backbone, enabling fine-tuning of the catalyst's electronic and steric properties to match specific reaction requirements. By operating under mild conditions, typically at room temperature in methanol, this novel approach drastically simplifies the manufacturing process, eliminating the need for specialized equipment or extreme thermal controls. The result is a robust catalytic system that delivers excellent yields and stereoselectivity while significantly lowering the barrier to entry for industrial scale-up.
Mechanistic Insights into Bifunctional Hydrogen Bond Catalysis
The efficacy of these cinchona-squaramide catalysts stems from their unique bifunctional mechanism, which simultaneously activates both the nucleophile and the electrophile through distinct interaction sites. The tertiary amine within the quinuclidine ring of the cinchona skeleton acts as a Brønsted base, deprotonating or coordinating with the nucleophilic species to enhance its reactivity. Concurrently, the two NH protons of the squaramide unit serve as strong hydrogen bond donors, stabilizing the developing negative charge on the electrophile, such as a nitroalkene or a cyclic anhydride. This dual activation brings the reactants into close proximity within a highly organized chiral environment, ensuring that the bond formation occurs with precise stereochemical control. The rigidity of the squaramide ring prevents conformational freedom that could otherwise lead to non-selective background reactions, thereby maximizing the enantiomeric excess of the product.
This mechanistic advantage is vividly demonstrated in asymmetric Michael addition reactions, where the catalyst achieves remarkable levels of stereoinduction. 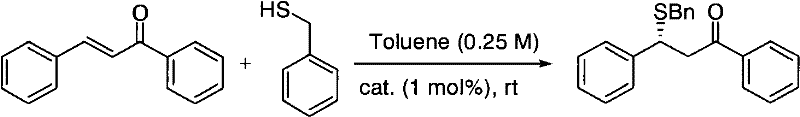 For instance, in the reaction between trans-chalcone and benzyl mercaptan, the catalyst facilitates the formation of the C-S bond with a yield of 97% and an ee value of 96% under ambient conditions. The electron-withdrawing nature of the aryl group attached to the squaramide nitrogen further amplifies the acidity of the NH protons, strengthening the hydrogen bond network and accelerating the reaction rate. This deep understanding of the catalytic cycle allows process chemists to predict substrate compatibility and optimize reaction parameters with greater confidence, reducing the trial-and-error phase typically associated with new method development. Such predictability is invaluable for maintaining consistent quality in the production of high-purity pharmaceutical intermediates.
For instance, in the reaction between trans-chalcone and benzyl mercaptan, the catalyst facilitates the formation of the C-S bond with a yield of 97% and an ee value of 96% under ambient conditions. The electron-withdrawing nature of the aryl group attached to the squaramide nitrogen further amplifies the acidity of the NH protons, strengthening the hydrogen bond network and accelerating the reaction rate. This deep understanding of the catalytic cycle allows process chemists to predict substrate compatibility and optimize reaction parameters with greater confidence, reducing the trial-and-error phase typically associated with new method development. Such predictability is invaluable for maintaining consistent quality in the production of high-purity pharmaceutical intermediates.
How to Synthesize Cinchona-Squaramide Catalysts Efficiently
The synthesis of these high-value organocatalysts is designed for operational simplicity, utilizing a convergent strategy that minimizes purification steps and maximizes overall throughput. The process begins with the preparation of a squaramide monoester intermediate, followed by coupling with a chiral amine derived from cinchona alkaloids. This modular approach allows for the rapid generation of a library of catalyst variants by simply changing the amine component, facilitating quick structure-activity relationship studies. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and isolation techniques, are outlined in the guide below to ensure reproducibility and safety in your laboratory.
- Prepare the squaramide intermediate by reacting dimethyl squarate with an aromatic amine (e.g., 3,5-dimethylaniline) in methanol at room temperature for 24 hours.
- Isolate the monomethyl ester intermediate via filtration and drying without requiring complex purification steps.
- React the isolated intermediate with a cinchona alkaloid amine (e.g., quinine amine) in methanol at room temperature to form the final bifunctional catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this cinchona-squaramide catalyst technology offers substantial strategic benefits that extend beyond mere technical performance. The reliance on commodity chemicals like dimethyl squarate and naturally derived cinchona alkaloids ensures a stable and resilient supply chain, mitigating the risks associated with scarce or geopolitically sensitive raw materials. The mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, leading to significant reductions in utility costs and carbon footprint. Furthermore, the absence of heavy metals in the catalyst structure removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet stringent regulatory limits for residual metals in pharmaceutical products. This simplification of the downstream process not only accelerates time-to-market but also enhances the overall sustainability profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of precious metal catalysts and hazardous reagents, which dramatically lowers the direct material costs and waste disposal expenses. By utilizing a simple precipitation and filtration method for product isolation, the process minimizes solvent consumption and reduces the load on recovery systems. This efficiency translates into a more competitive cost structure for the final chiral intermediates, allowing companies to maintain healthy margins even in price-sensitive markets. The elimination of complex purification chromatography steps further contributes to operational savings by reducing labor and consumable costs.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this catalyst system is straightforward, as both squaric acid derivatives and cinchona alkaloids are produced at a global scale with established supply networks. This abundance ensures that production schedules are not disrupted by raw material shortages, providing a reliable foundation for long-term planning. The robustness of the catalyst synthesis also means that quality variations between batches are minimized, ensuring consistent performance in downstream applications. This reliability is crucial for maintaining uninterrupted production lines and meeting strict delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in standard glassware without requiring specialized high-pressure or low-temperature equipment. This ease of scale-up facilitates a smooth transition from laboratory discovery to pilot plant and eventually to full commercial production. Additionally, the use of methanol as a primary solvent and the generation of minimal hazardous waste align with green chemistry principles, simplifying regulatory compliance and environmental permitting. The ability to operate under ambient conditions also enhances workplace safety, reducing the risk of thermal runaways or exposure to toxic vapors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of cinchona-squaramide catalysts in industrial processes. These insights are derived directly from the patent data and practical experience with similar organocatalytic systems, aiming to clarify potential concerns regarding stability, scope, and handling. Understanding these nuances is essential for making informed decisions about integrating this technology into your existing manufacturing workflows.
Q: What are the advantages of squaramide catalysts over traditional thiourea catalysts?
A: Squaramide catalysts possess a more rigid structure and stronger hydrogen-bonding donor ability compared to thioureas. This results in enhanced stereocontrol and often higher enantiomeric excess (ee) values in asymmetric reactions such as Michael additions and desymmetrization processes.
Q: Are the reaction conditions for synthesizing these catalysts suitable for large-scale production?
A: Yes, the synthesis involves mild conditions, typically utilizing methanol as a solvent at room temperature. The process avoids harsh reagents and extreme temperatures, facilitating easier scale-up and reducing energy consumption in industrial manufacturing.
Q: What types of asymmetric reactions can these catalysts facilitate?
A: These bifunctional catalysts are highly versatile and have been successfully applied in asymmetric Michael additions, desymmetrization of cyclic anhydrides, and other carbon-carbon or carbon-heteroatom bond-forming reactions requiring precise chiral induction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinchona-Squaramide Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern drug discovery and process development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst meets the highest standards of performance and consistency. We understand that the success of your asymmetric synthesis depends on the reliability of your catalyst supply, and we are committed to providing a partnership that supports your long-term growth and innovation goals.
We invite you to engage with our technical procurement team to discuss how our cinchona-squaramide catalysts can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this metal-free catalytic system. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project needs, allowing us to demonstrate our capability to deliver high-quality solutions that drive value across your entire supply chain.
