Advanced Copper-Catalyzed Synthesis of Nitro Aromatic Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of Nitro Aromatic Intermediates for Scalable Pharmaceutical Manufacturing
The landscape of organic synthesis is continuously evolving towards more sustainable and cost-effective methodologies, particularly in the production of complex pharmaceutical intermediates. A pivotal advancement in this domain is detailed in patent CN102516162A, which discloses a novel method for preparing copper-catalyzed nitroaryl (hetero) ring compounds. This technology leverages a pyridyl-directed C-H bond nitration strategy, utilizing copper acetate as an environmentally benign and economically superior catalyst compared to traditional precious metal systems. For R&D directors and procurement specialists, this represents a significant opportunity to optimize synthetic routes for nitro-functionalized heterocycles, which are critical building blocks in medicinal chemistry. The process achieves high regioselectivity and yield, addressing long-standing challenges in direct C-H functionalization while maintaining strict adherence to green chemistry principles.
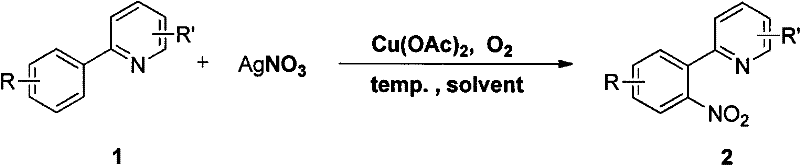
Traditional methods for introducing nitro groups onto aromatic rings have historically relied on classical electrophilic aromatic substitution, which often suffers from poor regiocontrol and harsh reaction conditions involving strong acids. Furthermore, recent transition-metal catalyzed approaches using Ruthenium, Rhodium, or Palladium, while effective, impose substantial financial burdens due to the high cost and supply volatility of these precious metals. The limitations of these conventional methods extend beyond mere economics; they often generate complex impurity profiles that require extensive downstream purification, thereby increasing the overall E-factor and waste disposal costs. In contrast, the novel approach outlined in the patent utilizes a base-metal copper catalyst that is not only abundant and inexpensive but also demonstrates exceptional compatibility with a wide range of functional groups. This shift from precious to base metal catalysis is a strategic move for any organization aiming to reduce the cost of goods sold (COGS) in their API manufacturing pipelines without compromising on quality or purity standards.
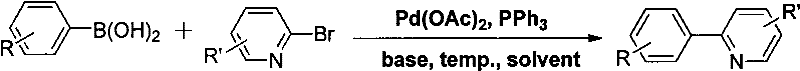
The mechanistic underpinnings of this copper-catalyzed transformation offer profound insights into how selectivity is achieved at the molecular level. The reaction proceeds through a directed C-H activation mechanism where the nitrogen atom of the pyridyl ring acts as an internal coordinating group. This coordination brings the copper catalyst into close proximity with the ortho-C-H bond of the adjacent phenyl ring, facilitating the formation of a stable metallacycle intermediate. This geometric constraint is the key to the observed high regioselectivity, effectively blocking nitration at other positions on the aromatic ring. Following C-H activation, the nitro group is transferred from the silver nitrate oxidant, likely regenerating the active copper species in the presence of oxygen or through the redox cycle involving silver. This precise control over the reaction trajectory minimizes the formation of regioisomeric impurities, which is a critical parameter for R&D teams focused on impurity profiling and regulatory compliance. The ability to predictably install a nitro group ortho to a pyridyl moiety opens up new retrosynthetic disconnections for complex drug candidates.
How to Synthesize Nitro Aryl Compounds Efficiently
The practical implementation of this synthesis route involves a two-step sequence that is highly amenable to standard laboratory and pilot plant operations. Initially, the requisite 2-arylpyridine substrate is constructed via a palladium-catalyzed Suzuki-Miyaura cross-coupling between substituted phenylboronic acids and 2-bromopyridine derivatives. This precursor synthesis is robust, utilizing aqueous conditions and inorganic bases like potassium phosphate, ensuring high purity of the starting material before the nitration step. The subsequent nitration is performed under elevated temperatures in halogenated solvents, specifically optimized to maximize conversion and selectivity. The detailed standardized synthesis steps, including precise stoichiometry and workup procedures, are provided in the guide below to ensure reproducibility and safety during scale-up efforts.
- Prepare the 2-arylpyridine substrate via Suzuki coupling of phenylboronic acid and 2-bromopyridine using Pd(OAc)2 and K3PO4 in water at 80°C.
- Combine the substrate with silver nitrate (1.5 equiv) and copper acetate (50 mol%) in 1,2,3-trichloropropane solvent.
- Heat the reaction mixture to 130°C under stirring until TLC indicates complete consumption of the starting material, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed nitration methodology offers transformative benefits for procurement managers and supply chain heads who are constantly pressured to optimize margins and secure raw material availability. The primary driver for cost reduction lies in the substitution of expensive precious metal catalysts with commodity-grade copper salts. Palladium and rhodium prices are subject to extreme market fluctuations and geopolitical supply risks, whereas copper remains a stable and affordable alternative. By eliminating the need for costly ligands often associated with noble metal catalysis, the overall reagent cost per kilogram of product is significantly decreased. Furthermore, the high selectivity of the reaction reduces the burden on purification processes, leading to lower solvent consumption and reduced waste treatment expenses, which directly contributes to a leaner and more sustainable manufacturing cost structure.
- Cost Reduction in Manufacturing: The economic advantage of this process is anchored in the drastic reduction of catalyst costs. Unlike palladium or platinum systems that require sophisticated ligands and rigorous metal scavenging steps to meet residual metal specifications in APIs, the copper-based system is inherently more economical. The removal of expensive heavy metal catalysts simplifies the downstream processing, potentially eliminating the need for specialized resin treatments or complex extraction protocols designed to lower ppm levels of toxic metals. This streamlining of the purification train translates into substantial operational savings and a faster time-to-market for new chemical entities, allowing companies to allocate resources more efficiently across their R&D portfolios.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and commoditized reagents such as copper acetate and silver nitrate. These materials are produced on a massive industrial scale globally, ensuring that procurement teams do not face the bottlenecks often associated with specialized organometallic catalysts. The robustness of the reaction conditions, which tolerate a variety of electronic substituents on the aromatic ring, means that a single platform technology can be applied to synthesize a diverse library of intermediates. This versatility reduces the need for multiple specialized suppliers and simplifies inventory management, thereby enhancing the overall reliability and continuity of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction temperatures and solvents that are manageable in large-scale reactors. The use of 1,2,3-trichloropropane as a solvent, while requiring appropriate handling, allows for high-temperature operations that drive reaction kinetics favorably. From an environmental standpoint, the shift towards base-metal catalysis aligns with increasingly stringent global regulations regarding heavy metal discharge and sustainability goals. The high atom economy and selectivity of the reaction minimize the generation of hazardous by-products, facilitating easier compliance with environmental protection standards and reducing the ecological footprint of the manufacturing facility, which is a key metric for modern corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed nitration technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of substrate scope, catalyst loading, and workup procedures is essential for successfully transferring this methodology from the bench to commercial production. We encourage technical teams to review these details thoroughly to assess the feasibility of integrating this route into their existing synthetic platforms.
Q: What are the advantages of using copper catalysts over palladium for C-H nitration?
A: Copper catalysts offer significant cost advantages due to the abundance of copper compared to precious metals like palladium or rhodium. Additionally, copper systems often exhibit distinct selectivity profiles and reduced environmental toxicity, aligning better with green chemistry principles for large-scale manufacturing.
Q: How does the pyridyl directing group influence the reaction selectivity?
A: The pyridyl nitrogen atom coordinates with the copper center, facilitating the activation of the ortho-C-H bond on the adjacent phenyl ring. This chelation assistance ensures high regioselectivity, preventing the formation of meta- or para-nitrated isomers that are common in traditional electrophilic aromatic substitution.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes robust reaction conditions (120-140°C) and commercially available reagents like silver nitrate and copper acetate. The workup involves standard extraction and chromatography, which can be adapted for continuous flow or large batch processing, ensuring reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitro Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the copper-catalyzed nitration pathway described in patent CN102516162A and possesses the technical capability to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nitro aromatic intermediates delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this copper-catalyzed method can reduce your overall production costs. Please contact us today to obtain specific COA data for similar compounds and to discuss route feasibility assessments that will accelerate your development timelines and secure your supply chain for the future.
