Advanced Manufacturing of High Enantioselective Pyrazolamine Compounds for Global Pharma
Advanced Manufacturing of High Enantioselective Pyrazolamine Compounds for Global Pharma
The pharmaceutical and agrochemical industries are increasingly demanding chiral intermediates with exceptional optical purity to ensure drug efficacy and safety. Patent CN107540616B introduces a groundbreaking methodology for the preparation of high enantioselective pyrazolamine compounds, specifically targeting the synthesis of chiral pyrazole derivatives that serve as critical building blocks for next-generation fungicides and therapeutic agents. This technology addresses the long-standing challenge of constructing chiral pyrazole cores without resorting to inefficient chiral resolution techniques. By leveraging a novel organocatalytic asymmetric aldol reaction followed by a robust pyrazole ring construction strategy, this process delivers the target molecule with an impressive 94% enantiomeric excess (ee). For R&D directors and procurement specialists, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of complex heterocyclic amines.
Furthermore, the strategic implementation of this synthesis route offers substantial advantages in supply chain stability. Traditional methods often rely on scarce chiral pool materials or expensive enzymatic resolutions that bottleneck production capacity. In contrast, the approach detailed in CN107540616B utilizes a synthetically accessible chiral guanidine catalyst to induce asymmetry early in the sequence. This early-stage stereocontrol ensures that the chirality is preserved throughout subsequent transformations, including the critical palladium-catalyzed amination step. The result is a streamlined process that minimizes waste and maximizes throughput, positioning manufacturers to meet the rigorous quality standards of global regulatory bodies while maintaining competitive pricing structures for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral pyrazole derivatives has been plagued by significant inefficiencies, primarily stemming from the reliance on racemic synthesis followed by chiral resolution. This traditional approach inherently caps the maximum theoretical yield at 50%, necessitating the recycling or disposal of the unwanted enantiomer, which drastically inflates production costs and environmental waste. Moreover, conventional routes often employ harsh reaction conditions or stoichiometric amounts of chiral auxiliaries that are difficult to recover, leading to complex purification protocols and inconsistent batch-to-batch optical purity. For supply chain managers, these limitations translate into volatile lead times and unpredictable inventory levels, as the separation of enantiomers often requires specialized chromatography columns that are not easily scalable to multi-ton production volumes.
The Novel Approach
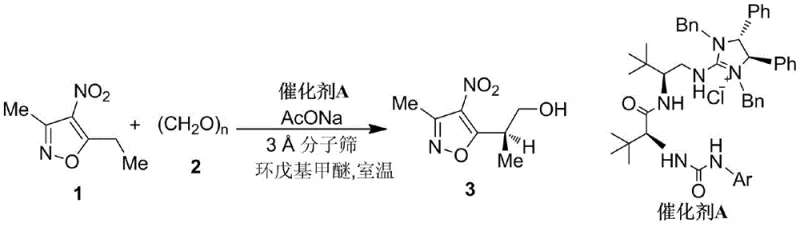
The innovative pathway disclosed in the patent overcomes these hurdles by employing a direct asymmetric catalytic strategy that constructs the chiral center with high fidelity from the outset. The process begins with an enantioselective aldol-type reaction between a nitroisoxazole and paraformaldehyde, catalyzed by a specialized amino acid-derived guanidine salt. This key transformation establishes the stereocenter with 97% ee, setting a high bar for purity that is maintained through the subsequent ring-opening and cyclization steps to form the pyrazole core. The entire sequence is designed for operational simplicity, utilizing common solvents like diethyl ether and THF, and avoiding cryogenic conditions except for a specific reduction step, thereby facilitating easier technology transfer to commercial manufacturing sites.
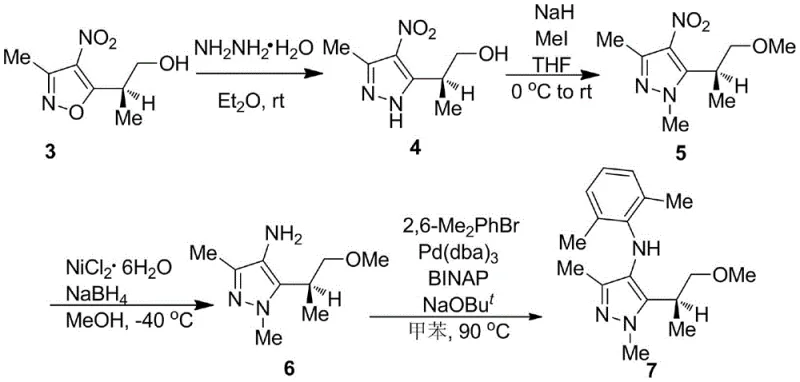
As illustrated in the reaction scheme, the conversion of the chiral isoxazole intermediate to the final pyrazolamine involves a logical progression of functional group manipulations that preserve stereochemical integrity. The use of hydrazine hydrate for ring transformation and sodium borohydride for nitro reduction demonstrates a commitment to using cost-effective reagents without compromising selectivity. This novel approach not only enhances the overall yield compared to resolution-based methods but also simplifies the impurity profile, making downstream processing significantly more manageable for quality control teams focused on meeting stringent residual solvent and heavy metal specifications.
Mechanistic Insights into Asymmetric Organocatalysis and Pd-Coupling
The cornerstone of this synthesis is the asymmetric organocatalytic step that generates the chiral isoxazole alcohol intermediate. The mechanism involves the activation of the nitroisoxazole substrate by the chiral guanidine catalyst, which acts as a bifunctional promoter to facilitate the nucleophilic attack on the formaldehyde equivalent. The steric bulk of the tert-leucine-derived backbone in the catalyst creates a well-defined chiral environment, effectively shielding one face of the reacting species and directing the formation of the desired enantiomer. This precise molecular recognition is crucial for achieving the reported 97% ee, as even minor deviations in catalyst structure can lead to significant erosion of optical purity. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as temperature and concentration to maximize catalyst turnover numbers.
Following the establishment of chirality, the subsequent steps involve a nickel-mediated reduction and a palladium-catalyzed cross-coupling, both of which require careful mechanistic consideration to prevent racemization. The reduction of the nitro group using NiCl2·6H2O and NaBH4 at -40°C is particularly sensitive; the low temperature suppresses potential side reactions and protects the labile chiral center adjacent to the reaction site. Finally, the Buchwald-Hartwig amination couples the amino-pyrazole with 2,6-dimethylbromobenzene using a Pd(0)/BINAP system. The choice of BINAP as the ligand is critical here, as its wide bite angle facilitates the reductive elimination step, ensuring high conversion rates even with the sterically hindered ortho-substituted aryl bromide, thus delivering the final product with 94% ee.
How to Synthesize (S)-N-(2,6-Dimethylphenyl)-5-(2-(1-methoxypropane))-1,3-dimethyl-1H-pyrazol-4-amine Efficiently
The synthesis of this high-value chiral intermediate is executed through a concise four-step sequence that balances reaction efficiency with ease of purification. The process initiates with the organocatalytic assembly of the chiral scaffold, followed by heterocyclic ring construction, functional group reduction, and final C-N bond formation. Each step has been optimized to minimize byproduct formation, ensuring that the intermediate streams require minimal workup before proceeding to the next stage. For technical teams looking to implement this route, it is essential to strictly adhere to the specified molar ratios and temperature controls, particularly during the reduction phase, to maintain the high enantiomeric excess established in the first step. Detailed standardized operating procedures for each transformation are outlined below to ensure reproducibility.
- Perform asymmetric aldol condensation of 3-methyl-5-ethyl-4-nitroisoxazole with paraformaldehyde using a chiral guanidine catalyst to obtain chiral isoxazole alcohol (97% ee).
- React the chiral isoxazole intermediate with hydrazine hydrate to form the pyrazole core, followed by N-methylation to protect the nitrogen.
- Reduce the nitro group to an amine using NiCl2 and NaBH4 at low temperature (-40°C), then couple with 2,6-dimethylbromobenzene using Pd(dba)3/BINAP.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative benefits for procurement managers seeking to optimize the cost structure of complex intermediate manufacturing. By eliminating the need for chiral resolution, the process effectively doubles the theoretical yield of the usable enantiomer compared to traditional racemic routes, leading to substantial raw material savings. The use of earth-abundant nickel for the reduction step, rather than more expensive noble metals, further contributes to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the robustness of the organocatalyst allows for potential recycling or the use of lower catalyst loadings in large-scale batches, driving down the cost of goods sold (COGS) significantly while maintaining high purity standards required for GMP production.
- Cost Reduction in Manufacturing: The direct asymmetric synthesis avoids the 50% yield loss inherent in chiral resolution, effectively maximizing the utility of every kilogram of starting material purchased. Furthermore, the avoidance of expensive enzymatic kits or specialized chiral columns for purification results in significant operational expenditure savings. The streamlined workflow reduces the number of unit operations, which lowers energy consumption and labor costs associated with extended processing times. These factors combine to create a highly competitive pricing model for the final chiral pyrazolamine product.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as nitroisoxazoles and paraformaldehyde, mitigates the risk of supply disruptions often associated with exotic chiral pool reagents. The synthetic route is designed for scalability, meaning that production can be ramped up from pilot plant to commercial scale without requiring major re-engineering of the process infrastructure. This flexibility ensures consistent delivery schedules and allows for rapid response to fluctuating market demands for agrochemical and pharmaceutical precursors.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents and avoids the generation of hazardous heavy metal waste streams typical of stoichiometric chiral reagents. The high atom economy of the catalytic steps aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental regulations simplifies waste disposal logistics and reduces the regulatory burden on the supply chain, ensuring uninterrupted production continuity in regions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral pyrazolamine synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the method's capabilities and constraints. For partners considering this route for their supply chain, these answers provide a foundational overview of the process performance and quality metrics.
Q: What is the enantiomeric excess (ee) achieved in this pyrazolamine synthesis?
A: The patented process achieves a high enantiomeric excess of 97% ee in the initial asymmetric step and maintains 94% ee in the final pyrazolamine product, eliminating the need for costly chiral resolution.
Q: Why is the nickel-mediated reduction performed at -40°C?
A: The low temperature (-40°C) is critical during the NiCl2/NaBH4 reduction step to prevent racemization of the chiral center and to control the exothermic reaction, ensuring high purity of the amino intermediate.
Q: Is this process scalable for commercial production of agrochemical intermediates?
A: Yes, the route utilizes commercially available starting materials and standard catalytic systems (Pd/BINAP), making it highly suitable for scale-up from kilogram to multi-ton production without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of safe and effective pharmaceuticals and agrochemicals. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR instrumentation to verify enantiomeric excess and chemical identity at every stage of production.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-quality chiral pyrazolamine compounds for your global operations.
