Advanced Axial Chiral Styrene Tertiary Amine Thiourea Catalysts for High-Enantioselectivity Manufacturing
Advanced Axial Chiral Styrene Tertiary Amine Thiourea Catalysts for High-Enantioselectivity Manufacturing
In the rapidly evolving landscape of asymmetric organocatalysis, the demand for highly efficient and stereoselective catalysts remains a critical bottleneck for the production of complex pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN112264091A, which discloses a novel class of axially chiral styrene tertiary amine thiourea catalysts. These innovative structures, represented generally by formulas 3 and 4, represent a paradigm shift from traditional carbon-centered chiral catalysts. By integrating an axially chiral styrene segment derived from oxindoles with a chiral tertiary amine thiourea motif, this technology offers a robust solution for controlling stereochemistry in challenging organic transformations. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic advantages of these dual-chirality systems is essential for optimizing synthetic routes.
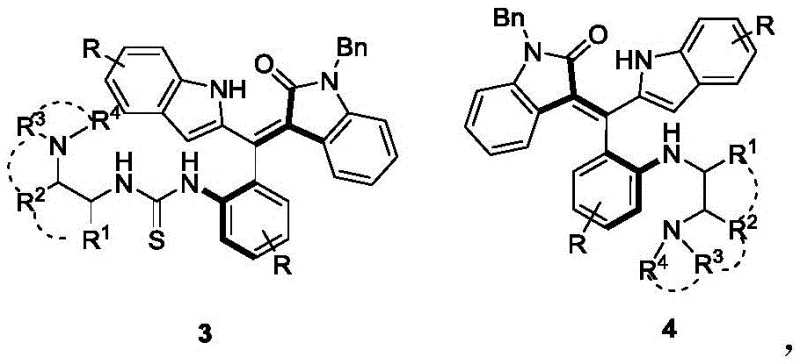
The strategic design of these catalysts addresses long-standing limitations in enantioselective synthesis. Traditional chiral tertiary amine thiourea catalysts often rely exclusively on point chirality (carbon chiral centers), which can sometimes lack the rigidity required for high-fidelity stereocontrol in bulky substrates. In contrast, the catalysts described in CN112264091A leverage axial chirality—a form of stereoisomerism arising from the spatial arrangement of groups about an axis—to create a more constrained and predictable chiral pocket. This structural innovation not only enhances the catalytic effect but also broadens the substrate scope, making it a valuable asset for the commercial scale-up of complex pharmaceutical intermediates. The ability to access two distinct configurational isomers (formulas 3 and 4) from a single synthetic sequence further adds to the versatility of this platform.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of organocatalysis has been dominated by catalysts possessing central chirality, such as those based on cinchona alkaloids or binaphthyl skeletons. While effective in many contexts, these conventional systems often struggle with specific reaction classes where the chiral environment is not sufficiently rigid to differentiate between transition states effectively. For instance, prior art documents like Adv. Synth. Catal. 2007 and Chem. Commun. 2007 describe axial chiral binaphthyl-based thioureas, yet these often exhibit suboptimal stereoselectivity control and limited catalytic turnover in certain [4+2] cyclization reactions. Furthermore, the synthesis of highly specialized chiral ligands often involves multi-step sequences requiring stringent conditions, which drives up the cost and complexity of manufacturing. For procurement managers focused on cost reduction in fine chemical manufacturing, the reliance on scarce or difficult-to-synthesize chiral pools can be a significant supply chain vulnerability.
The Novel Approach
The novel approach presented in the patent data utilizes an oxindole-derived axially chiral styrene framework as the core scaffold. This design choice is transformative because it combines the proven hydrogen-bonding activation capability of the thiourea moiety with the steric bulk and rigidity of the axially chiral backbone. The synthesis is remarkably streamlined, proceeding through a mild two-step sequence at ambient temperature (25°C). This contrasts sharply with older methods that might require cryogenic temperatures or hazardous reagents. By achieving high enantioselectivity (up to 93% ee) and excellent yields (up to 98%) in model reactions, this new class of catalysts demonstrates superior performance over commercially available alternatives like catalysts 7, 8, and 9. This performance leap translates directly into higher process efficiency and reduced waste, key metrics for any sustainable chemical operation.
Mechanistic Insights into Dual-Activation Organocatalysis
The exceptional performance of these axially chiral catalysts can be attributed to their sophisticated dual-activation mechanism. In the asymmetric [4+2] cyclization of p-methoxyphenyl-substituted o-methylenebenzoquinone with malononitrile, the catalyst operates by simultaneously activating both the electrophile and the nucleophile. The thiourea moiety forms strong hydrogen bonds with the electron-deficient quinone methide (electrophile), thereby lowering its LUMO energy and increasing its reactivity. Concurrently, the tertiary amine group interacts with the acidic proton of the malononitrile (nucleophile), enhancing its nucleophilicity. This bifunctional activation brings the reactants into close proximity within a well-defined chiral environment created by the axially chiral styrene backbone.
Crucially, the axial chirality imposes a specific geometric constraint that dictates the facial selectivity of the attack. Unlike flexible carbon chains, the rigid styrene-oxindole axis prevents free rotation, locking the catalyst into a conformation that favors the formation of one enantiomer over the other. This rigidity is the key to the observed high enantiomeric excess values. For example, catalyst 3a achieved 93% ee, while catalyst 3d reached 91% ee in the same transformation. This level of control minimizes the formation of unwanted diastereomers and enantiomers, simplifying downstream purification processes. For quality control teams, this means a cleaner impurity profile and a more robust process capable of meeting stringent regulatory specifications for high-purity pharmaceutical intermediates.
How to Synthesize Axial Chiral Styrene Tertiary Amine Thiourea Catalyst Efficiently
The preparation of these high-value catalysts is designed for operational simplicity and scalability, avoiding the need for exotic equipment or extreme conditions. The process begins with the activation of the axially chiral amine precursor using thiophosgene, followed by immediate coupling with a chiral diamine. The entire sequence is performed in common organic solvents like dichloromethane, facilitating easy solvent recovery and recycling. The mild reaction temperature of 25°C ensures energy efficiency and safety, while the use of pyridine as a base scavenges the generated HCl effectively. Detailed standardized synthesis steps for replicating this high-performance catalyst are provided in the guide below.
- React the axially chiral styrene compound (Formula 1) with thiophosgene in dichloromethane with pyridine at 25°C to form an isothiocyanate intermediate.
- Add the chiral tertiary amine diamine (Formula 2) to the intermediate solution in dichloromethane and stir at 25°C to complete the thiourea formation.
- Concentrate the reaction mixture and purify the resulting catalysts (Formula 3 and 4) using silica gel column chromatography with a dichloromethane/methanol eluent.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this catalytic technology offers compelling strategic advantages beyond mere technical performance. The synthesis route described in the patent is inherently cost-effective due to its brevity and the use of commodity chemicals. The elimination of transition metals removes the need for expensive metal scavengers and rigorous heavy metal testing, which are significant cost drivers in API manufacturing. Furthermore, the ability to generate two different catalytic isomers from the same intermediate streamlines inventory management, allowing manufacturers to switch between enantiomers without changing the raw material supply base. This flexibility is crucial for maintaining supply chain reliability in the face of market fluctuations.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of precious metal catalysts or cryogenic cooling systems, which are capital-intensive and operationally costly. By operating at ambient temperature and utilizing standard silica gel chromatography for purification, the process significantly lowers the barrier to entry for production. The high yields observed (up to 98%) mean that less raw material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final catalyst. Additionally, the simplified workup procedure reduces labor hours and solvent consumption, contributing to substantial overall cost savings in the production of chiral building blocks.
- Enhanced Supply Chain Reliability: The starting materials for this catalyst, including oxindole derivatives and chiral diamines, are readily available from established chemical suppliers, reducing the risk of raw material shortages. The robustness of the reaction conditions—tolerant to minor variations in temperature and stoichiometry—ensures consistent batch-to-batch quality, which is vital for long-term supply contracts. By adopting a catalyst that is easier to manufacture and purify, companies can reduce lead times for high-purity pharmaceutical intermediates and ensure a steady flow of materials to downstream synthesis units, thereby mitigating the risk of production delays.
- Scalability and Environmental Compliance: The process is inherently green, utilizing mild conditions and avoiding toxic heavy metals, which aligns with increasingly strict environmental regulations. The simplicity of the two-step synthesis makes it highly amenable to scale-up from gram to kilogram scales without significant re-engineering of the process. The use of dichloromethane, while requiring careful handling, allows for efficient recycling, minimizing waste generation. This scalability ensures that the technology can meet the demands of commercial production, supporting the commercial scale-up of complex pharmaceutical intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral catalyst technology. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, offering a clear perspective on its practical utility.
Q: What distinguishes this axial chiral catalyst from conventional carbon-centered chiral catalysts?
A: Unlike conventional catalysts that rely solely on carbon chiral centers, this novel catalyst incorporates an axially chiral styrene backbone derived from oxindoles. This dual-chirality design provides a more rigid and defined chiral environment, resulting in significantly improved stereoselectivity control and catalytic efficiency in asymmetric reactions.
Q: What are the optimal reaction conditions for the catalyst synthesis?
A: The synthesis is conducted under remarkably mild conditions. Both the activation step with thiophosgene and the subsequent coupling with the diamine occur at room temperature (25°C) in dichloromethane. This eliminates the need for cryogenic conditions or harsh reagents, simplifying the operational protocol.
Q: Can this catalyst be applied to large-scale asymmetric cyclization reactions?
A: Yes, the catalyst has demonstrated high efficacy in asymmetric [4+2] cyclization reactions, achieving yields up to 98% and enantiomeric excess (ee) values up to 93%. The simple purification via silica gel chromatography and the use of common solvents make it highly suitable for scale-up in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Thiourea Catalyst Supplier
The development of axially chiral styrene tertiary amine thiourea catalysts represents a significant advancement in the field of asymmetric organocatalysis, offering a powerful tool for the synthesis of enantiopure compounds. At NINGBO INNO PHARMCHEM, we recognize the critical importance of such technologies in driving innovation within the pharmaceutical and fine chemical sectors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of catalyst or intermediate meets the highest global standards. We are committed to bridging the gap between academic innovation and industrial reality.
We invite R&D directors and procurement managers to explore how our expertise can accelerate your project timelines. Whether you require custom synthesis of these novel catalysts or assistance in optimizing their application in your specific reaction workflows, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can enhance your supply chain efficiency and product quality.
