Advanced Catalytic Oxidation for High-Purity Alpha-Beta Dicarbonyl-1,2,3-Triazole Intermediates
Introduction to Next-Generation Triazole Synthesis
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for more efficient and sustainable pathways to complex scaffolds. A pivotal advancement in this domain is detailed in patent CN113912556B, which discloses a robust method for synthesizing α,β-dicarbonyl-1,2,3-triazole compounds. These structures serve as critical building blocks for a wide array of bioactive molecules, including imidazoles, quinoxalines, and triazines, making them indispensable in modern drug discovery pipelines. The patented technology represents a paradigm shift from traditional multi-step sequences to a streamlined, copper-catalyzed oxidative coupling strategy. By leveraging readily available aryl butanones or aryl butanols as starting materials, this approach circumvents the logistical and economic bottlenecks associated with acquiring specialized precursors like acid chlorides and silyl alkynes. For R&D directors and procurement specialists alike, understanding the nuances of this innovation is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
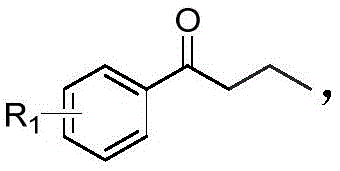
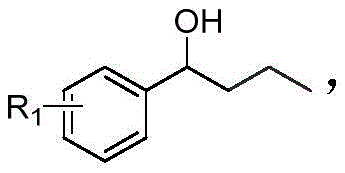
Historically, the construction of α,β-dicarbonyl-1,2,3-triazole cores has been fraught with synthetic challenges that hinder scalability and cost-efficiency. Conventional literature methods typically rely on a convoluted sequence beginning with the reaction of acid chlorides and hexamethylsilylacetylene to generate trimethylsilyl acetylene ketone intermediates. This initial step is problematic due to the high cost and limited commercial availability of hexamethylsilylacetylene, creating a significant barrier for large-scale production. Furthermore, the subsequent conversion to a mono-carbonyl triazole via reaction with organic azides, followed by a final oxidation step to introduce the second carbonyl group, results in a low overall yield. Each additional isolation and purification stage in this multi-step protocol not only consumes valuable time and resources but also increases the potential for impurity accumulation, complicating the downstream processing required for pharmaceutical grade materials.
The Novel Approach
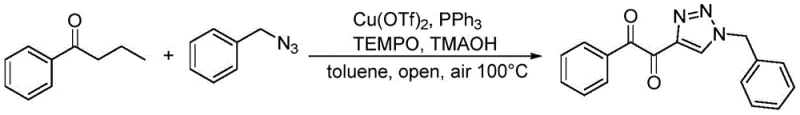
In stark contrast to these legacy techniques, the methodology outlined in CN113912556B offers a direct and atom-economical solution. The novel approach utilizes a one-pot tandem reaction where aryl butanone or aryl butanol serves as the direct precursor, reacting with organic azides in the presence of a copper catalyst, TEMPO oxidant, and a base. This strategy effectively merges the oxidation and cyclization events into a single operational unit, drastically simplifying the workflow. As illustrated in the reaction scheme below, the process operates under mild thermal conditions (90-100°C) using air as the terminal oxidant, which is a substantial improvement over stoichiometric chemical oxidants. This transition not only enhances the safety profile of the manufacturing process but also aligns with green chemistry principles by minimizing waste generation. For a reliable pharmaceutical intermediate supplier, adopting such a streamlined route translates directly into improved throughput and reduced lead times for clients.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
To fully appreciate the technical sophistication of this synthesis, one must delve into the catalytic cycle that drives the transformation. The reaction mechanism, depicted in the following diagram, initiates with the oxidation of the alcohol substrate (if aryl butanol is used) to the corresponding ketone via a Cu(II)/air system, ensuring quantitative conversion to the active carbonyl species. Subsequently, the Cu(II) species facilitates the generation of a carbon-centered radical intermediate at the alpha-position of the ketone through a single-electron transfer process. This radical species is then intercepted by the TEMPO radical or a copper-TEMPO complex, leading to further oxidation and the formation of an enolate-like intermediate. The presence of triphenylphosphine (PPh3) acts as a crucial ligand, stabilizing the copper center and modulating its redox potential to sustain the catalytic turnover. Finally, the activated dicarbonyl species undergoes a [3+2] cycloaddition with the organic azide, facilitated by the Lewis acidic nature of the copper catalyst, to forge the triazole ring with high regioselectivity.
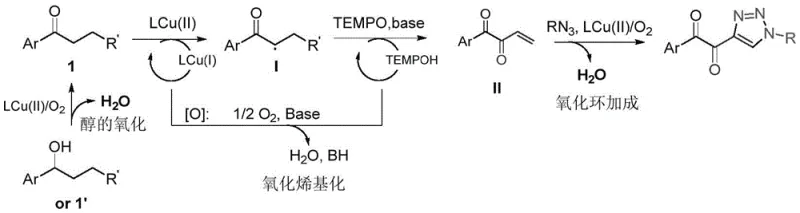
Beyond the primary catalytic cycle, the control of impurities is a critical aspect of this mechanism that appeals to quality-focused R&D teams. The use of tetramethylammonium hydroxide (TMAOH) as a base ensures a controlled pH environment that promotes the desired cyclization while suppressing side reactions such as over-oxidation or polymerization of the radical intermediates. The specific choice of Cu(OTf)2 as the catalyst, as evidenced by comparative examples in the patent, provides a balance of Lewis acidity and solubility that minimizes the formation of metal-complexed byproducts which are often difficult to remove. This mechanistic clarity allows for precise tuning of reaction parameters, such as the molar ratio of azide to ketone (optimized at 1:1.5 to 1:2), to maximize the yield of the target α,β-dicarbonyl-1,2,3-triazole. Understanding these subtleties is vital for scaling the process from gram-scale laboratory experiments to multi-kilogram commercial batches without compromising purity specifications.
How to Synthesize Alpha,Beta-Dicarbonyl-1,2,3-Triazole Efficiently
Implementing this synthesis in a practical setting requires adherence to specific operational protocols to ensure reproducibility and safety. The patent details a straightforward procedure where the substrates and catalysts are combined in toluene, a solvent chosen for its ability to dissolve both organic substrates and inorganic salts while maintaining a suitable boiling point for the reaction temperature. The process is designed to be operationally simple, requiring no inert atmosphere gloveboxes, as the reaction thrives under open air conditions. This accessibility makes it an ideal candidate for technology transfer to manufacturing sites equipped with standard glass-lined reactors. For those seeking to replicate or license this technology, the following guide outlines the critical steps derived from the patent's preferred embodiments, ensuring that the high yields reported (up to 92%) can be consistently achieved.
- Mix aryl butanone or aryl butanol with organic azide, copper salt (e.g., Cu(OTf)2), TEMPO, triphenylphosphine, and tetramethylammonium hydroxide in toluene.
- Heat the reaction mixture to 90-100°C and stir under an air atmosphere for 16-24 hours to facilitate oxidative coupling.
- Upon completion, extract with ethyl acetate, separate the organic phase, concentrate, and purify via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain heads tasked with optimizing costs and ensuring continuity. The primary driver of value lies in the drastic simplification of the raw material portfolio. By eliminating the need for exotic silyl reagents and acid chlorides, the process relies on commodity chemicals that are widely available from multiple global vendors, thereby mitigating supply risk. Furthermore, the use of air as the oxidant removes the cost and safety hazards associated with purchasing, storing, and disposing of hazardous stoichiometric oxidants. This shift towards safer, cheaper reagents inherently lowers the cost of goods sold (COGS), allowing for more competitive pricing strategies in the final API market. The robustness of the copper catalytic system also implies a longer catalyst life and potentially lower metal loading, contributing further to economic efficiency.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from fundamental changes in the reaction design rather than marginal optimizations. By consolidating multiple synthetic steps into a single pot, the method eliminates the need for intermediate isolations, which are typically the most labor-intensive and solvent-consuming parts of chemical manufacturing. The removal of expensive silyl protecting groups and the reliance on atmospheric oxygen as a free oxidant significantly reduces the material cost per kilogram of product. Additionally, the simplified work-up procedure, involving basic extraction and crystallization, reduces the demand for specialized purification equipment and silica gel, leading to substantial cost savings in downstream processing operations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of aryl butanones and aryl butanols, which are stable, shelf-stable commodities with established global supply chains. Unlike the moisture-sensitive acid chlorides and silyl alkynes required by older methods, these starting materials do not require stringent storage conditions or expedited shipping, reducing logistics costs and the risk of spoilage during transit. The tolerance of the reaction to air and moisture further enhances reliability, as it reduces the dependency on specialized inert gas infrastructure, making the process adaptable to a wider range of manufacturing facilities worldwide and ensuring consistent delivery schedules for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by its inherent safety and environmental profile. The absence of highly exothermic steps or toxic byproducts simplifies heat management and waste treatment protocols. Toluene, the solvent of choice, is a well-understood industrial solvent with established recovery and recycling streams, minimizing environmental impact. The catalytic nature of the copper system means that heavy metal waste is minimized compared to stoichiometric methods, aiding in compliance with increasingly strict environmental regulations regarding heavy metal discharge. This alignment with green chemistry principles not only future-proofs the manufacturing process but also enhances the brand value of the final pharmaceutical products by reducing their overall carbon footprint.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis, we have compiled a set of questions based on the specific data points and claims within the patent documentation. These answers are designed to provide clarity on the operational parameters and the comparative advantages of this technology over existing alternatives. Whether you are evaluating the feasibility of in-house production or assessing a potential supplier's capabilities, understanding these details is crucial for making informed strategic decisions regarding your intermediate sourcing strategy.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike conventional methods requiring difficult-to-source acid chlorides and hexamethylsilylacetylene, this patent utilizes readily available aryl butanones or butanols. It simplifies the process into a one-pot reaction using air as the terminal oxidant, significantly reducing step count and raw material costs.
Q: Which copper catalyst provides the optimal yield for this transformation?
A: According to the experimental data in patent CN113912556B, Copper(II) triflate (Cu(OTf)2) demonstrates superior catalytic activity compared to other salts like Cu(OAc)2 or CuBr2, achieving yields up to 92% under optimized conditions.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the use of common solvents like toluene, ambient air as the oxidant, and robust copper catalysis suggests high scalability. The elimination of sensitive silyl reagents and harsh acidic conditions enhances safety and operational feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Dicarbonyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex heterocycles requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We understand the critical importance of stringent purity specifications in the pharmaceutical sector and utilize rigorous QC labs to verify that every batch of α,β-dicarbonyl-1,2,3-triazole meets the highest international standards. Our capability to implement the advanced copper-catalyzed oxidative coupling described in CN113912556B positions us as a strategic partner capable of delivering high-quality intermediates with consistent reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined route for your specific API candidates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in your supply chain strategy and accelerate your drug development timelines.
