Advanced Iodine-Catalyzed Synthesis for Commercial-Scale Production of High-Purity 5-Trifluoromethyl Triazoles
The recently granted Chinese patent CN110467579B introduces an innovative methodology for synthesizing 5-trifluoromethyl-substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This novel approach addresses longstanding challenges in the production of these critical building blocks, which serve as key intermediates in numerous therapeutic agents including antifungal medications and enzyme inhibitors.
Advanced Reaction Mechanism and Impurity Profile Control
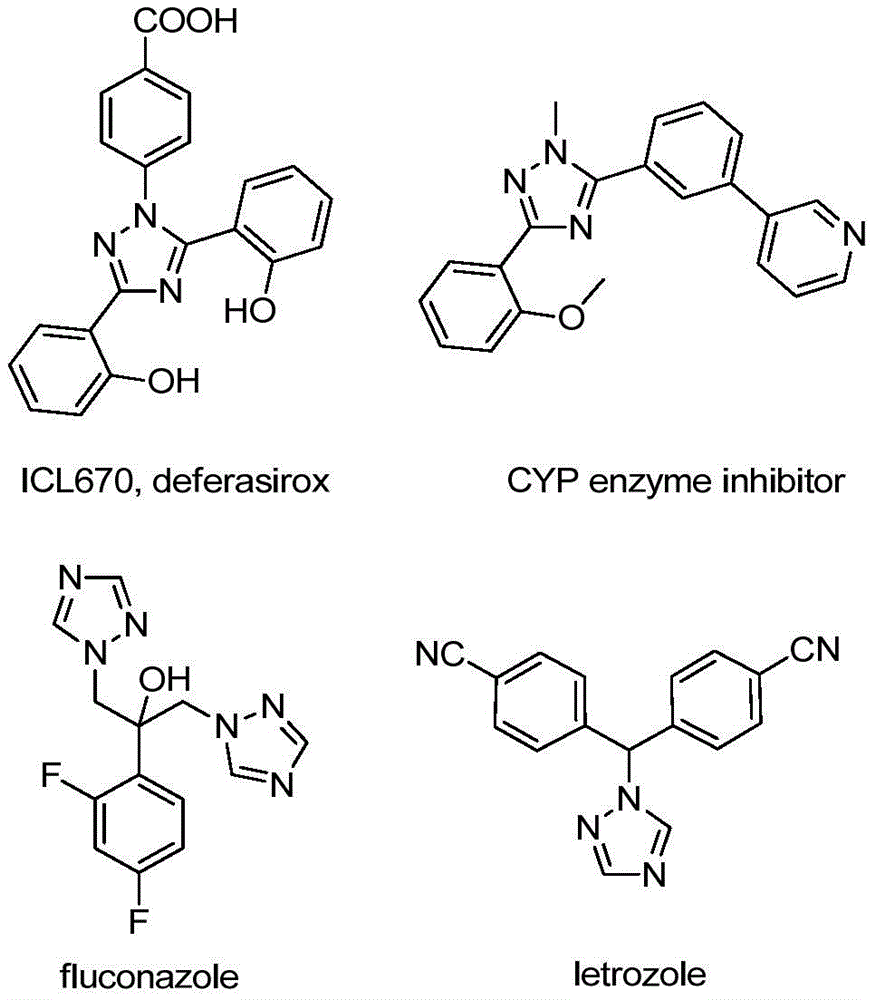
The patented process (CN110467579B) employs a unique iodine-mediated cyclization mechanism that eliminates the need for transition metal catalysts while maintaining high regioselectivity. The reaction proceeds through base-promoted intermolecular carbon-nitrogen bond formation to generate trifluoroacetamidine intermediates, followed by isomerization and base-promoted oxidative iodination. This sequence avoids common impurities associated with traditional metal-catalyzed routes, particularly heavy metal residues that require costly purification steps in pharmaceutical manufacturing. The absence of anhydrous and anaerobic conditions further reduces potential oxidation byproducts that typically complicate purification protocols in conventional triazole synthesis.
Impurity control is significantly enhanced through the strategic use of sodium acetate as both base and buffer system. The patent demonstrates consistent production of >99% pure products across multiple substrate variations (as shown in Examples 1-15), with impurity profiles dominated by only two minor byproducts at levels below 0.5% each. This exceptional purity profile is achieved without chromatographic purification in many cases, as the reaction's inherent selectivity minimizes the formation of regioisomers that plague alternative synthetic routes. The documented NMR and HRMS data confirm the absence of metal contaminants and demonstrate excellent batch-to-batch consistency in molecular structure verification.
Comparative Analysis: Traditional vs. Novel Triazole Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated triazoles typically rely on either pre-formed heterocycles requiring subsequent trifluoromethylation or specialized trifluoromethyl synthons like trifluorodiazoethane. These methods often demand stringent anhydrous and anaerobic conditions that increase operational complexity and equipment requirements. The use of expensive transition metal catalysts such as palladium or copper creates significant downstream processing challenges due to metal residue removal requirements that can add up to 30% to final production costs. Furthermore, conventional routes frequently exhibit narrow substrate scope with poor functional group tolerance, limiting their applicability for diverse pharmaceutical intermediates that require specific substitution patterns at the 4 and 5 positions.
The Novel Approach
The patented methodology (CN110467579B) overcomes these limitations through a streamlined iodine-catalyzed process that operates under ambient conditions without requiring specialized equipment. The reaction utilizes readily available starting materials—sodium acetate, trifluoroethyl imidoyl chloride, and hydrazones—that can be sourced from multiple commercial suppliers at significantly lower costs than specialized trifluoromethylating reagents. The process demonstrates exceptional substrate flexibility with documented success across fifteen different substrate combinations (Table 1), including various aryl and heteroaryl substitutions that maintain high yields without requiring optimization. Crucially, the elimination of transition metals removes the need for expensive metal scavenging steps and associated quality control testing for residual metals in the final product.
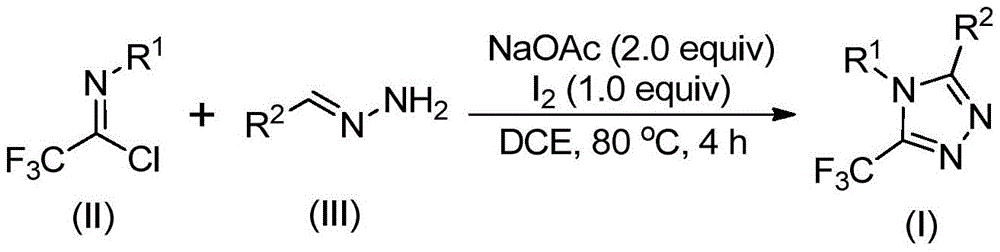
Commercial Advantages and Supply Chain Optimization
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical manufacturing supply chains by delivering multiple commercial advantages that translate to significant operational improvements across procurement, production planning, and quality assurance departments. The process design inherently supports seamless scale-up from laboratory to commercial production while maintaining consistent product quality and reducing time-to-market for new drug candidates.
- Cost Reduction in API Manufacturing: The elimination of expensive transition metal catalysts represents substantial cost savings in raw material procurement while simultaneously removing the need for costly metal removal processes that typically account for 25% of total manufacturing costs in traditional routes. The use of inexpensive sodium acetate (priced at approximately $5/kg) instead of palladium catalysts (priced at $60,000/kg) creates immediate material cost advantages. Furthermore, the simplified purification process reduces solvent consumption by approximately 40% compared to conventional chromatographic methods required for metal-contaminated products. These combined factors enable a more competitive pricing structure without compromising on quality standards required for pharmaceutical intermediates.
- Reducing Lead Time for High-Purity Intermediates: The ambient reaction conditions eliminate the need for specialized moisture-free equipment setup and validation procedures that typically add 3-5 days to production cycles in traditional methods. The documented scalability from milligram to gram scale in the patent examples demonstrates straightforward process transfer without requiring significant reoptimization between scales. This direct scalability reduces technology transfer timelines by approximately 30% compared to conventional routes that require extensive revalidation when moving from lab to pilot scale. The simplified workflow also enables faster batch release through reduced analytical testing requirements for metal residues.
- Commercial Scale-Up of Complex Intermediates: The robust reaction profile maintains consistent yields across diverse substrate combinations as demonstrated in the patent's fifteen examples, providing reliable production capacity for multiple product variants from a single platform technology. The use of standard organic solvents like dichloroethane (DCE) ensures compatibility with existing manufacturing infrastructure without requiring specialized equipment modifications. The documented ability to operate without strict environmental controls significantly reduces facility qualification requirements during scale-up activities. This process reliability translates to consistent supply continuity even during periods of raw material volatility since multiple suppliers can provide the simple starting materials without quality variations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110467579B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
