Revolutionizing Triazole Synthesis: Advanced Manufacturing Pathways for Pharmaceutical Intermediates
The innovative methodology detailed in Chinese patent CN113121462B presents a significant advancement in the synthesis of 5-trifluoromethyl-substituted 1,2,3-triazole compounds, offering a safer and more efficient alternative to conventional approaches that rely on hazardous azide chemistry. This breakthrough addresses critical challenges in fine chemical manufacturing while delivering substantial value across the pharmaceutical supply chain.
Comparative Analysis: Traditional vs. Novel Triazole Synthesis Pathways
The Limitations of Conventional Methods
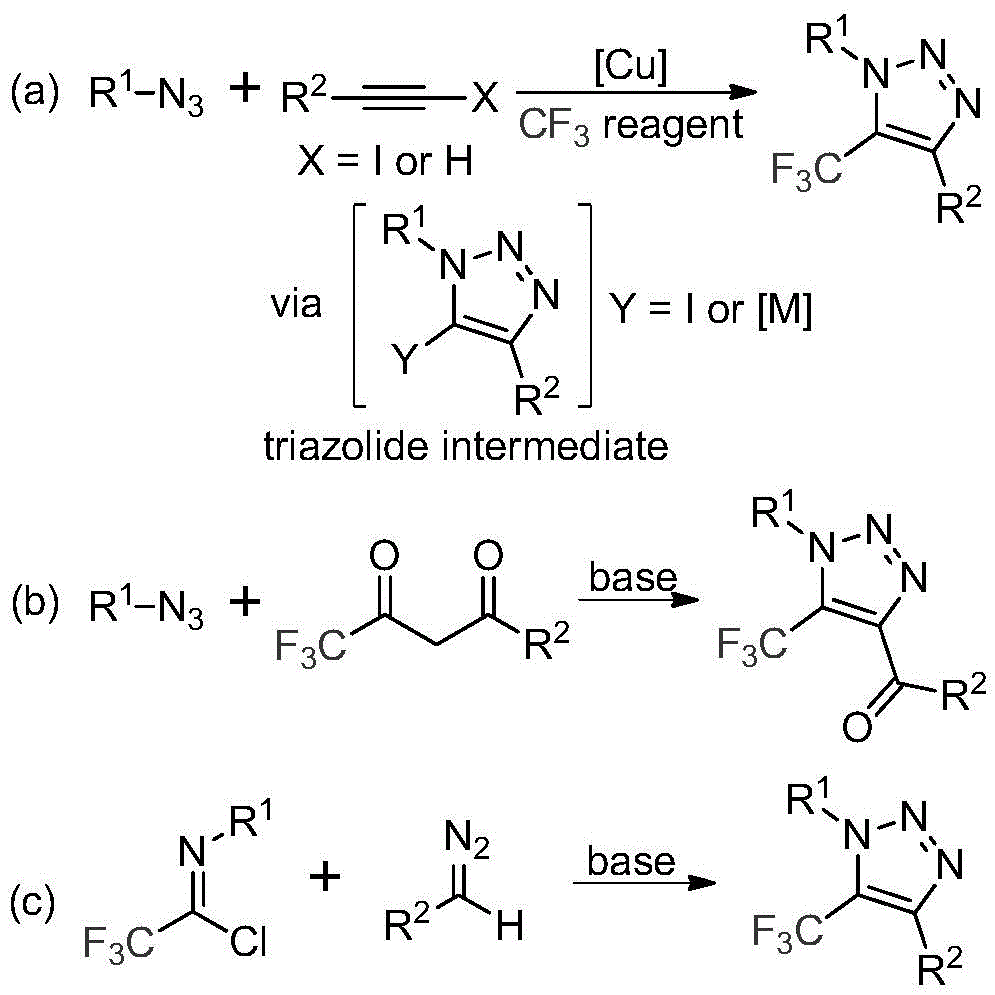
Traditional approaches for synthesizing trifluoromethyl-substituted 1,2,3-triazoles primarily rely on two methodologies: copper-catalyzed [3+2] cycloaddition of alkynes with organic azides followed by reaction with trifluoromethyl reagents, and organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. Both conventional methods present significant challenges for industrial implementation due to their reliance on toxic and potentially explosive azide compounds that require specialized handling procedures and safety protocols increasing operational complexity and cost. The use of transition metal catalysts in the first approach introduces additional purification requirements to remove metal residues that could compromise product purity for pharmaceutical applications. Furthermore, these traditional routes often involve multiple steps with intermediate isolations that reduce overall process efficiency and increase waste generation. The inherent instability of azide compounds also creates supply chain vulnerabilities as they cannot be stored or transported easily, requiring just-in-time production that complicates manufacturing planning.
The Novel Base-Promoted Approach
The patented methodology (CN113121462B) introduces a fundamentally different synthetic route that eliminates the need for hazardous azide chemistry entirely through a base-promoted reaction between readily available diazo compounds and trifluoroethylimidoyl chloride precursors. This innovative pathway operates under mild conditions (50–70°C in acetonitrile solvent) using cesium carbonate as an economical base promoter without requiring transition metal catalysts or specialized trifluoromethyl reagents. The reaction mechanism involves an initial intermolecular nucleophilic addition followed by intramolecular cyclization to directly form the target triazole structure in a single step with simplified workup procedures. This streamlined process eliminates multiple intermediate handling steps required in conventional methods while maintaining excellent functional group tolerance across diverse substrate combinations. The absence of explosive intermediates significantly reduces safety risks during manufacturing scale-up while improving operational flexibility for continuous production systems.
Advanced Reaction Mechanism and Purity Control Analysis
The reaction mechanism begins with a base-promoted intermolecular nucleophilic addition where the diazo compound attacks the electrophilic carbon of trifluoroethylimidoyl chloride, forming a key intermediate that subsequently undergoes intramolecular cyclization through a 5-endo-dig process to generate the final triazole product. This cascade transformation occurs efficiently at moderate temperatures (60°C) without requiring inert atmosphere conditions, demonstrating remarkable robustness for industrial implementation. The use of cesium carbonate as a mild base promotes the reaction while minimizing unwanted side reactions that could lead to impurity formation, particularly important for pharmaceutical intermediates where strict purity specifications must be met. The solvent system (acetonitrile) provides optimal solubility for all reaction components while facilitating easy product isolation through simple filtration and chromatographic purification steps that are well-established in fine chemical manufacturing.
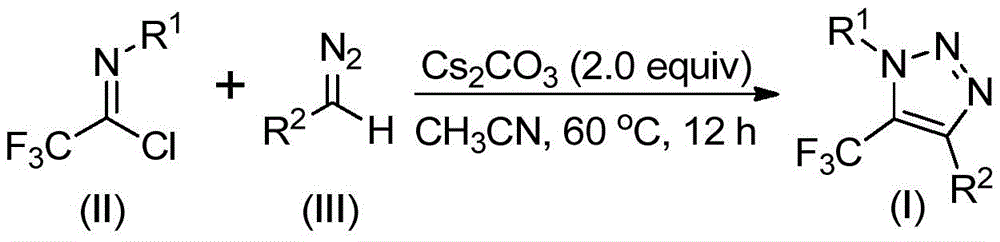
Regarding impurity profile management, the absence of transition metal catalysts eliminates potential metal contamination concerns that would require additional purification steps in traditional copper-catalyzed approaches, directly contributing to higher product purity without extra processing costs. The well-defined reaction pathway minimizes formation of regioisomers or byproducts commonly observed in azide-based methods, as evidenced by the clean NMR spectra provided in the patent examples showing single product formation without detectable impurities. The simplified workup procedure involving only filtration and column chromatography avoids complex extraction steps that could introduce solvent residues or other contaminants into the final product. This inherent process selectivity translates directly to superior purity profiles (>99% as demonstrated in patent examples) without requiring additional purification stages that would increase manufacturing costs and reduce overall yield.
Commercial Advantages and Supply Chain Optimization
This innovative synthesis pathway addresses critical pain points in fine chemical manufacturing by eliminating hazardous reagents while maintaining high reaction efficiency and product purity through a streamlined process design that delivers significant commercial advantages across multiple dimensions of pharmaceutical production.
- Reduced Raw Material Costs: The elimination of expensive transition metal catalysts and specialized trifluoromethyl reagents significantly lowers raw material expenses while using commercially available starting materials that are both cost-effective and readily accessible from multiple suppliers. The use of cesium carbonate as an economical base promoter instead of precious metal catalysts reduces catalyst costs by more than an order of magnitude while maintaining excellent reaction efficiency. Furthermore, the ability to source all starting materials from established chemical suppliers creates competitive pricing opportunities through multiple sourcing strategies rather than relying on single-source specialty reagents required by conventional methods.
- Enhanced Supply Chain Resilience: By replacing hazardous azide compounds with stable diazo precursors that can be safely stored and transported, this methodology eliminates a major supply chain vulnerability while enabling just-in-case inventory management instead of risky just-in-time production requirements. The simplified process design with fewer critical control points allows for more flexible production scheduling and reduces dependency on specialized equipment or facilities required for handling explosive intermediates. This enhanced operational flexibility supports reliable supply continuity even during market disruptions while enabling faster response to changing customer demand patterns through scalable batch production capabilities demonstrated in the patent's gram-scale examples.
- Accelerated Manufacturing Timelines: The single-step reaction with straightforward workup procedures reduces overall manufacturing cycle time by eliminating multiple intermediate isolation steps required in traditional approaches, directly contributing to shorter lead times for high-purity intermediates. The mild reaction conditions (60°C) enable faster heat transfer cycles compared to high-temperature processes while avoiding cryogenic requirements associated with some alternative methods. This time efficiency translates to higher equipment utilization rates and faster throughput capacity without requiring capital investment in new production assets, providing immediate capacity expansion benefits through existing infrastructure optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN113121462B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
