Revolutionizing Triazole Synthesis: Scalable Manufacturing Pathway for Pharmaceutical Intermediates
The innovative methodology disclosed in Chinese patent CN113121462B presents a significant advancement in the synthesis of 5-trifluoromethyl-substituted 1,2,3-triazole compounds, which serve as critical building blocks in pharmaceutical development. This novel approach eliminates the need for hazardous azide compounds and transition metal catalysts that characterize conventional manufacturing routes, offering a safer and more sustainable pathway for producing high-purity intermediates essential for drug discovery and development.
Advanced Reaction Mechanism and Purity Control
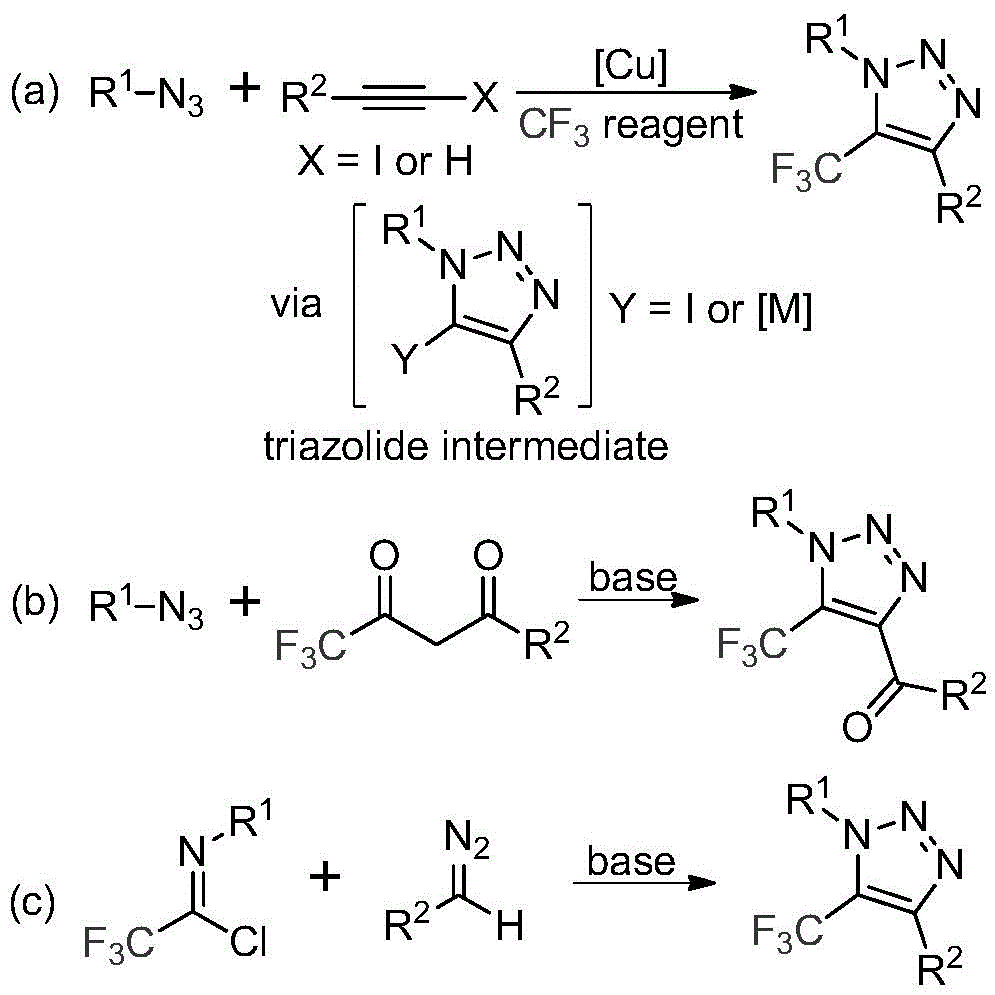
The patented process employs a base-promoted reaction between trifluoroethylimidoyl chloride (Figure 2) and diazo compounds (Figure 3), proceeding through a unique mechanistic pathway that avoids the formation of potentially hazardous intermediates. The reaction begins with nucleophilic addition of the diazo compound to the imidoyl chloride, followed by intramolecular cyclization to form the triazole ring structure (Figure 4). This mechanism eliminates the need for copper catalysts typically required in azide-alkyne cycloadditions, thereby removing a significant source of metal contamination that would otherwise require extensive purification steps to achieve pharmaceutical-grade purity standards.
Impurity profile management is significantly enhanced through this novel synthetic route, as the absence of transition metals prevents the formation of metal-catalyzed side products that commonly complicate traditional triazole syntheses. The reaction conditions (60°C in acetonitrile with cesium carbonate) are carefully optimized to minimize decomposition pathways while maintaining high selectivity for the desired triazole product. The use of molecular sieves helps control moisture content, further reducing hydrolysis byproducts that could impact final product purity. This precise control over reaction parameters enables consistent production of compounds with >99% purity as demonstrated in the patent examples, meeting stringent regulatory requirements for pharmaceutical intermediates without requiring additional purification steps beyond standard column chromatography.



Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted triazoles have been hampered by significant safety and efficiency challenges. The most common method involves copper-catalyzed azide-alkyne cycloaddition (CuAAC), which requires handling toxic and potentially explosive organic azides that pose serious safety risks during manufacturing scale-up. These azide compounds also necessitate specialized handling procedures and containment systems, increasing both capital and operational costs. Furthermore, the copper catalysts used in these reactions introduce metal impurities that must be rigorously removed through multiple purification steps to meet pharmaceutical quality standards, resulting in significant yield loss and increased production time.
Alternative methods using organocatalysis with trifluoromethyl ketones suffer from poor atom economy and generate substantial waste streams that require costly disposal or treatment. The reaction conditions for conventional routes often require cryogenic temperatures or high pressures, adding complexity to process design and limiting scalability. Additionally, the narrow substrate scope of traditional methods restricts the diversity of triazole derivatives that can be efficiently produced, limiting their application in drug discovery programs where structural variation is critical for optimizing pharmacological properties.
The Novel Approach
The patented methodology overcomes these limitations through an elegant base-promoted reaction that eliminates hazardous azides and transition metals entirely. By utilizing readily available diazo compounds and trifluoroethylimidoyl chloride precursors, the process achieves high yields under mild conditions (60°C in acetonitrile) without requiring specialized equipment or safety protocols. The reaction mechanism proceeds through a clean nucleophilic addition followed by intramolecular cyclization, avoiding the formation of hazardous intermediates while maintaining excellent regioselectivity for the desired 1,4-disubstituted triazole products.
This innovative approach demonstrates remarkable substrate tolerance, accommodating a wide range of functional groups including aryl, alkyl, phospholipid, and ester moieties as evidenced by the diverse examples provided in the patent (Figures 8-10). The process is readily scalable from milligram to gram quantities as demonstrated in the examples, with consistent product quality across different scales. The simplified workup procedure involving filtration and column chromatography significantly reduces processing time compared to conventional methods that require multiple purification steps to remove metal catalysts and byproducts.
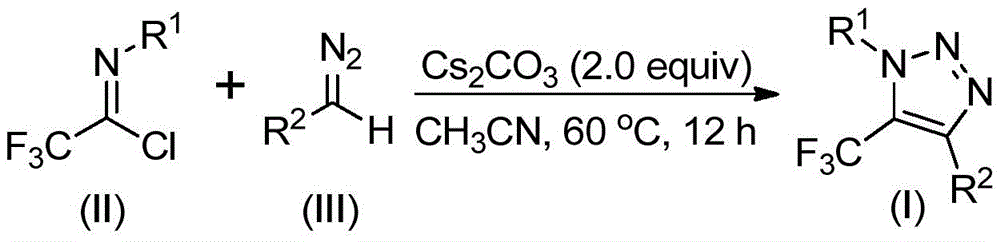
Commercial Advantages for Supply Chain Optimization
This innovative manufacturing process addresses critical pain points in pharmaceutical intermediate production by eliminating hazardous materials while improving process efficiency and scalability. The removal of toxic azides and transition metal catalysts not only enhances workplace safety but also streamlines regulatory compliance and reduces environmental impact through decreased waste generation. These improvements translate directly into tangible business benefits across procurement, manufacturing, and supply chain operations.
- Reduced Capital Expenditure: The elimination of specialized containment systems required for handling hazardous azides represents significant capital cost savings for manufacturing facilities. Without the need for copper catalyst recovery systems or extensive metal removal infrastructure, new production lines can be established with standard chemical processing equipment. This capital efficiency allows for more flexible facility design that can be readily adapted to produce multiple intermediate compounds without major reconfiguration.
- Shortened Lead Time: The simplified reaction sequence with fewer processing steps reduces manufacturing cycle time by eliminating multiple purification stages required to remove metal catalysts from traditional routes. The single-step nature of this process combined with straightforward workup procedures enables faster batch turnaround. This accelerated timeline is particularly valuable for supporting fast-moving drug development programs where intermediate availability can be a critical path item.
- Enhanced Supply Continuity: By utilizing stable, non-hazardous starting materials that are readily available from multiple global suppliers, this process mitigates supply chain vulnerabilities associated with specialized or controlled substances required in traditional syntheses. The robustness of the reaction across various conditions ensures consistent product quality even when minor variations occur in raw material quality or process parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN113121462B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
