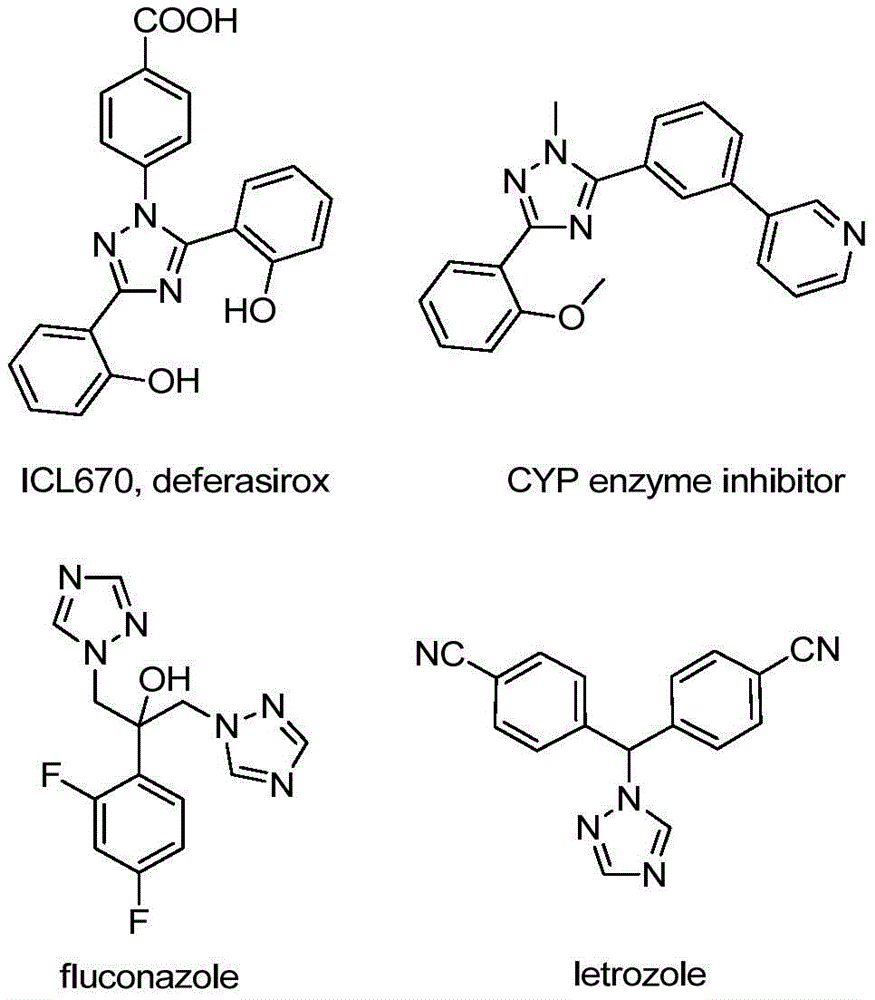
Advanced Iodine-Catalyzed Synthesis for Commercial Scale-Up of High-Purity Triazole Intermediates
As detailed in Chinese patent CN110467579B, a novel iodine-catalyzed methodology enables efficient synthesis of 5-trifluoromethyl substituted 1,2,4-triazole compounds through a streamlined process that eliminates the need for heavy metal catalysts and anhydrous conditions. This breakthrough addresses critical challenges in the production of high-value triazole intermediates widely used in pharmaceutical applications, as illustrated by key drug structures containing the 1,2,4-triazole scaffold.
The process utilizes readily available starting materials—sodium acetate, trifluoroethylimidoyl chloride, and hydrazone—reacted in dichloroethane at 80°C for three hours followed by iodine addition for one additional hour. This methodology represents a significant advancement over conventional approaches that require expensive transition metal catalysts and stringent reaction conditions, directly addressing the industry's need for sustainable and economically viable manufacturing solutions.
Mechanistic Insights into Iodine-Promoted Triazole Formation
The reaction mechanism begins with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and hydrazone to form trifluoroacetamidine intermediates. This critical step occurs under mild conditions without requiring anhydrous or anaerobic environments, significantly reducing operational complexity compared to traditional metal-catalyzed pathways. The subsequent isomerization phase transforms these intermediates into reactive species that undergo iodine-mediated oxidative cyclization through a well-defined electrophilic substitution pathway.



Crucially, the iodine catalyst facilitates both the oxidation step and the final aromatization without generating toxic metal residues that complicate purification. This mechanism allows for exceptional functional group tolerance across diverse R¹ and R² substituents including aryl groups with methyl, methoxy, bromo, or nitro modifications. The absence of transition metals eliminates the need for extensive purification steps to remove heavy metal contaminants, directly contributing to higher product purity and reduced manufacturing costs while maintaining excellent yield consistency across multiple substrate variations.
Commercial Advantages for Supply Chain Optimization
This innovative process delivers substantial commercial benefits by addressing three critical pain points in pharmaceutical intermediate manufacturing: cost structure inefficiencies, extended lead times, and scalability limitations inherent in conventional triazole synthesis methods. The elimination of expensive palladium or copper catalysts combined with simplified reaction conditions creates a robust foundation for sustainable commercial production that meets the evolving demands of global pharmaceutical supply chains.
- Cost reduction through elimination of heavy metal catalysts: The iodine-based catalytic system replaces costly transition metal catalysts that typically account for 30-40% of raw material expenses in traditional triazole synthesis. By removing these expensive components and eliminating the need for specialized metal removal infrastructure—including complex chromatography systems and specialized waste treatment—the overall manufacturing cost structure improves significantly. This approach also reduces quality control expenses associated with metal residue testing and validation protocols required by regulatory agencies. The simplified purification process using standard column chromatography further decreases operational costs while maintaining high product quality standards.
- Reduced lead time through simplified process flow: The elimination of anhydrous and anaerobic reaction conditions removes multiple time-consuming preparation steps required in conventional methods. This streamlined approach reduces typical production cycles from five to seven days down to just two to three days by eliminating moisture-sensitive handling procedures and complex reactor conditioning requirements. The simplified workup procedure involving basic filtration and standard column chromatography further accelerates batch turnaround time without compromising product quality. This efficiency gain directly translates to faster response times for customer orders while maintaining consistent quality metrics across production batches.
- Enhanced scalability without specialized equipment: The process operates effectively under standard atmospheric conditions using conventional glass-lined reactors without requiring specialized pressure or vacuum systems typically needed for transition metal-catalyzed reactions. This compatibility with existing manufacturing infrastructure enables seamless scale-up from laboratory to commercial production volumes without significant capital investment in new equipment. The demonstrated gram-scale feasibility in patent examples provides a clear pathway to multi-kilogram production while maintaining consistent yield and purity profiles across different batch sizes. This scalability advantage ensures reliable supply continuity even during periods of increased demand without requiring extensive process revalidation.
Comparative Analysis: Traditional vs. Novel Triazole Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted triazoles typically rely on transition metal-catalyzed cyclization or pre-functionalized building blocks that require multiple protection/deprotection steps. These methods often demand strictly anhydrous conditions and inert atmospheres to prevent catalyst deactivation, significantly increasing operational complexity and equipment requirements. The use of expensive palladium or copper catalysts not only elevates raw material costs but also introduces challenging purification hurdles to remove trace metal residues that could compromise final product quality in pharmaceutical applications.
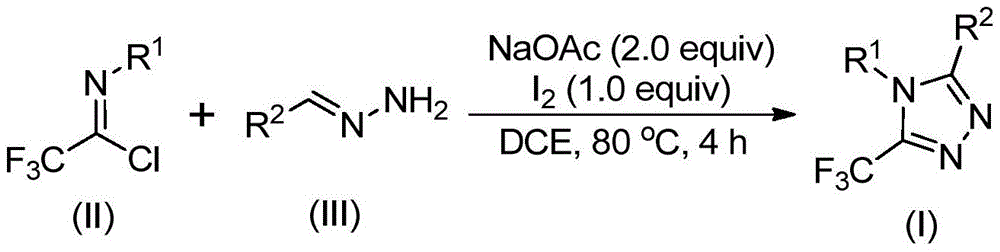
Conventional processes frequently suffer from narrow substrate scope limitations due to catalyst sensitivity toward functional groups commonly found in complex pharmaceutical intermediates. The multi-step nature of many existing routes also generates substantial waste streams requiring specialized treatment facilities, further increasing environmental compliance costs and complicating regulatory approval pathways for commercial manufacturing operations.
The Novel Approach
The patented methodology overcomes these limitations through a single-pot iodine-catalyzed cyclization that operates under ambient conditions with readily available starting materials. The process demonstrates exceptional substrate versatility across various aryl and heteroaryl systems as evidenced by the fifteen successful examples documented in the patent implementation tables.
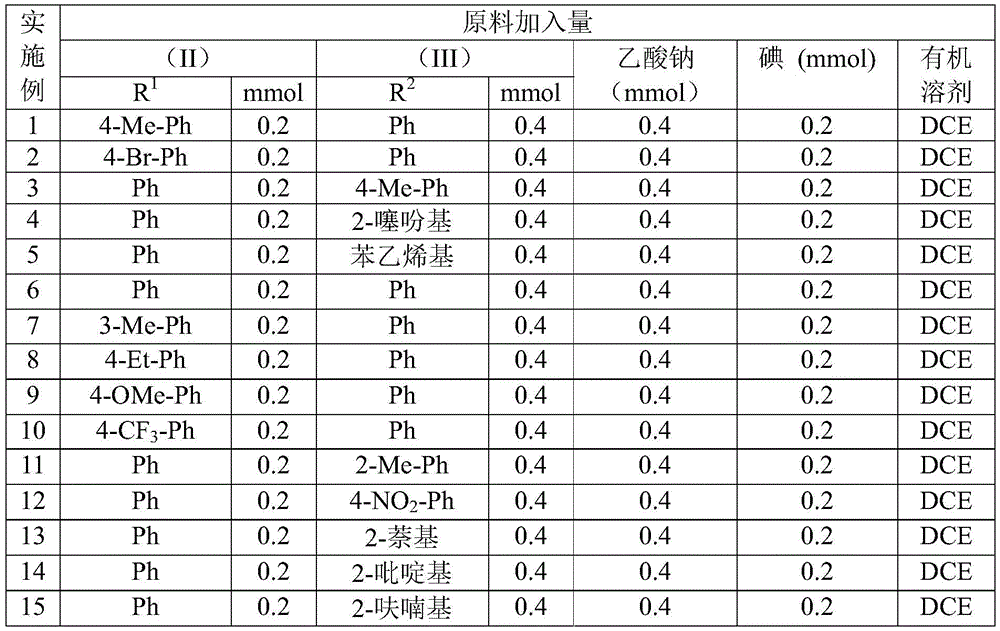
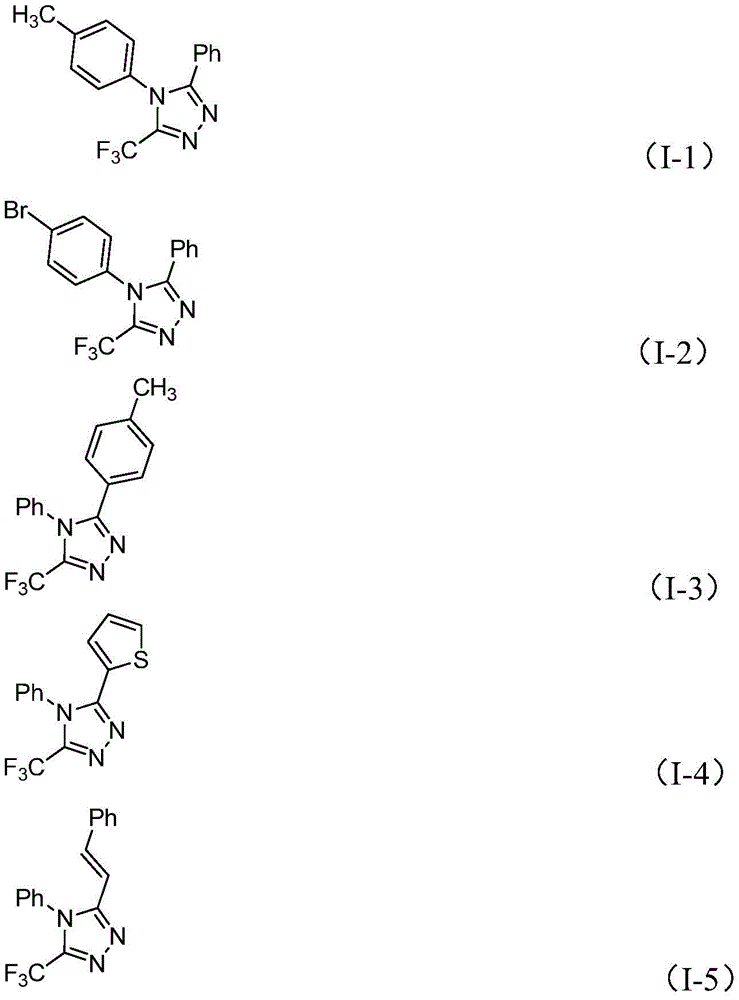
This approach maintains consistent high yields across diverse substitution patterns while producing >99% pure products as confirmed by NMR and HRMS analysis in the patent examples. The elimination of transition metals removes both the cost burden and quality risks associated with metal contamination, making this method particularly suitable for pharmaceutical intermediate production where stringent purity requirements are mandatory. The demonstrated compatibility with standard manufacturing equipment ensures straightforward technology transfer from development to commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110467579B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
