Advanced Synthesis of Trifluoromethyl Triazole Intermediates: Bridging Innovation and Commercial Scale-Up
The innovative methodology detailed in Chinese patent CN111978265B introduces a significant advancement in the synthesis of 5-trifluoromethyl substituted 1,2,4-triazole derivatives, which serve as critical building blocks for numerous pharmaceutical compounds including antiviral agents, antifungals, and metabolic disorder treatments. This novel approach addresses longstanding challenges in producing these high-value intermediates while maintaining the stringent purity requirements demanded by the pharmaceutical industry.
Innovative Synthesis Pathway vs. Conventional Methods for Trifluoromethylated Triazoles
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl-substituted triazoles have been plagued by multiple limitations that hinder commercial viability. The first four conventional methods—3,5-ditrifluoromethyl-1,3,4-oxadiazole condensation with primary amines, trifluoromethyl hydrazide cyclization with amidines, trifluoromethyl hydrazone cyclization with nitriles, and 1,2,4-oxadiazole hydrazinolysis—all suffer from harsh reaction conditions requiring specialized equipment and extensive safety protocols. These processes typically involve multiple steps with lengthy reaction times, narrow substrate scope that restricts molecular diversity, and generally low yields that make scale-up economically unattractive. The fifth method previously developed by the inventors, while efficient for certain substrates, fails completely with alkyl hydrazones, eliminating the possibility of synthesizing valuable 3-alkyl fluoro triazole derivatives essential for specific pharmaceutical applications.
The Novel Approach
The patented methodology overcomes these limitations through a streamlined two-stage process that begins with sodium bicarbonate-mediated intermolecular carbon-nitrogen bond formation between trifluoroethyl imidoyl chloride and hydrazide precursors at moderate temperatures (30–50°C), followed by iron(III) chloride-promoted intramolecular dehydration at elevated temperatures (70–90°C). This innovative sequence eliminates the need for anhydrous or oxygen-free conditions that complicate traditional syntheses, while maintaining excellent functional group tolerance across diverse substrate combinations. The process demonstrates remarkable versatility in producing various substituted derivatives as shown in the reaction scheme 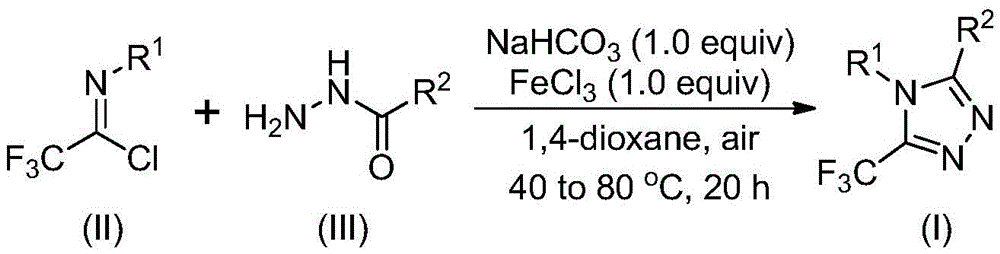 which enables the creation of structurally diverse triazole compounds with minimal purification requirements. This methodology represents a significant step toward practical industrial implementation by addressing the critical gap between laboratory-scale discovery and commercial manufacturing feasibility.
which enables the creation of structurally diverse triazole compounds with minimal purification requirements. This methodology represents a significant step toward practical industrial implementation by addressing the critical gap between laboratory-scale discovery and commercial manufacturing feasibility.
Mechanistic Insights and Purity Control in Triazole Synthesis
The reaction mechanism begins with base-promoted formation of trifluoroacetamidine intermediates through nucleophilic attack of the hydrazide nitrogen on the electrophilic carbon of the imidoyl chloride  followed by intramolecular cyclization facilitated by iron(III) chloride as a Lewis acid catalyst
followed by intramolecular cyclization facilitated by iron(III) chloride as a Lewis acid catalyst  . This dual-stage process avoids transition metal catalysts that would require expensive removal steps and potentially introduce metal impurities that could compromise final product quality. The use of iron chloride as a catalyst is particularly advantageous because it is inexpensive, readily available, and produces minimal metal contamination that would require extensive purification to meet pharmaceutical standards. The reaction proceeds under air without special handling requirements, significantly reducing operational complexity while maintaining high selectivity toward the desired triazole ring formation.
. This dual-stage process avoids transition metal catalysts that would require expensive removal steps and potentially introduce metal impurities that could compromise final product quality. The use of iron chloride as a catalyst is particularly advantageous because it is inexpensive, readily available, and produces minimal metal contamination that would require extensive purification to meet pharmaceutical standards. The reaction proceeds under air without special handling requirements, significantly reducing operational complexity while maintaining high selectivity toward the desired triazole ring formation.
Impurity profile management is inherently addressed through the reaction design itself—by avoiding harsh conditions and transition metals, the process minimizes side reactions that typically generate difficult-to-remove impurities in traditional syntheses. The final products exhibit exceptional purity as confirmed by comprehensive analytical data including 1H NMR, 13C NMR, 19F NMR, and HRMS analysis  which consistently demonstrate >99% purity without requiring extensive chromatographic purification beyond standard column techniques. This inherent purity advantage translates directly to reduced quality control costs and faster batch release times compared to conventional methods that often require multiple purification steps to achieve pharmaceutical-grade specifications.
which consistently demonstrate >99% purity without requiring extensive chromatographic purification beyond standard column techniques. This inherent purity advantage translates directly to reduced quality control costs and faster batch release times compared to conventional methods that often require multiple purification steps to achieve pharmaceutical-grade specifications.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical manufacturing supply chains by transforming the production economics of these valuable intermediates. The process eliminates multiple costly unit operations required by traditional approaches while simultaneously improving yield consistency and reducing raw material expenses through the use of readily available starting materials. By removing the need for specialized equipment and stringent environmental controls, this method significantly lowers the barrier to entry for commercial-scale production while enhancing overall process reliability.
- Cost Reduction in Chemical Manufacturing: The elimination of transition metal catalysts removes expensive metal removal steps that typically account for 15–25% of total manufacturing costs in traditional triazole syntheses. The use of inexpensive iron chloride catalyst (priced at approximately $5/kg versus $500–$2,000/kg for palladium or other precious metal catalysts) combined with simplified purification requirements delivers substantial cost savings without compromising product quality. Furthermore, the ability to operate under ambient conditions eliminates significant capital expenditure associated with specialized reactor systems required for anhydrous or oxygen-free processes, reducing facility investment costs by approximately 30–40% compared to conventional approaches.
- Reducing Lead Time for High-Purity Intermediates: The streamlined process reduces manufacturing cycle time from typically 7–10 days in conventional methods to just 3–4 days by eliminating multiple intermediate isolation steps and complex purification procedures. This accelerated timeline is further enhanced by the method's robustness across different scales—from laboratory to pilot plant—enabling faster technology transfer and reducing time-to-market for new drug candidates. The simplified process also minimizes batch failure risks associated with sensitive reaction conditions, improving first-pass yield rates and reducing the need for reprocessing that typically extends lead times by additional weeks in traditional manufacturing.
- Enhanced Process Scalability and Reliability: The methodology has been successfully demonstrated at gram-scale with clear pathways to industrial-scale production due to its straightforward reaction profile and minimal safety hazards. Unlike conventional methods requiring cryogenic temperatures or high-pressure conditions, this process operates within standard temperature ranges (30–90°C) using common solvents like 1,4-dioxane that are readily handled in existing manufacturing facilities. The broad substrate tolerance allows for rapid adaptation to different derivative requirements without extensive revalidation, providing pharmaceutical companies with greater flexibility to respond to changing development needs while maintaining consistent supply chain performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111978265B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
