Advanced Cross-Coupling Strategies for the Commercial Production of Abiraterone Acetate Intermediates
Advanced Cross-Coupling Strategies for the Commercial Production of Abiraterone Acetate Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical oncology intermediates, and the preparation of Abiraterone Acetate stands as a prime example of this technological evolution. Patent CN103864878A introduces a transformative methodology that shifts the paradigm from traditional boron-based Suzuki couplings to more versatile organozinc, organomagnesium, and organoindium cross-coupling reactions. This innovation addresses long-standing challenges in yield optimization and impurity control, providing a viable pathway for the commercial scale-up of complex steroid intermediates. By leveraging transition metal catalysis with earth-abundant metals, this approach not only enhances chemical efficiency but also aligns with modern green chemistry principles required by regulatory bodies. For R&D teams evaluating process robustness, the ability to toggle between Palladium, Nickel, Copper, and Iron catalysts offers unprecedented flexibility in managing production costs and supply chain risks. This report delves into the technical nuances of this patent, highlighting its potential to redefine the manufacturing landscape for this vital prostate cancer treatment precursor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Abiraterone Acetate has relied heavily on Suzuki-Miyaura coupling strategies utilizing diethyl(3-pyridyl)borane as the nucleophilic partner, a route exemplified in earlier patents such as GB2265624A. While chemically feasible, these legacy processes suffer from significant drawbacks that hinder large-scale adoption, particularly regarding the preparation of the boron reagent itself which requires hazardous Grignard reactions and subsequent transmetallation steps. Furthermore, the conventional route starting from dehydroepiandrosterone acetate often encounters severe selectivity issues where the acetoxy group is prone to elimination, generating a persistent androst-3,5,16-triene-17-trifluoromethanesulfonate by-product. 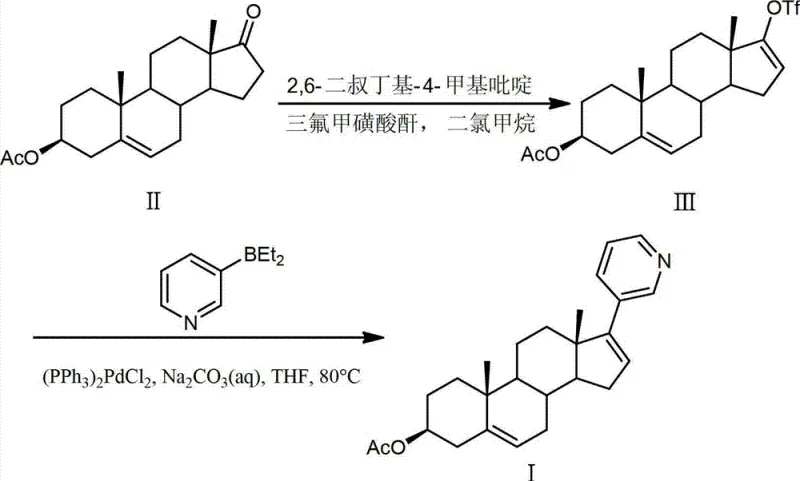 This specific impurity is notoriously difficult to remove via standard recrystallization techniques, forcing manufacturers to rely on costly and time-consuming column chromatography which is entirely unsuitable for multi-ton production. Additionally, alternative routes involving vinyl iodide intermediates, as seen in GB2282377A, extend reaction times to upwards of two days and introduce risks of Heck coupling side reactions, ultimately capping the total yield at approximately 41.5% to 48.7%. These inefficiencies create bottlenecks in cost reduction in pharmaceutical intermediates manufacturing and complicate the supply chain for downstream API producers.
This specific impurity is notoriously difficult to remove via standard recrystallization techniques, forcing manufacturers to rely on costly and time-consuming column chromatography which is entirely unsuitable for multi-ton production. Additionally, alternative routes involving vinyl iodide intermediates, as seen in GB2282377A, extend reaction times to upwards of two days and introduce risks of Heck coupling side reactions, ultimately capping the total yield at approximately 41.5% to 48.7%. These inefficiencies create bottlenecks in cost reduction in pharmaceutical intermediates manufacturing and complicate the supply chain for downstream API producers.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN103864878A utilizes a direct coupling strategy between steroidal triflates or vinyl iodides and pyridyl-metal reagents containing Zinc, Magnesium, or Indium. This novel approach fundamentally simplifies the reaction landscape by eliminating the need for pre-formed borane species, thereby reducing the number of unit operations and the associated safety hazards. The patent demonstrates that by employing catalysts such as bis(triphenylphosphine)palladium dichloride or even non-precious metal alternatives like nickel chloride, the reaction can proceed with remarkable speed and efficiency. For instance, the coupling of 3β-acetoxyandrosta-5,16-dien-17-trifluoromethanesulfonate with a pyridyl-zinc reagent achieved an impressive isolated yield of 85% in just 20 minutes at 100°C. 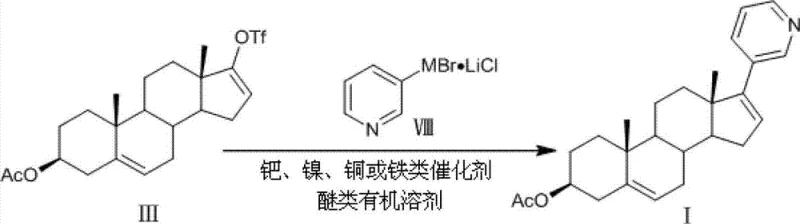 This dramatic improvement in throughput directly translates to enhanced capacity utilization for manufacturing facilities. Moreover, the versatility of the system allows for the use of various ether solvents like THF or MTBE, providing process engineers with the flexibility to optimize solvent recovery and recycling protocols. The ability to bypass the formation of the troublesome triene by-product significantly streamlines the purification workflow, making this method a superior choice for establishing a reliable API intermediate supplier network capable of meeting stringent quality standards.
This dramatic improvement in throughput directly translates to enhanced capacity utilization for manufacturing facilities. Moreover, the versatility of the system allows for the use of various ether solvents like THF or MTBE, providing process engineers with the flexibility to optimize solvent recovery and recycling protocols. The ability to bypass the formation of the troublesome triene by-product significantly streamlines the purification workflow, making this method a superior choice for establishing a reliable API intermediate supplier network capable of meeting stringent quality standards.
Mechanistic Insights into Transition Metal Catalyzed Cross-Coupling
The core of this technological advancement lies in the mechanistic efficiency of the transmetallation step when using organozinc or organomagnesium species compared to their organoboron counterparts. In the catalytic cycle, the oxidative addition of the steroidal electrophile (triflate or iodide) to the low-valent metal center is followed by a rapid transmetallation with the pyridyl-metal reagent, which is generally more nucleophilic and reactive than boronic acid derivatives. This heightened reactivity allows the reaction to proceed under milder conditions or shorter timeframes, reducing the thermal stress on the sensitive steroid backbone and minimizing degradation pathways. The presence of lithium chloride in the reagent preparation plays a crucial role in stabilizing the organometallic species and enhancing solubility in ether solvents, ensuring a homogeneous reaction environment that promotes consistent kinetics. Furthermore, the patent highlights the unique capability of Iron and Nickel catalysts to facilitate this transformation, suggesting a mechanism that may involve single-electron transfer pathways distinct from the traditional two-electron processes of Palladium. This mechanistic diversity is critical for R&D directors focused on high-purity Abiraterone Acetate, as it opens avenues to suppress specific metal-catalyzed side reactions that plague standard Suzuki conditions.
From an impurity control perspective, the selection of the leaving group and the metal catalyst combination dictates the profile of side products generated during the synthesis. The use of triflate leaving groups in conjunction with Zinc or Magnesium reagents appears to suppress the beta-elimination of the 3-acetoxy group, a major failure mode in previous iterations of this chemistry. By carefully tuning the basicity of the reaction medium—using inorganic bases like potassium carbonate or hydroxides only when necessary with Magnesium reagents—the process maintains the integrity of the steroid skeleton. The patent data indicates that even when using Iron catalysts, which are typically more prone to promoting radical side reactions, the selectivity remains high enough to produce crystalline product after simple workup. This level of control over the reaction trajectory ensures that the final impurity profile is manageable and predictable, a key requirement for regulatory filings. Understanding these mechanistic subtleties allows process chemists to design robust control strategies that guarantee batch-to-batch consistency, essential for reducing lead time for high-purity API intermediates in a commercial setting.
How to Synthesize Abiraterone Acetate Efficiently
The implementation of this synthesis route requires precise attention to the preparation of the organometallic reagent and the selection of the appropriate catalyst system based on cost and availability targets. The process begins with the activation of metal powders such as Zinc or Magnesium in the presence of lithium chloride and a small amount of 1,2-dibromoethane to initiate the formation of the pyridyl-metal species from 3-bromopyridine. Once the reagent is generated, it is introduced to the steroidal electrophile in a dry ether solvent under an inert atmosphere to prevent oxidation or hydrolysis. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures described in the patent examples, are provided in the guide below to ensure reproducibility and safety during scale-up.
- Preparation of the pyridyl-metal reagent (Zn, Mg, or In) from 3-bromopyridine and the corresponding metal powder in the presence of lithium chloride.
- Coupling reaction of 3β-acetoxyandrosta-5,16-dien-17-trifluoromethanesulfonate or 17-iodo-androsta-5,16-dien-3β-ol with the pyridyl-metal reagent using a transition metal catalyst.
- Workup and purification involving aqueous quenching, extraction, and recrystallization to obtain high-purity Abiraterone Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel coupling methodology presents a compelling value proposition centered on raw material accessibility and process simplification. The shift away from specialized borane reagents, which often require custom synthesis and have limited global suppliers, to commodity metals like Zinc and Magnesium drastically reduces supply chain vulnerability and price volatility. This substitution eliminates the complex multi-step preparation of diethyl(3-pyridyl)borane, effectively collapsing the supply chain and reducing the number of external vendors required to support production. Consequently, this leads to significant cost savings in the overall bill of materials, allowing manufacturers to offer more competitive pricing without compromising on quality. The ability to utilize base metal catalysts such as Iron and Nickel further amplifies these economic benefits by removing dependence on precious metals like Palladium, whose prices are subject to intense market fluctuations and geopolitical instability.
- Cost Reduction in Manufacturing: The elimination of expensive boron reagents and precious metal catalysts directly lowers the variable cost per kilogram of the intermediate. By replacing Palladium with Nickel or Iron, the catalyst cost component is reduced by orders of magnitude, while the simplified purification process reduces solvent consumption and waste disposal fees. This structural cost advantage creates a sustainable margin buffer that protects against raw material inflation and enhances long-term profitability for the manufacturing entity.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity chemicals like Zinc powder, Magnesium turnings, and common ether solvents ensures that production is not held hostage by the capacity constraints of niche reagent suppliers. This diversification of the raw material base significantly mitigates the risk of supply disruptions, ensuring continuous operation even during global logistics crises. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain against upstream variability.
- Scalability and Environmental Compliance: The high yields and simplified workup procedures described in the patent facilitate a smoother transition from pilot plant to full commercial scale, reducing the time and capital expenditure required for process validation. Additionally, the potential to use less toxic Iron catalysts and the reduction in heavy metal waste align with increasingly stringent environmental regulations, lowering the compliance burden and associated remediation costs. This eco-efficient profile enhances the corporate sustainability rating of the manufacturer, appealing to environmentally conscious partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cross-coupling technology for Abiraterone Acetate production. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using organozinc reagents over organoboron reagents in this synthesis?
A: Organozinc reagents eliminate the need for complex borane preparation steps involving Grignard reagents and borates, significantly simplifying the supply chain and reducing raw material costs while maintaining high coupling efficiency.
Q: Can base metal catalysts like Iron or Nickel replace Palladium in this process?
A: Yes, the patent demonstrates that Nickel, Copper, and Iron catalysts are effective alternatives to Palladium, offering substantial cost savings and reducing the burden of heavy metal residue removal in the final API.
Q: How does this method address the impurity issues found in prior art routes?
A: By optimizing the coupling conditions and avoiding the specific elimination pathways associated with previous triflate formations, this method minimizes the formation of difficult-to-remove triene by-products, facilitating easier purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN103864878A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors and rigorous QC labs capable of handling sensitive organometallic reactions while adhering to the most stringent purity specifications required for oncology intermediates. Our commitment to technical excellence ensures that we can translate these patent innovations into reliable, GMP-compliant manufacturing processes that deliver consistent quality.
We invite you to collaborate with us to leverage these cost-effective and scalable synthesis routes for your Abiraterone Acetate requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall cost of goods sold.
