Advanced Synthesis of Pyridyl Bridged Pyrazolyl Indole Derivatives for High-Efficiency Catalysis
Advanced Synthesis of Pyridyl Bridged Pyrazolyl Indole Derivatives for High-Efficiency Catalysis
The landscape of modern organometallic catalysis is constantly evolving, driven by the demand for more sustainable and efficient synthetic methodologies. Patent CN108148046B introduces a significant breakthrough in the preparation of pyridyl bridged pyrazolyl indole derivatives, a class of compounds that serve as critical precursors for high-performance catalysts. This technology addresses the longstanding challenges associated with synthesizing complex nitrogen-containing heterocycles, which are ubiquitous in pharmaceutical intermediates and advanced functional materials. By leveraging a straightforward copper-catalyzed substitution reaction, this innovation bypasses the need for harsh oxidants and multi-step sequences that have traditionally plagued the industry. For R&D directors and procurement specialists alike, this represents a pivotal shift towards greener chemistry that does not compromise on performance or scalability. The ability to generate these electron-rich derivatives from readily available starting materials like 2-bromo-6-pyrazolylpyridine opens new avenues for the development of robust catalytic systems capable of driving difficult transformations such as acceptorless dehydrogenation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocyclic compounds has been fraught with inefficiencies that hinder large-scale commercial adoption. Traditional pathways often rely heavily on the use of stoichiometric amounts of oxidants, which not only increases the raw material costs but also generates substantial quantities of hazardous waste that require expensive disposal protocols. Furthermore, conventional methods frequently involve tedious experimental steps, including multiple protection and deprotection cycles, which drastically reduce the overall atom economy and extend the production lead time. These factors create significant bottlenecks for supply chain managers who struggle to maintain consistent quality and volume when relying on outdated synthetic routes. The reliance on precious metal catalysts without efficient ligand systems often leads to poor turnover numbers, necessitating higher catalyst loadings that further inflate the cost of goods sold. Consequently, the industry has been in urgent need of a streamlined approach that can deliver high-purity intermediates without the environmental and economic baggage of legacy technologies.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by utilizing a direct substitution reaction between 2-bromo-6-pyrazolylpyridine and various indole compounds. This novel approach capitalizes on the reactivity of the bromo-substituted pyridine scaffold, allowing for the efficient construction of the pyridyl-bridged architecture under relatively mild conditions. 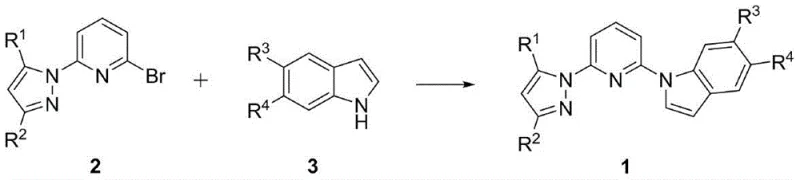 As illustrated in the reaction scheme, the process employs inexpensive copper catalysts such as CuBr or CuI, which are far more cost-effective than the palladium or iridium systems often seen in similar cross-coupling reactions. The reaction proceeds smoothly in common solvents like 1,4-dioxane or toluene, using simple inorganic bases like sodium carbonate to drive the transformation. This simplicity translates directly into operational excellence, as it reduces the complexity of the work-up procedure and minimizes the risk of side reactions that could compromise product purity. For manufacturers, this means a more reliable supply of high-quality intermediates that can be consistently produced at scale, thereby enhancing the stability of the entire downstream value chain.
As illustrated in the reaction scheme, the process employs inexpensive copper catalysts such as CuBr or CuI, which are far more cost-effective than the palladium or iridium systems often seen in similar cross-coupling reactions. The reaction proceeds smoothly in common solvents like 1,4-dioxane or toluene, using simple inorganic bases like sodium carbonate to drive the transformation. This simplicity translates directly into operational excellence, as it reduces the complexity of the work-up procedure and minimizes the risk of side reactions that could compromise product purity. For manufacturers, this means a more reliable supply of high-quality intermediates that can be consistently produced at scale, thereby enhancing the stability of the entire downstream value chain.
Mechanistic Insights into Copper-Catalyzed Substitution and Ligand Coordination
At the heart of this technological advancement lies a sophisticated understanding of copper-catalyzed C-N bond formation and subsequent coordination chemistry. The reaction mechanism likely involves the oxidative addition of the copper catalyst to the carbon-bromine bond of the pyridyl precursor, followed by coordination with the nitrogen atom of the indole ring. The presence of the pyrazole moiety adjacent to the pyridine ring creates a unique electronic environment that facilitates this coupling, stabilizing the transition state and ensuring high regioselectivity. This precision is crucial for R&D teams focused on impurity profiling, as it minimizes the formation of structural isomers that are notoriously difficult to separate. Once the pyridyl bridged pyrazolyl indole derivative is formed, its true potential is unlocked through its ability to act as a tridentate ligand. The three nitrogen donor atoms align perfectly to coordinate with transition metals like ruthenium, forming stable complexes that are resistant to decomposition under rigorous reaction conditions.
The resulting ruthenium complexes exhibit exceptional catalytic activity, particularly in the dehydrogenation of saturated nitrogen heterocycles, a reaction of paramount importance in the synthesis of quinoline derivatives found in many bioactive molecules. 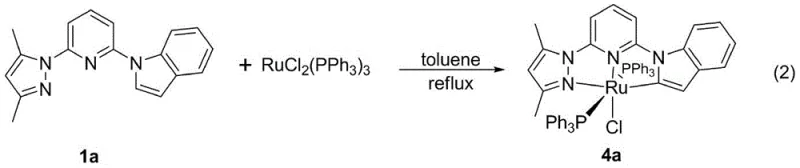 The formation of the ruthenium complex, as shown in the coordination diagram, demonstrates how the ligand wraps around the metal center, creating a rigid and well-defined active site. This structural integrity is what allows the catalyst to achieve high conversion rates, such as the 90% conversion of tetrahydroquinoline observed in the patent examples, without the need for external oxidants. The mechanism operates through a hydrogen borrowing strategy where the metal center temporarily accepts hydrogen from the substrate and releases it as molecular hydrogen gas. This atom-economical process aligns perfectly with green chemistry principles, producing water or hydrogen as the only byproducts. For technical leaders, understanding this mechanistic robustness provides confidence in the scalability of the process, as the catalyst's longevity reduces the frequency of replenishment and lowers the total cost of operation.
The formation of the ruthenium complex, as shown in the coordination diagram, demonstrates how the ligand wraps around the metal center, creating a rigid and well-defined active site. This structural integrity is what allows the catalyst to achieve high conversion rates, such as the 90% conversion of tetrahydroquinoline observed in the patent examples, without the need for external oxidants. The mechanism operates through a hydrogen borrowing strategy where the metal center temporarily accepts hydrogen from the substrate and releases it as molecular hydrogen gas. This atom-economical process aligns perfectly with green chemistry principles, producing water or hydrogen as the only byproducts. For technical leaders, understanding this mechanistic robustness provides confidence in the scalability of the process, as the catalyst's longevity reduces the frequency of replenishment and lowers the total cost of operation.
How to Synthesize Pyridyl Bridged Pyrazolyl Indole Derivatives Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and industrial-scale production. The protocol begins with the precise weighing of 2-bromo-6-pyrazolylpyridine and the chosen indole derivative, ensuring a molar ratio that favors the formation of the desired product while minimizing unreacted starting materials. The choice of solvent plays a critical role, with 1,4-dioxane demonstrating superior performance compared to alternatives like o-xylene, likely due to its ability to solubilize both the organic substrates and the inorganic base effectively. Maintaining an inert atmosphere, typically using nitrogen or argon, is essential to prevent the oxidation of the copper catalyst and ensure reproducible yields. The detailed standardized synthesis steps provided below outline the exact parameters for temperature, time, and purification that have been validated to produce high-purity material suitable for sensitive catalytic applications.
- Prepare the reaction mixture by combining 2-bromo-6-pyrazolylpyridine and indole compounds with a copper catalyst such as CuBr in a suitable solvent like 1,4-dioxane.
- Add an appropriate base such as sodium carbonate and heat the mixture to a temperature range of 70-150°C for 5-15 hours under inert atmosphere.
- Upon completion, cool the reaction, remove volatiles under reduced pressure, and purify the target derivative using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis technology offers profound advantages that resonate deeply with procurement managers and supply chain heads tasked with optimizing operational expenditures. The primary driver of value is the drastic simplification of the supply chain for raw materials; since the key starting materials like 2-bromo-6-pyrazolylpyridine and indoles are either commercially available or easily synthesized from commodity chemicals, the risk of supply disruption is significantly mitigated. This reliability ensures that production schedules can be maintained without the delays often caused by the scarcity of exotic reagents. Furthermore, the elimination of stoichiometric oxidants and the use of base metal catalysts instead of precious metals result in substantial cost savings in terms of raw material procurement and waste management. The process inherently reduces the environmental footprint, which simplifies regulatory compliance and avoids the hefty fines associated with hazardous waste disposal, thereby protecting the company's bottom line and reputation.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental shift away from expensive and hazardous reagents towards benign and abundant alternatives. By utilizing copper salts which are orders of magnitude cheaper than palladium or iridium, the direct material cost per kilogram of product is significantly lowered. Additionally, the high atom efficiency of the substitution reaction means that less raw material is wasted as byproduct, maximizing the yield from every batch. The simplified work-up procedure, which avoids complex extraction and purification steps required by oxidant-heavy methods, reduces labor costs and energy consumption during the manufacturing process. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediate, allowing downstream customers to achieve better margins on their own products.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available chemical building blocks. Unlike specialized reagents that may have single-source suppliers and long lead times, the precursors for this synthesis are produced by multiple global manufacturers, ensuring a steady flow of materials even during market fluctuations. The mild reaction conditions, operating at temperatures between 70-150°C, reduce the stress on reactor equipment and lower the energy requirements, making the process adaptable to a wider range of manufacturing facilities. This flexibility allows for decentralized production strategies if needed, further insulating the supply chain from regional disruptions. The consistency of the reaction outcome also means that inventory planning becomes more accurate, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of exothermic hazards associated with strong oxidants, making the safety profile much more manageable for large reactors. The generation of hydrogen gas as a byproduct in the downstream application is clean and can be managed with standard venting systems, avoiding the accumulation of toxic waste streams. This alignment with green chemistry principles not only satisfies increasingly stringent environmental regulations but also enhances the brand image of the manufacturer as a sustainability leader. The ease of purification via standard column chromatography or crystallization ensures that high purity specifications can be met consistently, which is critical for pharmaceutical grade intermediates. Ultimately, the process design supports a circular economy approach by minimizing waste and maximizing resource utilization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel derivatives. They are based on the specific experimental data and advantageous effects detailed in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their existing workflows. Understanding these nuances is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the primary advantages of this copper-catalyzed substitution method over traditional oxidation routes?
A: This method eliminates the need for stoichiometric oxidants and繁琐 experimental steps often required in traditional nitrogen heterocycle synthesis. It utilizes cheap and easily obtained raw materials like 2-bromo-6-pyrazolylpyridine and commercial indoles, offering high synthesis efficiency and mild reaction conditions.
Q: Can the resulting pyridyl bridged pyrazolyl indole derivatives be used for other applications besides dehydrogenation?
A: While the patent specifically highlights their utility as tridentate ligands for synthesizing highly efficient ruthenium catalysts for saturated nitrogen heterocycle dehydrogenation, the electron-rich nature and derivatization potential of the core structure suggest broad applicability in organometallic chemistry and potentially in material science contexts requiring robust chelating agents.
Q: What specific reaction conditions optimize the yield of the target derivative?
A: Optimal results are achieved using CuBr as the catalyst with sodium carbonate as the base in 1,4-dioxane solvent at 100°C for 10 hours. Deviations such as using CuOAc, o-xylene solvent, or lower temperatures significantly reduce the reaction yield as demonstrated in the comparative examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridyl Bridged Pyrazolyl Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the pyridyl bridged pyrazolyl indole derivative technology in advancing the field of homogeneous catalysis. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or catalyst precursor we deliver performs exactly as expected in your critical reactions. We understand that in the high-stakes world of pharmaceutical and fine chemical synthesis, consistency is key, and our commitment to quality assurance is unwavering.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to this method can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market through superior chemical innovation and supply chain reliability.
