Revolutionizing Aryl Internal Alkyne Production with Light-Mediated Copper Catalysis for Commercial Scale
Introduction to Next-Generation Alkyne Synthesis
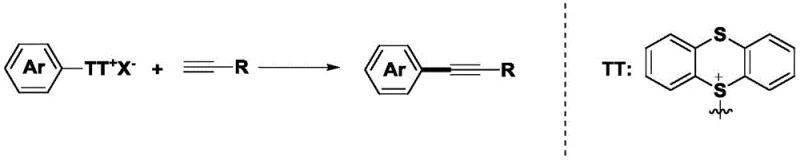
The synthesis of aryl internal alkynes represents a critical cornerstone in modern medicinal chemistry, serving as essential building blocks for a vast array of bioactive molecules, including kinase inhibitors and macrocyclic therapeutics. Traditionally, accessing these structural motifs has relied heavily on palladium-catalyzed cross-coupling methodologies, which, while effective, impose significant economic and environmental burdens due to the high cost of precious metals and the stringent requirement for removing trace metal residues from final drug substances. A groundbreaking advancement in this field is detailed in Chinese Patent CN111606785B, which discloses a novel, light-mediated copper catalytic system that fundamentally shifts the paradigm towards more sustainable and cost-effective manufacturing. This innovative approach utilizes readily available aryl sulfonium salts and terminal alkynes as coupling partners, driven by visible light irradiation and inexpensive copper(I) salts, thereby eliminating the dependency on palladium entirely. For global pharmaceutical manufacturers and fine chemical producers, this technology offers a compelling value proposition by merging high synthetic efficiency with a drastically reduced cost of goods sold (COGS), positioning it as a superior alternative for the large-scale production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the C(sp2)-C(sp) bond required for aryl internal alkynes has been dominated by the Sonogashira coupling reaction, which typically necessitates the use of aryl iodides or bromides in conjunction with palladium catalysts and copper co-catalysts. While robust, this classical approach suffers from several inherent drawbacks that complicate industrial adoption, particularly the exorbitant price volatility of palladium and the rigorous purification steps needed to meet strict regulatory limits on heavy metal impurities in active pharmaceutical ingredients (APIs). Furthermore, the preparation of the requisite aryl halide starting materials often involves additional synthetic steps utilizing hazardous halogenating agents, adding to the overall process mass intensity and waste generation. In many cases, the sensitivity of palladium catalysts to air and moisture requires strictly anhydrous conditions and specialized equipment, increasing the operational complexity and capital expenditure for manufacturing facilities. These cumulative factors create a significant bottleneck for procurement teams aiming to reduce supply chain costs and for R&D directors seeking greener, more streamlined synthetic routes for complex drug candidates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN111606785B introduces a transformative strategy that leverages aryl sulfonium salts as highly reactive electrophiles in a copper-catalyzed, photo-induced coupling reaction. This novel pathway bypasses the need for expensive palladium catalysts entirely, relying instead on abundant and low-cost copper(I) salts such as CuCl, CuBr, or CuI, which are activated by simple blue LED light sources at near-ambient temperatures. The use of aryl sulfonium salts is particularly advantageous as they can be easily derived from common aromatic precursors, offering a broader and more flexible substrate scope than traditional aryl halides. By operating under mild conditions (typically around 30°C) in polar aprotic solvents like DMSO, this method not only ensures excellent functional group tolerance but also significantly lowers the energy footprint of the reaction. This shift represents a major leap forward in process chemistry, enabling the efficient synthesis of diverse aryl internal alkynes with yields ranging from 50% to 86%, while simultaneously addressing key sustainability goals related to waste reduction and resource conservation.
Mechanistic Insights into Photo-mediated Copper Catalysis
The mechanistic underpinning of this transformation involves a sophisticated interplay between photo-excitation and copper redox chemistry, distinct from the oxidative addition/reductive elimination cycles typical of palladium catalysis. Upon irradiation with blue light, the copper(I) species interacts with the aryl sulfonium salt, likely facilitating a single-electron transfer (SET) process that generates a highly reactive aryl radical intermediate alongside a copper(II) species. This radical pathway allows for the activation of the C-S bond in the sulfonium salt under exceptionally mild conditions, avoiding the high thermal energy barriers associated with traditional thermal activation. The resulting aryl radical subsequently couples with the copper-acetylide species formed from the terminal alkyne and the base, eventually undergoing reductive elimination or radical rebound to forge the desired C-C triple bond linkage. This unique mechanism not only explains the high efficiency observed at room temperature but also accounts for the remarkable chemoselectivity, as the radical intermediates are generated and consumed rapidly within the coordination sphere of the copper catalyst, minimizing side reactions such as homocoupling of the alkyne.
From an impurity control perspective, this mechanism offers distinct advantages for the production of high-purity pharmaceutical intermediates. The absence of palladium eliminates the risk of difficult-to-remove Pd residues, which are a persistent challenge in API manufacturing and often require scavenging resins or complex crystallization protocols. Additionally, the mild reaction conditions prevent the thermal degradation of sensitive functional groups often present in advanced intermediates, such as esters, nitriles, and heterocycles, thereby reducing the formation of thermal decomposition byproducts. The use of potassium carbonate as a mild inorganic base further contributes to a cleaner reaction profile compared to strong organic bases that might induce elimination or hydrolysis side reactions. Consequently, the crude reaction mixtures obtained from this photo-mediated process are typically cleaner, simplifying downstream purification and enhancing the overall yield of the isolated product, which is critical for maintaining cost-effectiveness in commercial scale-up operations.
How to Synthesize Aryl Internal Alkynes Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment, requiring only a basic setup for light irradiation and inert gas protection. The general procedure involves charging a reaction vessel with the aryl sulfonium salt, the terminal alkyne substrate, a catalytic amount of copper(I) chloride, and an excess of potassium carbonate base in dimethyl sulfoxide (DMSO) solvent. Following the addition of reagents, the system is sealed and purged with nitrogen to create an oxygen-free environment, which is crucial for preventing the oxidation of the copper catalyst and ensuring the longevity of the catalytic cycle. The reaction mixture is then subjected to irradiation from a blue LED light source (typically 2 x 7W bulbs) while stirring at a controlled temperature of approximately 30°C for a period of roughly 20 hours. Detailed standardized synthetic steps, including precise stoichiometric ratios, workup procedures, and purification protocols for specific derivatives, are provided in the guide below.
- Combine aryl sulfonium salt, terminal alkyne, Cu(I) catalyst (e.g., CuCl), and base (e.g., K2CO3) in an organic solvent like DMSO under inert atmosphere.
- Seal the reaction vessel and purge with nitrogen gas to ensure an oxygen-free environment essential for the catalytic cycle.
- Irradiate the mixture with blue LED light (5-15W) at mild temperatures (approx. 30°C) for approximately 20 hours to drive the coupling reaction.
- Monitor reaction progress via TLC, then perform aqueous workup and purify the crude product using column chromatography to isolate the high-purity aryl internal alkyne.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this light-mediated copper catalysis technology presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. By replacing expensive palladium catalysts with commodity-grade copper salts, manufacturers can achieve substantial cost savings on raw materials, insulating their supply chains from the volatile pricing trends of precious metals. Furthermore, the simplified purification requirements resulting from the absence of heavy metal contaminants reduce the consumption of specialized scavenging agents and lower the overall processing time, leading to increased throughput and reduced manufacturing lead times. The mild reaction conditions also translate to lower energy consumption for heating and cooling, aligning with corporate sustainability targets and reducing utility costs associated with large-scale production. These factors collectively enhance the economic viability of producing complex aryl internal alkyne intermediates, making the supply of these critical building blocks more reliable and cost-effective for downstream API synthesis.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts removes one of the most significant cost drivers in traditional cross-coupling reactions, as copper salts are orders of magnitude cheaper and more abundant. This substitution not only lowers the direct material cost but also reduces the indirect costs associated with metal recovery and waste disposal, as copper residues are less regulated and easier to manage than palladium. Additionally, the high atom economy and efficient conversion rates observed in this process minimize the loss of valuable starting materials, further optimizing the cost structure for commercial manufacturing.
- Enhanced Supply Chain Reliability: Utilizing aryl sulfonium salts as coupling partners diversifies the sourcing options for arylating agents, as these salts can be synthesized from a wide range of commercially available aromatic compounds, reducing dependency on specific aryl halide suppliers. The robustness of the copper catalytic system under mild conditions ensures consistent batch-to-batch reproducibility, minimizing the risk of production delays caused by sensitive reaction parameters. This reliability is crucial for maintaining continuous supply lines for key pharmaceutical intermediates, especially in a global market where logistical disruptions can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The use of visible light and ambient temperature conditions makes this process inherently safer and easier to scale compared to high-temperature or high-pressure alternatives, reducing the engineering complexity required for reactor design. The green chemistry attributes, including the use of non-toxic copper catalysts and the generation of benign sulfur byproducts, facilitate compliance with increasingly stringent environmental regulations regarding hazardous waste discharge. This alignment with eco-friendly manufacturing principles not only mitigates regulatory risk but also enhances the brand reputation of chemical suppliers committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photo-mediated copper catalytic method, based on the detailed experimental data and scope exploration provided in the patent literature. These insights are designed to assist technical teams in evaluating the feasibility of adopting this route for their specific project needs, covering aspects from substrate compatibility to process safety. Understanding these nuances is essential for making informed decisions about integrating this technology into existing manufacturing workflows to maximize efficiency and product quality.
Q: What are the primary advantages of using aryl sulfonium salts over traditional aryl halides in this synthesis?
A: Aryl sulfonium salts offer superior stability and are often more readily available or easier to synthesize from common aromatic precursors compared to specific aryl iodides or bromides. Furthermore, they enable the use of earth-abundant copper catalysts instead of expensive palladium systems, significantly reducing raw material costs and heavy metal contamination risks in the final pharmaceutical intermediate.
Q: How does the light-mediated condition impact the scalability and safety of the process?
A: The reaction proceeds efficiently at mild temperatures (around 30°C) using visible blue LED light, eliminating the need for high-temperature heating or hazardous high-pressure conditions often associated with traditional thermal coupling methods. This mild operational window enhances process safety, reduces energy consumption for heating and cooling, and simplifies the engineering requirements for scaling up to multi-kilogram or ton-level production in standard photo-reactors.
Q: Is this method compatible with complex drug-like molecules containing sensitive functional groups?
A: Yes, the methodology demonstrates excellent functional group tolerance, successfully accommodating substrates with esters, nitriles, halides, ethers, and even complex heterocycles. The mild, neutral-to-basic conditions prevent the degradation of acid-sensitive moieties, making it highly suitable for late-stage functionalization of complex pharmaceutical scaffolds where chemoselectivity is paramount.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Internal Alkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this light-mediated copper catalysis technology in reshaping the landscape of pharmaceutical intermediate manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust, GMP-compliant industrial processes. Our state-of-the-art facilities are equipped with advanced photo-reactor systems capable of handling large-volume irradiation reactions, allowing us to leverage this cutting-edge methodology to deliver high-purity aryl internal alkynes with stringent purity specifications. Our rigorous QC labs employ sophisticated analytical techniques to verify the absence of heavy metal impurities and confirm the structural integrity of every batch, guaranteeing that our products meet the exacting standards required by global regulatory agencies.
We invite pharmaceutical companies and research institutions to collaborate with us to explore how this cost-effective and sustainable synthesis route can accelerate your drug development timelines. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, comparing the economic and operational benefits of this copper-catalyzed approach against traditional palladium methods. We encourage you to reach out today to obtain specific COA data for our catalog of aryl alkyne intermediates and to discuss route feasibility assessments for your custom synthesis projects, ensuring a reliable and competitive supply chain for your critical therapeutic candidates.
