Scalable One-Pot Synthesis of 3-Trifluoromethyl Isoxazoles for Advanced Drug Discovery
Introduction to Next-Generation Isoxazole Synthesis
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone strategy in modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of candidate drug molecules. Patent CN107963996B discloses a groundbreaking one-pot methodology for the preparation of 3-trifluoromethyl substituted isoxazole compounds, addressing long-standing inefficiencies in heterocyclic synthesis. This innovative approach leverages commercially available 2,2,2-trifluoroethylamine and terminal alkynes as primary feedstocks, bypassing the need for hazardous or expensive fluorinating agents. By utilizing a cost-effective copper-catalyzed system, the process achieves high yields under mild reaction conditions, offering a robust platform for the rapid generation of diverse chemical libraries essential for high-throughput screening in pharmaceutical and agrochemical research.

The significance of this technological advancement lies in its ability to streamline the supply chain for critical pharmaceutical intermediates. Traditional routes often involve multiple isolation steps and harsh reagents that complicate process safety and environmental compliance. In contrast, this disclosed method operates at room temperature in common solvents like chloroform, significantly lowering the energy footprint and operational complexity. For R&D teams focused on structure-activity relationship (SAR) studies, the broad substrate scope described in the patent allows for the rapid exploration of chemical space around the isoxazole core, accelerating the lead optimization phase without being bottlenecked by synthetic accessibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing 3-trifluoromethyl isoxazoles have been plagued by significant operational and economic drawbacks that hinder their utility in large-scale manufacturing. Early methods, such as those reported by Linderman in 1989, relied on the condensation of trifluoromethyl-substituted alkynones with hydroxylamine, a process characterized by harsh reaction conditions and notoriously poor regioselectivity. This lack of selectivity often results in complex mixtures of isomers, necessitating difficult and yield-eroding purification steps that are unacceptable in GMP environments. Furthermore, subsequent improvements by researchers like Sloop in 2002 utilized trifluoromethyl-substituted 1,3-diones as key intermediates, which themselves require multi-step synthesis, thereby elongating the production timeline and increasing the cumulative cost of goods.
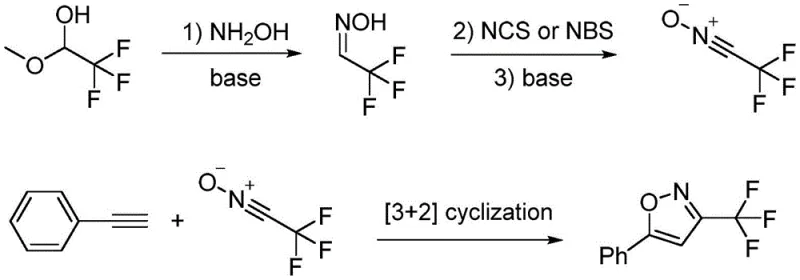
More recent methodologies, including the 2016 reports by Wu and Brown involving trifluoromethyl nitrile oxides, attempted to improve efficiency but still suffered from procedural inefficiencies. These routes typically require the in situ generation of unstable nitrile oxide species through oxidation with reagents like NCS or NBS, followed by a [3+2] cycloaddition. While chemically valid, these processes involve long reaction sequences, low atom economy, and the handling of potentially explosive intermediates. The cumulative effect of these limitations is a synthesis pathway that is fragile, expensive, and difficult to scale, creating a substantial barrier for procurement managers seeking reliable sources of high-purity isoxazole building blocks for commercial API production.
The Novel Approach
The novel one-pot method disclosed in CN107963996B fundamentally reimagines the synthetic logic by merging the formation of the reactive fluorinated species and the cyclization event into a single operational unit. By employing 2,2,2-trifluoroethylamine as a stable, safe, and inexpensive source of the trifluoromethyl group, the method avoids the hazards associated with handling gaseous fluorinating agents or unstable diazo compounds directly. The reaction proceeds via a copper-catalyzed coupling that is remarkably tolerant of ambient conditions, eliminating the need for cryogenic cooling or high-pressure reactors. This simplification translates directly into reduced capital expenditure for manufacturing facilities and lower operating costs, as the process can be executed in standard glass-lined or stainless steel reactors without specialized modifications.
Moreover, the new approach demonstrates superior regioselectivity, consistently delivering the desired 3-trifluoromethyl isoxazole isomer with minimal byproduct formation. This high fidelity in chemical transformation ensures that downstream purification is straightforward, often requiring only basic filtration and chromatography, which preserves yield and maximizes throughput. For supply chain stakeholders, this reliability means shorter lead times and more predictable inventory planning. The ability to access a wide array of substituted isoxazoles from simple terminal alkynes also enhances the flexibility of the supply base, allowing manufacturers to respond rapidly to changing demands in drug development pipelines without retooling entire production lines.
Mechanistic Insights into Copper-Catalyzed Cyclization
The mechanistic elegance of this transformation relies on the synergistic interaction between the copper catalyst and the zinc bromide additive to facilitate the in situ generation of a reactive trifluoromethyl diazo species. Upon mixing 2,2,2-trifluoroethylamine with tert-butyl nitrite (TBN) in the presence of the catalytic system, a fluorodiazomethane intermediate is generated under mild conditions. This highly reactive species then undergoes a concerted [3+2] cycloaddition with the electron-rich triple bond of the terminal alkyne substrate. The presence of zinc bromide is critical, as it likely acts as a Lewis acid to activate the diazo species or stabilize the transition state, thereby lowering the activation energy barrier and allowing the reaction to proceed efficiently at room temperature.
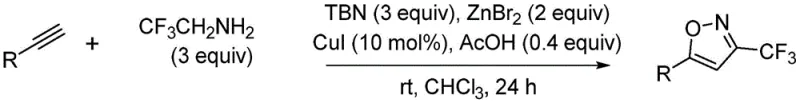
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product quality. Harsh acidic or basic conditions often lead to the decomposition of sensitive functional groups or the polymerization of reactive intermediates, generating complex impurity profiles that are difficult to characterize and remove. In this copper-catalyzed protocol, the neutral to slightly acidic environment maintained by the acetic acid additive prevents such degradation pathways. Additionally, the high chemoselectivity of the copper catalyst ensures that only the alkyne moiety participates in the cyclization, leaving other sensitive groups such as esters, nitriles, and halides intact. This orthogonal reactivity is essential for the synthesis of complex pharmaceutical intermediates where late-stage functionalization is required.
The broad substrate scope demonstrated in the patent underscores the robustness of this mechanistic pathway. A diverse library of terminal alkynes, ranging from simple phenylacetylenes to complex heterocycles and protected amino acids, successfully undergoes the transformation with yields ranging from 58% to 93%. This consistency suggests that the catalytic cycle is not easily poisoned by common coordinating groups found in drug molecules. For process chemists, this implies that the method can be applied to a wide variety of targets with minimal need for condition optimization, significantly accelerating the path from bench-scale discovery to pilot plant validation.
How to Synthesize 3-Trifluoromethyl Isoxazole Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and addition order to maximize the generation of the reactive diazo intermediate while minimizing side reactions. The standard procedure involves dissolving the terminal alkyne in chloroform, followed by the sequential addition of trifluoroethylamine, tert-butyl nitrite, and acetic acid. The catalytic system, consisting of cuprous iodide and zinc bromide, is then introduced, often with initial cooling to manage the exotherm of diazo formation before allowing the mixture to warm to room temperature for the extended reaction period. Detailed standardized operating procedures for scaling this reaction from milligram to kilogram quantities are provided in the technical documentation below.
- Dissolve the terminal alkyne substrate in chloroform and add 2,2,2-trifluoroethylamine, tert-butyl nitrite, and acetic acid under nitrogen protection.
- Introduce the catalytic system comprising cuprous iodide and zinc bromide reagent, ensuring full dissolution possibly with ice-water bath cooling initially.
- Stir the reaction mixture at room temperature for 24 hours, then proceed to workup via filtration, washing, and flash column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis method offers transformative benefits in terms of cost structure and supply security. The primary driver of cost reduction is the substitution of expensive, multi-step intermediates with commodity chemicals. 2,2,2-Trifluoroethylamine and terminal alkynes are produced on a massive industrial scale, ensuring a stable and competitive pricing landscape. By eliminating the need for custom-synthesized precursors like trifluoromethyl alkynones, the bill of materials is drastically simplified, leading to substantial savings in raw material costs. Furthermore, the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium removes the financial burden associated with metal recovery and residual metal testing, which are significant cost centers in API manufacturing.
Enhanced supply chain reliability is another critical advantage derived from the simplicity and robustness of this process. Traditional multi-step syntheses are prone to bottlenecks where a failure in any single step can halt the entire production campaign. The convergent nature of this one-pot reaction reduces the number of unit operations, thereby minimizing the risk of process failures and batch rejections. The mild reaction conditions also mean that the process can be manufactured in a wider range of facilities, including those without specialized high-pressure or cryogenic capabilities. This flexibility diversifies the potential supplier base, reducing the risk of supply disruption due to equipment downtime or regulatory issues at a single manufacturing site.
Scalability and environmental compliance are seamlessly addressed by this green chemistry approach. The reaction operates at room temperature, significantly reducing energy consumption compared to processes requiring reflux or cooling. The high atom economy and reduced solvent usage associated with one-pot procedures align with increasingly stringent environmental regulations and corporate sustainability goals. Waste generation is minimized as there are fewer intermediate isolation steps and less solvent required for purification. For companies aiming to reduce their carbon footprint and meet ESG (Environmental, Social, and Governance) targets, adopting this efficient synthesis route provides a tangible pathway to greener manufacturing without compromising on yield or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel isoxazole synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent literature, offering a realistic assessment of the method's capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this chemistry into existing production workflows or new drug development programs.
Q: What are the primary advantages of this one-pot method over traditional multi-step synthesis?
A: This method eliminates the need for pre-synthesized complex intermediates like trifluoromethyl alkynones or 1,3-diones, significantly reducing step count, waste generation, and overall production time while maintaining high regioselectivity.
Q: Is the catalytic system suitable for large-scale commercial production?
A: Yes, the process utilizes inexpensive and commercially available catalysts (CuI/ZnBr2) and operates at room temperature without stringent pressure or cryogenic requirements, making it highly amenable to kilogram-to-ton scale-up.
Q: What is the functional group tolerance of this synthesis protocol?
A: The protocol demonstrates exceptional tolerance for diverse functional groups including halogens, nitro, cyano, esters, and protected amines, allowing for the direct synthesis of complex drug-like molecules without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Isoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the success of modern drug discovery and development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including specialized containment and waste treatment systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl isoxazole meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this innovative one-pot synthesis can optimize your specific project requirements. Whether you need custom analogs for SAR studies or bulk quantities for clinical trials, we are prepared to provide a Customized Cost-Saving Analysis tailored to your molecule. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation that reflects the economic advantages of this advanced manufacturing technology.
