Revolutionizing Aminopyridine Production: A Solvent-Free, Catalyst-Free Thermal Amination Strategy for Commercial Scale-Up
Introduction to Breakthrough Aminopyridine Synthesis Technology
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and scalable synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN111732536A, which discloses a novel method for synthesizing aminopyridine compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a robust, solvent-free thermal amination strategy. By leveraging the thermal decomposition of solid ammoniating reagents, specifically zinc ammonium chloride, this method achieves high conversion rates at temperatures between 200°C and 240°C without the need for expensive palladium catalysts or voluminous organic solvents. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediates supplier, this patent offers a compelling value proposition: drastically simplified downstream processing, reduced environmental footprint, and enhanced economic viability for large-scale production.
The significance of aminopyridine derivatives in the life sciences cannot be overstated, as they serve as critical building blocks for a myriad of bioactive molecules, including kinase inhibitors and agrochemical agents. However, their synthesis has historically been plagued by inefficiencies associated with noble metal catalysis and hazardous solvent usage. The methodology outlined in CN111732536A addresses these pain points directly by utilizing a closed autoclave system where the amination reagent decomposes in situ to generate ammonia gas. This internal pressure generation facilitates the nucleophilic substitution of halogen atoms on the pyridine ring, resulting in high-purity products after a straightforward aqueous workup. This report delves deep into the mechanistic advantages, operational parameters, and commercial implications of adopting this cutting-edge synthesis route for your supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the amination of halopyridines has relied heavily on palladium-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination. While effective for complex substrates, these methods introduce significant bottlenecks for industrial scale-up. As illustrated in prior art literature, reactions employing Pd(OAc)2/XantPhos catalytic systems often require microwave heating and organic solvents like dioxane, yet still suffer from disappointingly low yields, frequently hovering around only 30%. 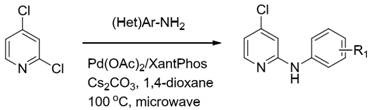
Beyond the issue of poor atom economy, the reliance on transition metals necessitates rigorous purification steps to ensure residual metal levels meet stringent regulatory standards for pharmaceutical ingredients. This adds layers of complexity and cost to the manufacturing process, involving specialized scavengers and additional chromatography or crystallization steps. Furthermore, the use of volatile organic solvents generates substantial hazardous waste, increasing the burden on environmental compliance and waste treatment facilities. For a procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing, these factors translate into higher raw material costs, longer lead times, and increased operational risk.
The Novel Approach
In stark contrast, the novel approach described in CN111732536A eliminates the need for external catalysts and solvents entirely. The core innovation lies in the use of solid zinc ammonium chloride as an ammonia source, which undergoes thermal decomposition at elevated temperatures to release ammonia gas directly within the reaction vessel. This solvent-free protocol not only simplifies the reaction setup but also dramatically improves the reaction efficiency, with experimental data showing yields reaching up to 75-80% for key substrates like 2-chloro-3-nitropyridine. 
The operational simplicity of this method is a major advantage for supply chain heads concerned with scalability. By removing the solvent, the volume of the reaction mixture is significantly reduced, allowing for higher throughput in existing reactor infrastructure. The workup procedure is equally streamlined, involving a simple water quench followed by extraction and pH adjustment to precipitate the product. This reduction in unit operations translates to faster batch cycles and lower energy consumption. For organizations seeking a reliable pharmaceutical intermediates supplier, this technology promises a more resilient supply chain capable of delivering high-purity aminopyridines with consistent quality and reduced dependency on volatile precious metal markets.
Mechanistic Insights into Solvent-Free Thermal Amination
The success of this synthesis route hinges on the unique thermal properties of the ammoniating reagent, zinc ammonium chloride. Upon heating to the optimal range of 220°C to 240°C, the solid reagent undergoes a stepwise decomposition process. Initially, it releases ammonia molecules, which build up pressure within the sealed autoclave. This high-pressure environment is crucial for driving the nucleophilic aromatic substitution (SnAr) reaction, particularly for less activated halopyridines. The in situ generation of ammonia ensures a high local concentration of the nucleophile at the reaction interface, overcoming the kinetic barriers that typically limit gas-liquid reactions. This mechanism allows for the efficient displacement of chlorine or bromine atoms even in the absence of a metal catalyst to lower the activation energy.
Furthermore, the reaction conditions are finely tuned to maximize selectivity and minimize byproduct formation. The patent specifies a molar ratio of halopyridine to ammoniating reagent between 1:2 and 1:5. This excess of the solid reagent serves a dual purpose: it ensures complete consumption of the valuable halogenated substrate and maintains sufficient internal pressure throughout the 4 to 7-hour reaction window. The substrate scope is remarkably broad, accommodating various electron-withdrawing groups such as nitro, trifluoromethyl, and acetyl moieties, which further activate the pyridine ring towards nucleophilic attack. 
Impurity control is inherently built into the process design. Since no external ligands or metal salts are introduced, the impurity profile is significantly cleaner compared to catalytic methods. Post-reaction, the unreacted zinc salts and ammonium byproducts are highly water-soluble. A simple aqueous workup effectively separates these inorganic residues from the organic aminopyridine product. Subsequent pH adjustment to the 8-9 range induces precipitation of the target amine, facilitating isolation via filtration. This elegant separation strategy avoids the need for complex distillation or column chromatography, ensuring that the final product meets high-purity specifications required for downstream drug synthesis.
How to Synthesize Aminopyridine Compounds Efficiently
Implementing this solvent-free thermal amination protocol requires precise control over temperature and pressure parameters to ensure safety and reproducibility. The process begins with the careful loading of stoichiometric amounts of the halopyridine substrate and solid zinc ammonium chloride into a high-pressure autoclave rated for temperatures exceeding 250°C. It is critical to maintain an inert atmosphere during loading to prevent oxidation, although the reducing environment of the reaction generally mitigates this risk. Once sealed, the reactor is heated to the target temperature of 220°C to 240°C with continuous stirring to ensure uniform heat distribution and efficient mass transfer between the solid reagent and the molten or dissolved substrate.
- Load halogenated pyridine substrate and solid zinc ammonium chloride (molar ratio 1: 2 to 1:5) into a high-pressure autoclave.
- Heat the mixture to 200-240°C under stirring for 4-7 hours to facilitate thermal decomposition and nucleophilic substitution.
- Quench the reaction with water, extract impurities with organic solvents like MTBE, and adjust aqueous pH to 8-9 to precipitate the pure aminopyridine product.
Following the reaction period, typically 5 hours for optimal conversion, the system must be cooled safely before opening. The crude reaction mass is then treated with water to dissolve inorganic salts, followed by extraction with green solvents such as methyl tert-butyl ether (MTBE) or isopropyl acetate to remove non-polar organic impurities. The aqueous phase containing the protonated amine is then basified with sodium hydroxide to pH 8-9, causing the free amine to precipitate as a solid. This solid is filtered, washed, and dried to yield the high-purity aminopyridine compound, ready for further functionalization or salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis method offers transformative benefits for the commercial viability of aminopyridine production. From a procurement perspective, the elimination of palladium catalysts and phosphine ligands removes a significant cost driver and supply risk. Precious metals are subject to extreme price volatility and geopolitical supply constraints; replacing them with commodity chemicals like zinc ammonium chloride stabilizes the cost of goods sold (COGS). Additionally, the solvent-free nature of the reaction drastically reduces the volume of hazardous waste generated, leading to substantial cost savings in waste disposal and environmental compliance fees. These factors combined contribute to a more predictable and economical manufacturing model.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated purification steps significantly lowers the overall production cost. Without the need for metal scavengers or extensive chromatography, the operational expenditure is reduced, allowing for more competitive pricing in the market. The high yield achieved (up to 80% in some examples) further enhances material efficiency, minimizing the loss of valuable starting materials and maximizing output per batch.
- Enhanced Supply Chain Reliability: Relying on readily available solid reagents rather than specialized catalytic systems improves supply chain resilience. Zinc ammonium chloride is a stable, easy-to-transport commodity, reducing the risk of delays associated with sourcing sensitive organometallic reagents. The robustness of the high-temperature process also ensures consistent batch-to-batch quality, reducing the likelihood of production failures or out-of-specification results that could disrupt downstream manufacturing schedules.
- Scalability and Environmental Compliance: The solvent-free design is inherently safer and easier to scale from pilot plant to commercial production. The absence of flammable organic solvents reduces fire hazards and simplifies reactor design requirements. Moreover, the significant reduction in volatile organic compound (VOC) emissions aligns with increasingly strict global environmental regulations, positioning manufacturers as leaders in sustainable chemistry and facilitating smoother regulatory approvals for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free thermal amination technology. These insights are derived directly from the experimental data and technical specifications provided in patent CN111732536A, offering clarity on process safety, substrate compatibility, and quality control measures. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the solvent-free thermal amination method superior to traditional Pd-catalyzed routes?
A: Traditional Pd-catalyzed methods often suffer from low yields (around 30%), high catalyst costs, and complex purification requirements to remove heavy metals. The solvent-free thermal method eliminates expensive catalysts entirely, operates without organic solvents to reduce waste, and achieves significantly higher conversion rates through high-pressure in situ ammonia generation.
Q: What are the safety considerations for operating at 200-240°C?
A: The reaction utilizes solid zinc ammonium chloride which decomposes to release ammonia gas in a controlled manner within a closed autoclave system. This high-pressure environment enhances the contact between ammonia and the halopyridine substrate, driving the reaction forward while maintaining safety through standard high-pressure reactor protocols.
Q: Which types of halopyridine substrates are compatible with this synthesis method?
A: The method demonstrates broad substrate tolerance, successfully converting various chloro- and bromo-pyridines substituted with electron-withdrawing groups such as nitro, trifluoromethyl, and acetyl groups. This versatility makes it suitable for synthesizing a wide range of pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminopyridine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the solvent-free thermal amination process described in CN111732536A and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications for complex aminopyridine intermediates.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this technology can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most advanced and reliable chemistry available.
