Advanced Purification Technology for Elagolix Sodium Intermediates Enabling Commercial Scale-Up
Advanced Purification Technology for Elagolix Sodium Intermediates Enabling Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies to enhance the quality of active pharmaceutical ingredients (APIs), particularly for complex molecules like Elagolix Sodium, a potent GnRH antagonist. Patent CN112694445A introduces a groundbreaking purification strategy specifically designed for the key intermediate, known chemically as the compound of Formula II. This innovation addresses a critical bottleneck in the supply chain where traditional synthesis routes often result in intermediates with insufficient purity, typically hovering around 80%, which compromises the quality of the final drug substance. By implementing a specialized salt formation technique using L-tartaric acid, manufacturers can now achieve purity levels exceeding 99%, ensuring that the downstream synthesis of Elagolix Sodium meets rigorous regulatory standards. This technological leap not only resolves long-standing impurity issues but also streamlines the production workflow, making it an essential advancement for reliable pharmaceutical intermediate supplier networks aiming for excellence.
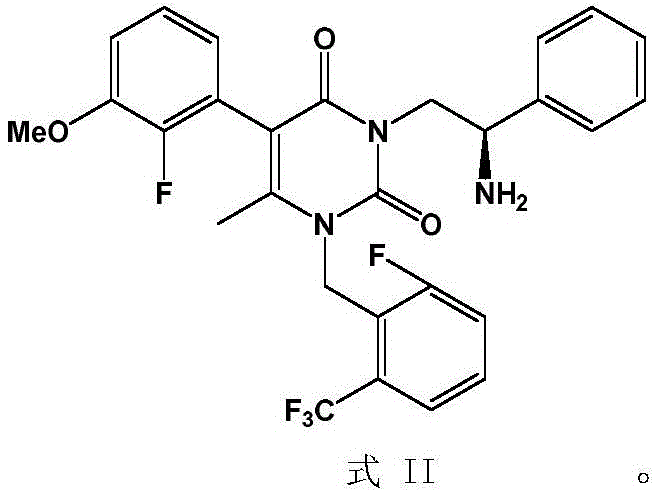
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Elagolix Sodium has relied on multi-step routes, such as the one disclosed in WO2005007164a1, which involves ring closing, iodination, coupling, and hydrolysis steps. While these pathways successfully construct the molecular skeleton, they frequently generate a crude intermediate product laden with structurally similar impurities and by-products that are difficult to remove. The lack of an effective purification protocol for this specific intermediate means that manufacturers often struggle with batch-to-batch variability and suboptimal yields in the final salification step. Furthermore, attempting to purify such complex heterocyclic compounds using standard recrystallization or column chromatography on an industrial scale is often economically unviable and technically challenging due to solubility issues. Consequently, the accumulation of impurities from the intermediate stage propagates through the synthesis, leading to costly rework or even batch rejection, thereby disrupting the continuity of the supply chain for this vital medication.
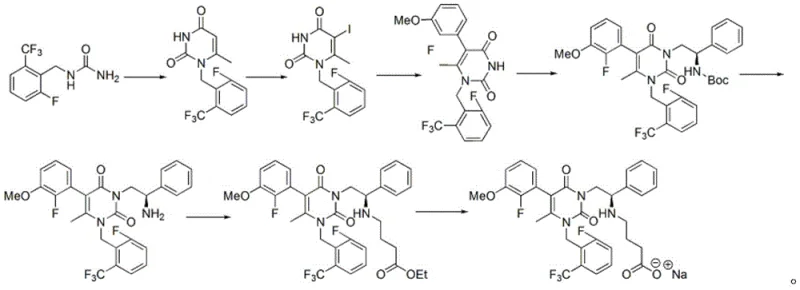
The Novel Approach
In stark contrast to these cumbersome traditional methods, the novel approach detailed in the patent utilizes a highly selective salt formation and dissociation strategy that dramatically simplifies the purification landscape. By reacting the crude intermediate with L-tartaric acid in a specific solvent system, the process selectively precipitates the desired compound as a stable tartrate salt, effectively leaving the majority of impurities in the mother liquor. This method transforms a difficult liquid-liquid separation or chromatographic challenge into a straightforward solid-liquid filtration operation, which is inherently more scalable and reproducible. The subsequent liberation of the free base using mild inorganic bases ensures that the chemical integrity of the sensitive uracil backbone is maintained while achieving exceptional purity. This shift from complex separation techniques to crystallization-driven purification represents a paradigm shift in process chemistry, offering a clear path toward cost reduction in pharmaceutical intermediates manufacturing by minimizing solvent consumption and processing time.
Mechanistic Insights into L-Tartaric Acid Mediated Salt Formation
The core of this purification technology lies in the specific molecular recognition and lattice energy differences between the target intermediate and its impurities when interacting with L-tartaric acid. When the crude amine-containing intermediate is introduced to L-tartaric acid in ethanol, a diastereomeric salt or a specific crystalline complex is formed that possesses significantly lower solubility than the corresponding salts of the impurities. This differential solubility is the driving force behind the high efficiency of the purification, as it allows the target molecule to nucleate and grow into large, filterable crystals while excluding foreign species. The choice of L-tartaric acid is critical, as screening data revealed that other common acids like fumaric or citric acid failed to induce precipitation, highlighting the unique stereochemical and hydrogen-bonding compatibility required for this specific substrate. Understanding this mechanism allows process chemists to fine-tune parameters such as temperature and solvent ratio to maximize the recovery of the pure salt without compromising the exclusion of contaminants.
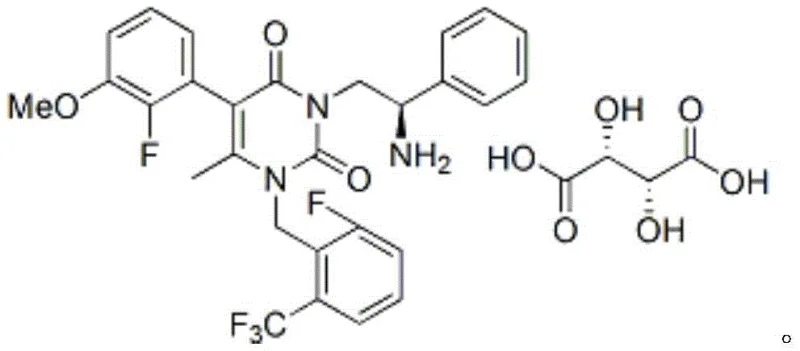
Furthermore, the dissociation step is meticulously designed to ensure that the high purity achieved during salt formation is not lost during the regeneration of the free base. By utilizing a biphasic system involving water and an organic solvent like ethyl acetate or dichloromethane, the process ensures that the inorganic salts and any residual tartaric acid remain in the aqueous phase. The use of mild bases such as sodium carbonate or potassium bicarbonate prevents the degradation of the ester or amide functionalities present in the molecule, which could occur under strongly alkaline conditions. This careful control of pH and phase separation ensures that the final isolated product retains the structural fidelity required for the subsequent coupling reactions in the API synthesis. The result is a highly pure intermediate that serves as a robust foundation for the final drug substance, minimizing the risk of genotoxic impurities or difficult-to-remove by-products in the final API.
How to Synthesize Elagolix Sodium Intermediate Efficiently
To implement this purification protocol effectively, manufacturers must adhere to precise operational parameters regarding solvent volumes and temperature controls to ensure optimal crystal growth and yield. The process begins with the dissolution of the crude material in ethanol, followed by the controlled addition of the acid to initiate nucleation, a step that requires careful monitoring to prevent oiling out or the formation of amorphous solids. Detailed standardized operating procedures are essential to replicate the high success rates observed in the patent examples, particularly regarding the stirring times and filtration techniques used to isolate the tartrate salt. For a comprehensive breakdown of the exact reagent quantities, reaction times, and workup procedures, please refer to the technical guide below.
- Dissolve the crude compound of Formula II in ethanol (3-10 volumes) at 25°C and add L-tartaric acid (1 equivalent) to form the tartrate salt.
- Stir the mixture at 20-40°C for approximately 22 hours to allow complete crystallization of the pure tartrate salt solid.
- Suspend the isolated salt in an aqueous organic solvent system, treat with an inorganic base like sodium carbonate to liberate the free base, and extract to obtain high-purity Formula II.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification method offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for complex pharmaceutical intermediates. The ability to consistently produce intermediates with purity greater than 99% significantly reduces the risk of downstream processing failures, thereby enhancing the overall reliability of the API supply chain. By eliminating the need for expensive and time-consuming chromatographic purification steps, manufacturers can achieve significant cost savings in terms of both consumables and labor, translating into more competitive pricing for the final active ingredient. Moreover, the use of common, non-hazardous solvents like ethanol and ethyl acetate simplifies waste management and aligns with increasingly stringent environmental regulations, reducing the compliance burden on production facilities. This combination of high yield, superior purity, and operational simplicity makes the technology a cornerstone for establishing a resilient and cost-effective supply network for Elagolix Sodium.
- Cost Reduction in Manufacturing: The elimination of complex chromatography and the use of inexpensive, recyclable solvents like ethanol drastically lower the variable costs associated with production. By converting a low-yield, high-waste process into a high-efficiency crystallization workflow, the overall cost of goods sold is significantly reduced, allowing for better margin management in a competitive generic market.
- Enhanced Supply Chain Reliability: The robustness of the salt formation method ensures consistent batch quality, minimizing the likelihood of production delays caused by out-of-specification materials. This reliability is crucial for maintaining continuous API production schedules and meeting the demanding delivery timelines required by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable from kilogram to tonne levels without requiring specialized equipment, facilitating rapid capacity expansion to meet market demand. Additionally, the reduced solvent footprint and avoidance of heavy metal catalysts or toxic reagents contribute to a greener manufacturing profile, satisfying corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and quality advantages of the L-tartaric acid method for stakeholders evaluating this process for potential adoption. Understanding these details is vital for making informed decisions about process validation and technology transfer.
Q: Why is L-tartaric acid preferred over other acids for this purification?
A: Experimental data indicates that L-tartaric acid uniquely facilitates the precipitation of a stable crystalline salt with the intermediate, whereas acids like fumaric or citric acid fail to induce solid formation under identical conditions.
Q: What purity levels can be achieved with this new method compared to conventional synthesis?
A: While conventional synthetic routes often yield crude intermediates with purity around 80%, this salt formation and dissociation method consistently achieves final product purity exceeding 99.3%.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like ethanol and ethyl acetate and avoids complex chromatography, making it highly scalable and cost-effective for manufacturing multi-kilogram batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Elagolix Sodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful manufacture of life-saving medications like Elagolix Sodium. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle complex purification challenges, leveraging technologies like the one described in CN112694445A to guarantee the supply of premium-grade intermediates.
We invite you to collaborate with us to explore how our advanced manufacturing capabilities can support your specific project requirements and drive efficiency in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to being your trusted partner in pharmaceutical development and commercialization.
