Advanced Posaconazole Synthesis: A Safer, High-Purity Debenzylation Strategy for Global Pharma
Advanced Posaconazole Synthesis: A Safer, High-Purity Debenzylation Strategy for Global Pharma
The pharmaceutical landscape for antifungal treatments continues to evolve, with Posaconazole standing as a critical second-generation triazole drug for managing invasive fungal infections. As detailed in patent CN109970725B, a significant technological breakthrough has been achieved in the final synthetic step of this potent API. This patent discloses a novel preparation method that utilizes ammonium formate-mediated transfer hydrogenation to convert the key benzyl-protected intermediate (Compound I) into the final active pharmaceutical ingredient. This approach represents a paradigm shift from traditional, hazardous debenzylation protocols, offering a pathway that is not only chemically elegant but also industrially robust. For R&D directors and process chemists, this method solves long-standing issues regarding impurity profiles and reaction safety, while providing supply chain leaders with a more scalable and cost-effective manufacturing route.
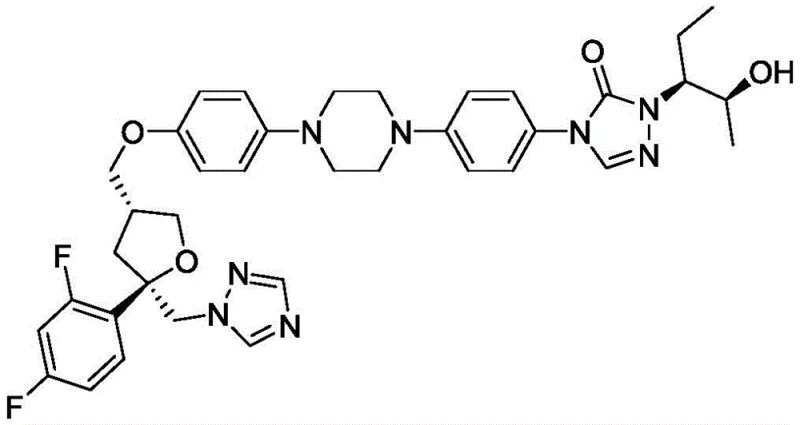
The structural complexity of Posaconazole demands precise synthetic control, particularly in the final deprotection stages where the integrity of the chiral centers and the triazole rings must be preserved. The innovation described in CN109970725B leverages the mild reducing power of ammonium formate in the presence of a palladium catalyst to cleave the benzyl ether bond. This specific transformation is pivotal because it avoids the harsh acidic or high-pressure conditions that have historically plagued the production of this molecule. By adopting this methodology, manufacturers can achieve superior product quality with significantly reduced operational risks, positioning this synthesis route as a preferred choice for reliable pharmaceutical intermediates supplier networks aiming for excellence in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the debenzylation of Compound I to produce Posaconazole has relied on several problematic methodologies that pose severe challenges for industrial scale-up. Traditional protocols often employ strong inorganic protonic acids, such as concentrated hydrochloric acid or hydrobromic acid, which necessitate heating and create highly corrosive environments. These acidic conditions not only demand expensive, acid-resistant reactor materials but also frequently lead to the formation of halogenated genotoxic impurities, a critical quality attribute failure in modern pharmaceutical manufacturing. Furthermore, the solubility differences between the starting material and the product in these acidic media are often minimal, making purification arduous and resulting in lower overall yields.
Alternative methods utilizing boron trichloride require cryogenic conditions, typically ranging from -80°C to -40°C, which imposes a massive energy burden and limits batch sizes due to cooling capacity constraints. Additionally, boron trichloride is moisture-sensitive and hazardous to transport and handle, introducing significant safety liabilities. Another common approach involves catalytic hydrogenation using gaseous hydrogen, which requires high-pressure autoclaves and rigorous safety protocols to mitigate explosion risks. These conventional routes are fraught with operational complexities, high capital expenditure for specialized equipment, and environmental concerns regarding waste disposal, making them suboptimal for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the method disclosed in CN109970725B introduces a remarkably mild and efficient strategy using ammonium formate as a hydrogen donor. This transfer hydrogenation technique operates under atmospheric pressure and moderate temperatures, typically between 60°C and 75°C, completely eliminating the need for high-pressure vessels or cryogenic cooling. The reaction proceeds smoothly in common alcohol solvents like methanol or ethanol, facilitating easy handling and workup. Crucially, this neutral reaction environment prevents the formation of acid-induced degradation products and halogenated impurities, ensuring a cleaner crude profile that simplifies downstream purification.
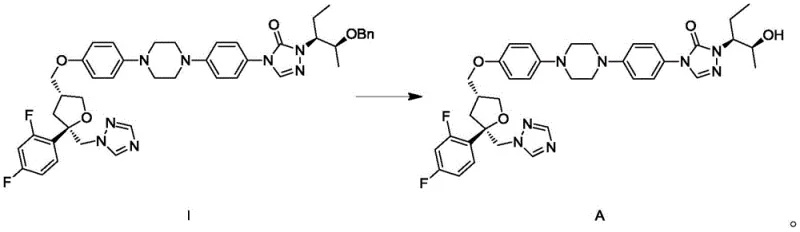
The novelty of this approach lies in its operational simplicity and safety profile. By replacing hazardous reagents with solid ammonium formate and standard palladium catalysts, the process becomes inherently safer and more amenable to large-scale production. The absence of strong acids means there is no need for subsequent neutralization steps with alkaline solutions, which traditionally generate large volumes of saline wastewater. This streamlined workflow not only enhances the commercial scale-up of complex pharmaceutical intermediates but also aligns perfectly with green chemistry principles by reducing waste generation and energy consumption. The result is a high-yielding process that delivers Posaconazole with exceptional purity, addressing the critical needs of both quality assurance and production efficiency.
Mechanistic Insights into Ammonium Formate-Mediated Transfer Hydrogenation
The core of this innovative synthesis lies in the mechanism of transfer hydrogenation, where ammonium formate serves as a convenient source of nascent hydrogen. In the presence of a palladium catalyst, such as wet palladium on carbon or palladium hydroxide, ammonium formate decomposes to generate hydrogen gas and carbon dioxide in situ. However, rather than evolving as a gas, the hydrogen is adsorbed onto the surface of the palladium catalyst, forming active palladium-hydride species. These surface-bound hydrides are then transferred directly to the benzyl ether substrate (Compound I), facilitating the cleavage of the carbon-oxygen bond. This heterogeneous catalytic cycle ensures that the reduction occurs selectively at the benzyl group without affecting other sensitive functional groups within the complex Posaconazole scaffold, such as the triazole rings or the chiral alcohol moieties.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed hydrolysis. Since the reaction does not involve free protons or halide ions, pathways leading to nucleophilic substitution or acid-catalyzed rearrangement are effectively shut down. This mechanistic cleanliness is vital for preventing the formation of genotoxic alerts, which are strictly regulated in final drug substances. Furthermore, the use of wet palladium carbon allows for controlled reactivity, minimizing the risk of over-reduction or catalyst poisoning. The ability to monitor the reaction progress via HPLC until the complete disappearance of Compound I ensures consistent batch-to-batch reproducibility. This deep understanding of the catalytic cycle allows process chemists to fine-tune parameters such as catalyst loading and temperature to optimize the high-purity pharmaceutical intermediates output.
How to Synthesize Posaconazole Efficiently
The synthesis of Posaconazole via this novel debenzylation route is designed for operational ease and scalability. The process begins by dissolving the benzyl-protected precursor, Compound I, in a suitable alcohol solvent, with methanol being the preferred choice due to its excellent solubility properties and cost-effectiveness. To this solution, a specific grade of palladium catalyst, typically wet palladium on carbon with a defined water content, is added along with a stoichiometric excess of ammonium formate. The mixture is then heated to a moderate temperature range, allowing the transfer hydrogenation to proceed under atmospheric pressure. Detailed standardized synthesis steps see the guide below.
- Dissolve Compound I in an alcohol solvent such as methanol or ethanol within a standard reaction vessel.
- Add wet palladium on carbon catalyst and ammonium formate as the hydrogen donor under atmospheric pressure.
- Heat the mixture to 60-75°C for 16-18 hours, followed by hot filtration and recrystallization to obtain high-purity Posaconazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ammonium formate-based synthesis route offers transformative economic and logistical benefits. The elimination of high-pressure hydrogenation equipment and cryogenic reactors significantly lowers the capital expenditure required for manufacturing facilities. This reduction in infrastructure complexity translates directly into lower overhead costs and faster turnaround times for production campaigns. Moreover, the use of stable, solid reagents like ammonium formate simplifies raw material logistics, removing the hazards and regulatory burdens associated with transporting compressed gases or corrosive liquids. This stability ensures a more resilient supply chain, less susceptible to disruptions caused by hazardous material transport restrictions.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive acid-resistant equipment and complex waste treatment systems associated with neutralizing strong acids. The simplified workup procedure, which omits pH adjustment steps, reduces labor hours and consumable usage, leading to a more lean manufacturing operation. Additionally, the high selectivity of the reaction minimizes raw material loss, improving the overall mass balance and reducing the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: By utilizing commonly available solvents and catalysts, the method reduces dependency on specialized or scarce reagents. The robustness of the reaction conditions allows for flexible scheduling and easier technology transfer between different manufacturing sites. This flexibility is crucial for maintaining continuous supply during market fluctuations or unexpected demand surges, ensuring that downstream formulation partners receive their materials on time without compromise.
- Scalability and Environmental Compliance: The atmospheric pressure operation and moderate temperatures make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduction in hazardous waste generation, particularly the avoidance of halogenated byproducts and saline effluents, simplifies environmental compliance and lowers disposal costs. This alignment with sustainability goals enhances the corporate social responsibility profile of the manufacturing entity, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this advanced Posaconazole synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the practical implementation and benefits of this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is ammonium formate preferred over hydrogen gas for this debenzylation?
A: Ammonium formate acts as a safe, in-situ hydrogen donor, eliminating the need for high-pressure hydrogenation equipment and reducing explosion risks associated with gaseous hydrogen.
Q: Does this method avoid genotoxic impurities?
A: Yes, unlike traditional methods using concentrated hydrochloric or hydrobromic acid, this neutral transfer hydrogenation process prevents the formation of halogenated genotoxic impurities.
Q: What is the typical purity achieved with this novel process?
A: The process consistently yields Posaconazole with purity exceeding 99.5% after recrystallization, meeting stringent pharmaceutical quality standards without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ammonium formate debenzylation of Posaconazole are translated into reality with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of API intermediate meets the highest international standards, providing our partners with absolute confidence in product quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener, safer route can optimize your bottom line. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why NINGBO INNO PHARMCHEM is the preferred partner for reducing lead time for high-purity pharmaceutical intermediates in a competitive marketplace.
