Revolutionizing Olefin Functionalization: A Catalyst-Free One-Pot Route for High-Value Intermediates
Introduction to Patent CN111620798A
The landscape of organic synthesis for functionalized olefins is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes. Patent CN111620798A introduces a groundbreaking methodology for the synthesis of (Z)-beta-iodo-beta-methylthio olefins, a class of compounds that serves as pivotal building blocks in the construction of complex biomedical molecules and photochemical materials. This technology represents a paradigm shift from traditional multi-step organometallic pathways to a streamlined, catalyst-free thermal process. By utilizing simple terminal alkenes, dimethyl sulfoxide (DMSO), and elemental iodine, this invention eliminates the reliance on expensive and hazardous metal reagents, thereby addressing critical pain points in the supply chain for reliable pharmaceutical intermediates supplier networks. The ability to generate these high-value scaffolds in a single pot under mild conditions not only enhances operational safety but also drastically simplifies the purification workflow, positioning this technology as a cornerstone for next-generation fine chemical manufacturing.
The strategic importance of this patent lies in its dual functionality; it acts as both a synthetic breakthrough and a supply chain optimizer. For R&D directors focused on impurity profiles and process robustness, the elimination of transition metal residues is a game-changer, ensuring higher purity specifications without the need for costly scavenging steps. Furthermore, the reaction demonstrates exceptional substrate tolerance, accommodating a wide array of electronic and steric environments on the aromatic ring, from electron-donating groups like tert-butyl to electron-withdrawing halogens. This versatility ensures that the method is not limited to model substrates but is applicable to the diverse library of compounds required in modern drug discovery. As we delve deeper into the technical specifics, it becomes evident that this approach offers a viable pathway for cost reduction in fine chemical manufacturing, aligning perfectly with the goals of procurement managers seeking to stabilize raw material costs and reduce dependency on volatile metal markets.
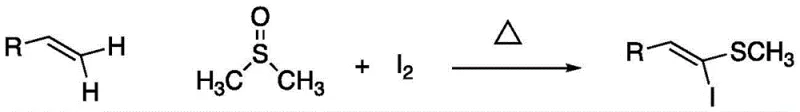
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-iodo-beta-alkylthio arylethenes has been fraught with complexity and inefficiency, relying heavily on alkyne precursors that require extensive pre-functionalization. Conventional literature, such as the work by Jin et al. and Cai et al., describes routes that necessitate the preparation of alkynyl sulfides through reactions with thioalkanes or chlorothioalkanes, often involving sensitive Grignard reagents or organoaluminum species. These multi-step sequences are not only time-consuming but also introduce significant risks associated with handling pyrophoric materials and moisture-sensitive intermediates. The requirement for specialized reagents like trimethylsilyl iodide (TMS-I) further exacerbates the cost burden, as these materials are expensive to procure and store. Moreover, the use of organotin compounds in alternative pathways poses severe environmental and regulatory challenges due to the toxicity of tin residues, which are notoriously difficult to remove to the stringent levels required for pharmaceutical applications. Consequently, the existing state-of-the-art creates a bottleneck in the commercial scale-up of complex polymer additives and API intermediates, limiting the availability of these valuable building blocks.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach disclosed in CN111620798A leverages a direct double substitution reaction on the terminal carbon atoms of simple olefins. This method bypasses the need for alkyne precursors entirely, starting instead from readily available terminal alkenes which are commodity chemicals with stable pricing and abundant global supply. The reaction proceeds via a unique mechanism where DMSO serves a dual role as both the solvent and the source of the methylthio group, while elemental iodine acts as the iodinating agent. This convergence of functions into a single pot reaction dramatically reduces the number of unit operations, solvent consumption, and waste generation. The process operates under atmospheric conditions without the need for inert gas protection or anhydrous environments, significantly lowering the barrier to entry for industrial adoption. By achieving high selectivity for the (Z)-isomer without the use of chiral ligands or transition metal catalysts, this technology offers a robust solution for reducing lead time for high-purity pharmaceutical intermediates, enabling faster iteration cycles in process development and quicker time-to-market for new therapeutic candidates.
Mechanistic Insights into Radical-Mediated Double Substitution
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction is proposed to proceed through a radical pathway initiated by the thermal homolysis of elemental iodine and the decomposition of DMSO. Under heating conditions, DMSO slowly cleaves to generate methanethiol and formaldehyde, while iodine molecules dissociate into iodine radicals. These iodine radicals then abstract a hydrogen atom from the methanethiol to produce methylthio radicals and hydrogen iodide. Concurrently, the terminal alkene undergoes an iodination-substitution reaction with molecular iodine to form a beta-iodoalkene intermediate. The key step involves the addition of the methylthio radical to this beta-iodoalkene intermediate, forming a carbon-centered radical species. This intermediate subsequently undergoes oxidation, facilitated by the presence of iodine or DMSO, to restore the double bond and yield the final (Z)-beta-iodo-beta-methylthio olefin product. The regeneration of iodine from hydrogen iodide via oxidation by DMSO completes the catalytic cycle, ensuring efficient utilization of the iodine source.
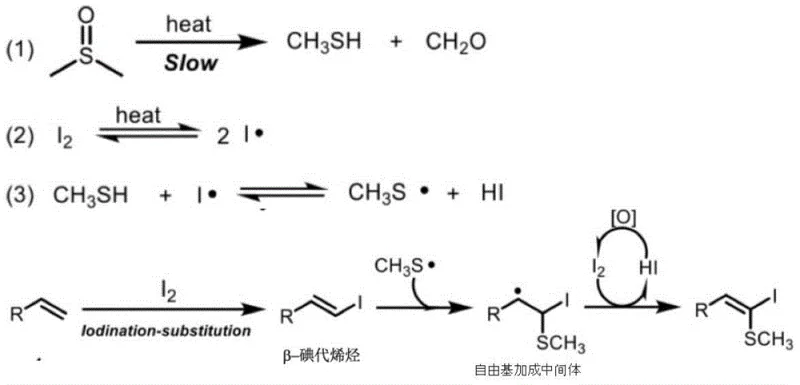
The control of stereochemistry and impurity formation is intrinsically linked to this radical mechanism. The preference for the (Z)-isomer suggests a specific transition state geometry during the radical addition and subsequent elimination steps, which is influenced by the steric bulk of the substituents on the alkene. Experimental evidence from radical inhibition studies using TEMPO and BHT confirms the involvement of free radical species, as the addition of these inhibitors significantly suppresses product formation. This mechanistic insight allows chemists to fine-tune reaction parameters such as temperature and concentration to minimize side reactions like poly-iodination or over-oxidation. For quality control purposes, understanding that the reaction generates formaldehyde as a byproduct is essential for designing appropriate workup procedures to ensure the final product meets strict residual solvent and impurity guidelines. The absence of metal catalysts means that the impurity profile is dominated by organic byproducts rather than heavy metals, simplifying the analytical characterization and release testing protocols required for GMP manufacturing environments.
How to Synthesize (Z)-beta-iodo-beta-methylthio olefin Efficiently
The practical implementation of this synthesis route is designed for simplicity and reproducibility, making it accessible for both laboratory-scale optimization and pilot plant operations. The standard protocol involves charging a reaction vessel with dimethyl sulfoxide, the terminal alkene substrate, and elemental iodine in a specific molar ratio optimized for maximum conversion. The mixture is then heated to a temperature range of 110-130°C, typically around 120°C, and stirred for a duration of 3 to 5 hours. Monitoring the reaction progress can be achieved through standard analytical techniques such as GC-MS or TLC, with the disappearance of the starting alkene indicating completion. Upon cooling, the reaction mixture is subjected to a straightforward aqueous workup involving extraction with ethyl acetate and washing with saturated brine and sodium thiosulfate to remove excess iodine. The organic layer is dried over anhydrous sodium sulfate and concentrated under reduced pressure, yielding a crude product that can be purified by flash column chromatography using petroleum ether and ethyl acetate as the eluent system.
- Mix terminal alkene substrate with dimethyl sulfoxide (DMSO) serving as both solvent and sulfur source in a reaction vessel.
- Add elemental iodine (I2) in a stoichiometric ratio of approximately 1.2 equivalents relative to the alkene substrate.
- Heat the mixture to 120°C under atmospheric conditions for 4 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis methodology offers profound advantages that extend beyond mere technical feasibility, directly impacting the bottom line and supply chain resilience. The elimination of expensive and hazardous organometallic reagents translates into a substantial reduction in raw material costs, as the process relies on commodity chemicals like DMSO and iodine which are produced on a massive global scale with stable pricing structures. This shift away from specialty reagents mitigates the risk of supply disruptions caused by the limited availability of niche catalysts or metal precursors. Furthermore, the simplified one-pot nature of the reaction reduces the demand for specialized equipment capable of handling air-sensitive or pyrophoric materials, allowing for the utilization of standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities. This compatibility with existing infrastructure accelerates the technology transfer process and lowers the capital expenditure required for implementation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the synthetic route. By removing the need for pre-synthesis of alkynyl sulfides and avoiding the use of precious metal catalysts or stoichiometric organometallic reagents, the overall cost of goods sold is significantly lowered. The removal of metal catalysts also eliminates the downstream costs associated with metal scavenging and validation testing for heavy metal residues, which are resource-intensive steps in pharmaceutical manufacturing. Additionally, the high atom economy of using DMSO as both solvent and reagent minimizes waste disposal costs, contributing to a more sustainable and economically viable production model that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced by the reliance on widely available starting materials. Terminal alkenes and elemental iodine are produced by numerous suppliers globally, reducing the risk of single-source dependency that often plagues processes requiring specialized ligands or custom-synthesized intermediates. The robustness of the reaction conditions, which do not require strict anhydrous or anaerobic environments, further reduces the logistical complexity of raw material storage and handling. This resilience ensures consistent production schedules and reliable delivery timelines, which are critical metrics for procurement managers responsible for maintaining continuous manufacturing operations and meeting customer demand without interruption.
- Scalability and Environmental Compliance: The scalability of this process is supported by its inherent safety profile and operational simplicity. The absence of exothermic hazards associated with organometallic additions and the use of high-boiling solvents like DMSO allow for safe scale-up to multi-ton quantities without significant engineering modifications. From an environmental compliance standpoint, the process generates less hazardous waste compared to traditional methods involving tin or aluminum reagents. The byproducts are primarily organic and can be managed through standard waste treatment protocols, facilitating easier permitting and regulatory approval. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for companies looking to reduce their carbon footprint and improve their sustainability ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing portfolios. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the fit of this technology within specific project constraints and regulatory frameworks.
Q: What are the primary advantages of this synthesis method over traditional alkyne functionalization?
A: Unlike traditional methods requiring multi-step preparation of alkynyl sulfides and expensive metal reagents like TMS-I or Grignard reagents, this patent describes a direct one-pot reaction using simple terminal alkenes, DMSO, and iodine, eliminating the need for transition metal catalysts.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under atmospheric pressure without sensitive metal-organic compounds, uses common solvents like DMSO, and achieves high yields (up to 86%) with simple workup procedures, making it highly suitable for commercial scale-up.
Q: What is the stereochemical outcome of the reaction?
A: The reaction selectively produces the (Z)-isomer of the beta-iodo-beta-methylthio olefin, which is critical for downstream coupling reactions in medicinal chemistry where geometric purity impacts biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-beta-iodo-beta-methylthio olefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN111620798A and are committed to leveraging this innovation to serve our global clientele. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this halogenation-thioetherification process, with rigorous QC labs dedicated to maintaining stringent purity specifications for every batch produced. We understand that in the competitive landscape of pharmaceutical and fine chemical intermediates, consistency and quality are non-negotiable, and our quality management systems are designed to deliver exactly that.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this catalyst-free method or need specific COA data to verify the quality of our pilot batches, we are ready to provide comprehensive support. Our team can also offer route feasibility assessments to determine the applicability of this chemistry to your proprietary substrates, ensuring that you can capitalize on the benefits of high-purity organic intermediates with confidence. Contact us today to explore how we can collaborate to drive innovation and efficiency in your supply chain.
