Industrial Preparation of Losartan 5-Carboxylate: A Scalable Route for High-Purity API Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active metabolites, and the preparation of Losartan 5-carboxylate (EXP-3174) stands as a prime example of process optimization. As detailed in Chinese Patent CN1931857A, a novel methodology has been established that transforms the production of this critical antihypertensive intermediate from a laboratory-scale curiosity into a viable industrial process. This patent discloses a direct oxidation strategy utilizing permanganate salts in polar solvent systems, effectively bypassing the severe bottlenecks associated with earlier techniques. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a transition from chromatography-dependent purification to straightforward crystallization-based isolation. The ability to produce Losartan 5-carboxylate with purity exceeding 98% using economical reagents like potassium permanganate underscores a significant advancement in cost-effective API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN1931857A, the synthesis of Losartan 5-carboxylate was plagued by methodologies that were inherently unsuitable for commercial scale-up. The background art highlights two primary existing methods, both of which rely heavily on active manganese dioxide as the oxidizing agent. The first method, referenced in US Patent 5,138,069, involves reacting Losartan with active manganese dioxide in chloroform at room temperature. While chemically feasible on a gram scale, this approach necessitates column chromatography for purification, a technique that is prohibitively expensive and slow for multi-kilogram production. Similarly, the second method utilizes microwave irradiation with manganese dioxide in water, followed by preparative chromatography. These chromatographic steps introduce massive inefficiencies, including high solvent consumption, significant product loss on the silica column, and extended processing times. Furthermore, the reliance on specialized equipment like microwave reactors and the generation of large volumes of hazardous organic waste render these conventional routes economically unviable for a reliable pharmaceutical intermediates supplier aiming for high-volume output.
The Novel Approach
In stark contrast, the novel approach presented in the patent leverages the oxidative power of permanganate salts—both inorganic (like potassium permanganate) and organic (like tetrabutylammonium permanganate)—within polar solvent environments. This method operates effectively at moderate temperatures ranging from 5°C to 80°C, utilizing common solvents such as dimethylformamide (DMF), pyridine, acetonitrile, or acetone, often in mixture with water. The true breakthrough lies in the workup procedure: instead of tedious chromatography, the reaction byproduct (manganese dioxide) is removed via simple filtration. The product is then isolated through acidification and crystallization, specifically recrystallization from isopropanol. This shift from separation science to unit operations like filtration and crystallization drastically simplifies the process flow. By enabling the use of various salt forms of the starting material, including potassium and calcium salts, the process offers remarkable flexibility in raw material selection, directly contributing to cost reduction in API manufacturing.
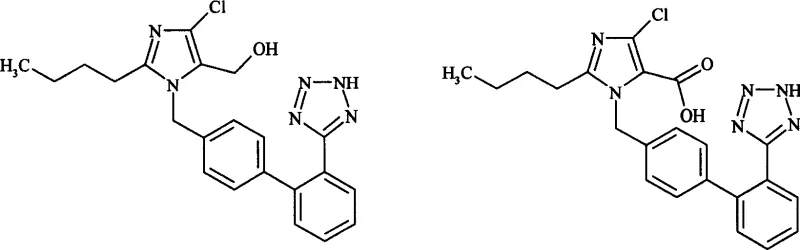
Mechanistic Insights into Permanganate-Catalyzed Oxidation
The core chemical transformation involves the oxidation of the primary hydroxymethyl group (-CH2OH) on the imidazole ring of Losartan to a carboxylic acid group (-COOH). In the patented process, this is achieved through the strong oxidizing capability of the permanganate ion (MnO4-). When conducted in a biphasic or polar medium containing a phase transfer catalyst such as tetrabutylammonium chloride, the permanganate anion is effectively transported into the organic phase where the Losartan substrate resides. This facilitates a homogeneous or pseudo-homogeneous reaction environment, ensuring efficient contact between the oxidant and the substrate. The reaction proceeds through the formation of intermediate aldehyde species which are rapidly further oxidized to the carboxylate under the reaction conditions. The use of polar aprotic solvents like DMF or pyridine stabilizes the transition states and solubilizes the ionic intermediates, allowing the reaction to proceed smoothly at temperatures as low as 20°C to 50°C, thereby minimizing thermal degradation of the sensitive biphenyl-tetrazole scaffold.
Impurity control is intrinsically built into this mechanistic pathway through the specific quenching and isolation steps. A critical aspect of the mechanism is the management of excess oxidant. The patent describes the addition of aqueous formaldehyde post-reaction, which serves to reduce any remaining purple permanganate to soluble or filterable manganese species, visually indicated by the disappearance of the red color. This step prevents over-oxidation or side reactions during the workup. Subsequently, the manganese dioxide precipitate formed during the reduction is filtered off, physically separating the bulk of inorganic impurities. The final acidification step to pH 2-3 protonates the carboxylate anion, causing the neutral Losartan 5-carboxylate to precipitate out of the aqueous-organic mixture. This pH-controlled crystallization acts as a powerful purification step, excluding non-acidic organic impurities and residual salts, resulting in the reported high purity of >98% without the need for chromatographic intervention.
How to Synthesize Losartan 5-Carboxylate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this chemistry from the bench to the pilot plant. The process begins with the preparation of the oxidizing solution, where potassium permanganate is dispersed in water and mixed with a polar organic solvent like pyridine or DMF, often aided by a phase transfer catalyst to enhance reactivity. The Losartan starting material, which can be introduced as the free acid or various salt forms, is added portion-wise to maintain thermal control, keeping the reaction temperature within the optimal 5°C to 80°C window. Following the completion of the oxidation, indicated by the persistence of the oxidant color, the mixture is treated with formaldehyde to quench excess permanganate. The detailed standardized synthesis steps for this process are provided below.
- Prepare the oxidation system by dissolving potassium permanganate in a mixture of water and a polar organic solvent such as pyridine or DMF, optionally adding a phase transfer catalyst like tetrabutylammonium chloride.
- Add Losartan or its salt (e.g., potassium or calcium salt) to the reaction mixture at controlled temperatures between 5°C and 80°C, maintaining stirring to ensure homogeneous oxidation.
- Quench excess oxidant with aqueous formaldehyde, filter off manganese dioxide precipitate, concentrate the filtrate, and acidify to pH 2-3 to crystallize the pure 5-carboxylate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this permanganate-based route offers transformative advantages over legacy methods. The most significant impact is the complete elimination of column chromatography, which is traditionally a major cost driver and bottleneck in fine chemical manufacturing. By replacing chromatography with filtration and crystallization, the process drastically reduces solvent consumption and waste disposal costs, leading to substantial cost savings in the overall production budget. Additionally, the cycle time for each batch is significantly shortened, as filtration and crystallization are much faster unit operations than running large columns, thereby enhancing throughput and reducing lead time for high-purity pharmaceutical intermediates. The use of commodity chemicals like potassium permanganate and common solvents ensures a stable and secure supply chain, mitigating risks associated with specialized reagent availability.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the downstream processing. Traditional methods require expensive silica gel and vast quantities of elution solvents, which must then be recovered or disposed of at high cost. In contrast, the novel method generates manganese dioxide sludge which is easily filtered, and the product is crystallized directly. This removal of complex purification steps lowers the operational expenditure (OpEx) significantly. Furthermore, the ability to use various salt forms of the starting material allows manufacturers to source the most cost-effective precursor available on the market at any given time, providing flexibility to capitalize on raw material price fluctuations and achieve optimized manufacturing costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction conditions. The process operates at mild temperatures and uses widely available industrial solvents like DMF, pyridine, and acetone, which are produced globally in massive quantities. This reduces the risk of supply disruptions caused by the scarcity of exotic reagents. Moreover, the tolerance of the process to different starting material salts (sodium, potassium, calcium) means that procurement teams are not locked into a single supplier specification. This flexibility ensures continuous production capability even if specific salt grades face temporary shortages, thereby guaranteeing consistent delivery schedules for downstream API manufacturers and securing the supply of this critical hypertension treatment intermediate.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is superior because it aligns with green chemistry principles by reducing solvent intensity. The absence of chromatographic solvents like chloroform or large volumes of ethyl acetate/hexane mixtures simplifies waste stream management and reduces the facility's environmental footprint. Scalability is inherently easier when the unit operations are filtration and crystallization, as these are standard in multi-purpose chemical plants. The process avoids the use of heavy metal catalysts that require stringent removal protocols, simplifying the quality control burden. This ease of scale-up from 100 kgs to 100 MT annual commercial production ensures that the technology can meet global demand without requiring bespoke reactor designs, facilitating rapid capacity expansion to match market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Losartan 5-carboxylate based on the patented technology. These insights are derived directly from the experimental data and claims within CN1931857A, providing clarity on purity, scalability, and process robustness. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their supply chain.
Q: Why is the permanganate oxidation method superior to active manganese dioxide oxidation for Losartan 5-carboxylate?
A: The permanganate method described in CN1931857A eliminates the need for column chromatography, which is required in traditional MnO2 methods. This allows for simple filtration and crystallization, making the process significantly more suitable for large-scale industrial production and reducing solvent waste.
Q: What represents the critical purity specification for this intermediate?
A: The patent data indicates that the synthesized 5-carboxylate losartan achieves a purity greater than 98% through recrystallization from isopropanol, meeting stringent requirements for downstream pharmaceutical applications without complex purification steps.
Q: Can this synthesis be adapted for different salt forms of the starting material?
A: Yes, the process is robust and accommodates various forms of Losartan, including the free base, ammonium salts, alkali metal salts (sodium, potassium), and alkaline earth metal salts (calcium, magnesium), providing flexibility in raw material sourcing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Losartan 5-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of Losartan 5-carboxylate (EXP-3174) in the global cardiovascular drug market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of this critical intermediate. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the >98% purity benchmark essential for downstream API synthesis. We combine the efficiency of the permanganate oxidation route with our own process optimization expertise to deliver a product that balances high quality with competitive pricing.
We invite pharmaceutical partners to collaborate with us to secure their supply chains for antihypertensive medications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can lower your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and efficient chemical partner in the industry.
