Advanced Catalytic Oxidation for High-Purity Alpha-Tricarbonyl Sulfur Ylide Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways to access complex molecular scaffolds. Patent CN110872249A introduces a groundbreaking synthesis method for α,α,β-tricarbonyl sulfur ylide compounds, which serve as critical precursors for the construction of tetraketone derivatives such as DPBT. Historically, the synthesis of such highly functionalized carbonyl systems relied heavily on hazardous reagents like mercury salts and concentrated nitric acid, posing severe safety and disposal challenges for large-scale manufacturing. This new methodology leverages a transition-metal catalyzed oxidation strategy, utilizing molecular oxygen as the terminal oxidant to transform sulfur oxide ylides into the desired tricarbonyl structures with exceptional efficiency. By shifting away from stoichiometric toxic oxidants to a catalytic aerobic system, this technology not only enhances the safety profile of the production process but also aligns with modern green chemistry principles, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains.
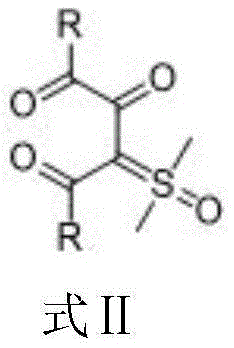
Furthermore, the structural versatility of this approach allows for the incorporation of diverse substituents, including electron-withdrawing and electron-donating groups on aryl rings, as well as heterocyclic and alkyl moieties. This broad substrate scope is pivotal for medicinal chemists who require rapid access to analog libraries for structure-activity relationship (SAR) studies. The ability to tolerate various functional groups without compromising the integrity of the sensitive ylide functionality underscores the robustness of the catalytic system. As a result, this patent represents a significant technological leap forward, offering a streamlined route to high-value intermediates that are essential for the development of next-generation bioactive molecules and advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating tetraketone compounds and their precursors have long been plagued by significant operational and environmental drawbacks. The reliance on mercury-based reagents introduces severe toxicity concerns, requiring specialized containment infrastructure and rigorous waste management protocols to prevent heavy metal contamination of the final product and the environment. Additionally, the use of strong mineral acids like nitric acid often leads to poor selectivity, resulting in complex mixtures of by-products that necessitate extensive and costly purification steps. These harsh conditions can also degrade sensitive functional groups, limiting the scope of substrates that can be successfully employed. From a supply chain perspective, the regulatory burden associated with handling and disposing of mercury waste adds substantial overhead costs and extends lead times, making conventional methods increasingly unsustainable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN110872249A utilizes a mild and efficient catalytic system driven by copper and silver salts under an oxygen atmosphere. This approach eliminates the need for stoichiometric toxic oxidants, replacing them with molecular oxygen, which is abundant, inexpensive, and generates water as the only by-product. The reaction proceeds under relatively moderate temperatures, typically between 70°C and 110°C, which reduces energy consumption compared to high-temperature pyrolytic methods. The use of 1,4-dioxane as a solvent provides a stable medium for the catalytic cycle, ensuring consistent reaction kinetics. This shift to a catalytic aerobic oxidation not only simplifies the work-up procedure but also dramatically improves the overall atom economy of the process. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing by removing the expenses associated with hazardous waste disposal and expensive stoichiometric reagents.
Mechanistic Insights into Copper-Catalyzed Aerobic Oxidation
The core of this innovation lies in the synergistic interaction between the copper and silver catalysts which facilitates the activation of molecular oxygen and the subsequent oxidation of the sulfur ylide carbon center. The proposed mechanism involves the coordination of the sulfur ylide substrate to the copper center, followed by the insertion of oxygen to form a peroxo-intermediate. The silver salt likely acts as a co-catalyst or Lewis acid to stabilize the transition state and promote the rearrangement of the oxygenated species into the tricarbonyl framework. This dual-catalyst system ensures high turnover numbers and minimizes catalyst deactivation, which is a common issue in single-metal catalyzed oxidations. The precise control over the oxidation state prevents over-oxidation or decomposition of the sensitive ylide moiety, thereby maintaining high product fidelity. Understanding this mechanistic pathway is crucial for R&D directors aiming to further optimize reaction conditions or adapt the protocol for continuous flow processing.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The high selectivity of the copper-silver catalytic system minimizes the formation of side products such as sulfones or desulfurized ketones, which are common impurities in non-catalytic oxidations. The mild reaction conditions preserve the integrity of other functional groups present on the R substituent, such as halogens, cyano, or nitro groups, preventing unwanted side reactions like hydrolysis or reduction. This inherent selectivity simplifies the downstream purification process, often allowing for high-purity isolation via standard silica gel chromatography without the need for preparative HPLC. For quality control teams, this means a cleaner impurity profile and easier validation of the manufacturing process, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications.
How to Synthesize Alpha-Tricarbonyl Sulfur Ylide Efficiently
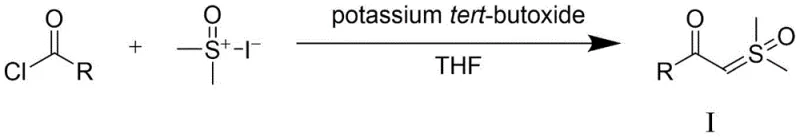
The practical implementation of this synthesis involves a straightforward two-stage process beginning with the preparation of the sulfur oxide ylide precursor followed by the catalytic oxidation step. The initial preparation of the starting material (Formula I) can be achieved using established literature procedures involving the reaction of acid chlorides with trimethylsulfoxonium iodide in the presence of a strong base like potassium tert-butoxide. Once the precursor is secured, the key oxidative transformation is conducted in a Schlenk tube or reactor equipped with an oxygen inlet. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification parameters, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Preparation of Sulfur Oxide Ylide (Formula I) from acid chlorides and trimethylsulfoxonium iodide using potassium tert-butoxide.
- Catalytic Oxidation Reaction: React Formula I with Cu(OAc)2 and CF3COOAg in 1,4-dioxane under oxygen atmosphere at 90°C.
- Purification via silica gel column chromatography using petroleum ether and ethyl acetate mixtures to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis route offers transformative benefits for supply chain stability and operational expenditure. By transitioning to a catalytic process that utilizes air-stable reagents and molecular oxygen, manufacturers can significantly reduce their dependency on regulated and hazardous chemicals. This shift mitigates the risk of supply disruptions caused by environmental regulations on mercury or nitric acid usage. Furthermore, the simplified work-up procedure, which involves basic silica gel chromatography, reduces the consumption of specialized purification resins and solvents. These operational efficiencies contribute to substantial cost savings and enhanced supply chain reliability, ensuring consistent delivery of critical intermediates to downstream clients without the volatility associated with traditional toxic synthesis methods.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic stoichiometric oxidants like mercury salts directly lowers the raw material costs per kilogram of product. Additionally, the removal of heavy metal waste treatment requirements drastically reduces the environmental compliance costs and disposal fees associated with the manufacturing process. The high yields reported, reaching up to 82%, further enhance the economic viability by maximizing the output from each batch of starting materials. This combination of lower input costs and higher output efficiency results in a significantly optimized cost structure for the production of these valuable intermediates.
- Enhanced Supply Chain Reliability: The starting materials, specifically substituted acid chlorides and dimethyl sulfoxide derivatives, are commodity chemicals that are readily available from multiple global suppliers. This abundance ensures that the supply chain is not bottlenecked by the availability of niche or regulated reagents. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in temperature or pressure, leading to more consistent batch-to-batch quality. For supply chain heads, this reliability translates to reduced lead time for high-purity pharmaceutical intermediates and a lower risk of production delays due to raw material shortages.
- Scalability and Environmental Compliance: The use of molecular oxygen as the oxidant makes this process inherently scalable, as oxygen can be supplied continuously without the accumulation of hazardous waste. The absence of heavy metals simplifies the regulatory approval process for new drug applications, as residual metal limits are easier to meet. The process generates minimal hazardous waste, aligning with increasingly strict global environmental standards and corporate sustainability goals. This environmental compatibility facilitates easier permitting for new manufacturing facilities and supports the long-term sustainability of the production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and technical stakeholders.
Q: What catalysts are utilized in this novel synthesis method?
A: The process employs a dual-catalyst system comprising anhydrous copper acetate (Cu(OAc)2) and silver trifluoroacetate (CF3COOAg) to facilitate the oxidative transformation efficiently.
Q: How does this method improve upon traditional tetraketone precursor synthesis?
A: Unlike conventional routes that rely on toxic mercury salts or nitric acid, this method utilizes molecular oxygen as a green oxidant, significantly reducing environmental hazards and waste treatment costs.
Q: What yields can be expected for this reaction?
A: Experimental data indicates that isolated yields can reach up to 82%, demonstrating high efficiency across a broad range of aryl and heterocyclic substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Tricarbonyl Sulfur Ylide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and green synthetic routes in the modern chemical landscape. Our team of expert chemists has extensively evaluated the catalytic oxidation technology described in CN110872249A and confirmed its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Alpha-Tricarbonyl Sulfur Ylide we produce, meeting the exacting demands of the global pharmaceutical market.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective supply of these critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
