Advanced Cobalt-Catalyzed Synthesis of Imidazolidinone Derivatives for Commercial Scale Production
Advanced Cobalt-Catalyzed Synthesis of Imidazolidinone Derivatives for Commercial Scale Production
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and cost-effective pathways to access high-value nitrogen-containing heterocycles. Patent CN113087667B introduces a groundbreaking synthetic methodology for imidazolidinone derivatives that addresses critical pain points in modern organic synthesis. By leveraging a recyclable cobalt catalyst and utilizing renewable polyol resources as key starting materials, this technology offers a robust alternative to traditional precious metal-catalyzed processes. For R&D directors and procurement managers alike, this innovation represents a significant opportunity to optimize supply chains while adhering to green chemistry principles. The ability to convert biomass-derived polyols directly into complex pharmacophores not only reduces dependency on petrochemical feedstocks but also streamlines the manufacturing workflow, making it an ideal candidate for reliable imidazolidinone supplier partnerships focused on long-term sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the imidazolidinone core has relied on methodologies that pose significant challenges for large-scale manufacturing. Traditional routes often involve the cyclization of diamides, the condensation of amino esters with cyanamides, or multi-component reactions catalyzed by expensive palladium complexes. These conventional approaches frequently necessitate harsh reaction conditions, including elevated temperatures and high pressures, which demand specialized equipment and increase operational risks. Moreover, the reliance on stoichiometric oxidants or precious metal catalysts generates substantial amounts of heavy metal waste and toxic byproducts, complicating downstream purification and environmental compliance. The cumulative effect of these factors is a manufacturing process characterized by low atom economy, high production costs, and extended lead times, which severely impacts the cost reduction in pharmaceutical intermediate manufacturing strategies for global enterprises.
The Novel Approach
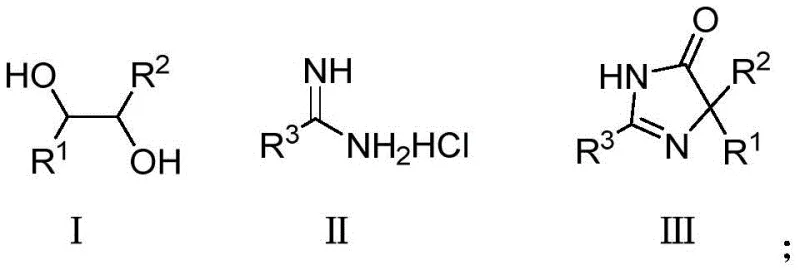
In stark contrast, the novel cobalt-catalyzed oxidative cyclization described in the patent data offers a streamlined, one-pot solution that dramatically simplifies the synthetic landscape. This method utilizes readily available polyols and amidines as building blocks, reacting them under mild conditions with air serving as the sole oxidant. The use of supported nano-cobalt catalysts ensures high activity while allowing for easy separation and reuse, effectively eliminating the contamination issues associated with homogeneous catalysis. As illustrated in the general reaction scheme below, the transformation proceeds with excellent functional group tolerance, accommodating diverse substituents on both the polyol and amidine components.
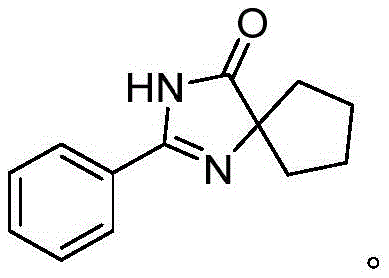
Furthermore, the versatility of this approach is evidenced by the successful synthesis of various derivatives, such as the cyclohexane-fused imidazolidinone shown in the following example, which demonstrates the method's capability to handle cyclic diols efficiently. This flexibility allows chemists to access a wide array of structural analogues without modifying the core protocol, thereby accelerating the drug discovery process and enhancing the commercial viability of the final active ingredients.
Mechanistic Insights into Cobalt-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the cobalt catalyst, the polyol substrate, and molecular oxygen. Initially, the nano-cobalt species activates the hydroxyl groups of the polyol, facilitating a selective dehydrogenation process that generates a reactive carbonyl intermediate in situ. This transient species then undergoes nucleophilic attack by the amidine nitrogen, followed by a cyclization event that constructs the five-membered imidazolidinone ring. The presence of a base, such as cesium carbonate or potassium hydroxide, is crucial for neutralizing the acidic byproducts and driving the equilibrium towards the desired product. Crucially, the use of air as the terminal oxidant regenerates the active cobalt species, closing the catalytic cycle and ensuring that the reaction proceeds with high turnover numbers. This mechanism not only explains the high yields observed across various substrates but also highlights the inherent safety of the process, as it avoids the accumulation of unstable peroxides often associated with other oxidation methods.
From an impurity control perspective, this catalytic system exhibits remarkable selectivity, minimizing the formation of over-oxidized byproducts or polymerization residues that typically plague polyol transformations. The heterogeneous nature of the supported catalyst prevents leaching of metal ions into the product stream, which is a critical parameter for meeting the stringent purity specifications required in API synthesis. Additionally, the mild reaction temperatures (ranging from 25°C to 130°C) prevent thermal degradation of sensitive functional groups, ensuring that the final crude product possesses a clean profile that simplifies subsequent purification steps. For quality assurance teams, this means reduced variability between batches and a more predictable impurity profile, which is essential for regulatory filings and maintaining consistent product quality in high-purity imidazolidinone derivatives manufacturing.
How to Synthesize Imidazolidinone Derivatives Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires careful attention to catalyst loading and reaction parameters to maximize efficiency. The standard protocol involves charging a reactor with the polyol and amidine substrates in a suitable solvent like pyridine or tert-amyl alcohol, followed by the addition of the base and the supported cobalt catalyst. The mixture is then heated under an air atmosphere, allowing the oxidative cyclization to proceed to completion. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and workup protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized steps outlined below.
- Mix polyol compounds and amidine compounds in a suitable solvent such as pyridine or tert-amyl alcohol with a base.
- Add a supported nano-cobalt catalyst (e.g., activated carbon or alumina supported) to the reaction mixture.
- Heat the mixture to 25°C-130°C under air atmosphere for 1-24 hours, then purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology translates into tangible strategic benefits that extend beyond simple unit price reductions. By shifting away from precious metal catalysts and hazardous oxidants, manufacturers can significantly lower their exposure to volatile commodity markets and reduce the costs associated with waste disposal and environmental remediation. The use of air as an oxidant eliminates the need for purchasing and storing dangerous chemical oxidants, thereby enhancing facility safety and reducing insurance premiums. Furthermore, the simplicity of the workup procedure, which often involves merely filtering off the solid catalyst and evaporating the solvent, drastically reduces processing time and energy consumption. These operational efficiencies contribute to a more resilient supply chain capable of responding quickly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or rhodium catalysts with earth-abundant cobalt represents a fundamental shift in cost structure for heterocycle synthesis. Since the cobalt catalyst is supported on inexpensive materials like activated carbon or alumina and can be recycled multiple times without significant loss of activity, the effective cost per kilogram of product is substantially decreased. Additionally, the elimination of stoichiometric oxidants removes a major line item from the bill of materials, while the simplified purification process reduces the consumption of chromatography media and solvents. This holistic reduction in material and processing costs allows for a much more competitive pricing strategy in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on renewable polyols and simple amidines as starting materials ensures a stable and secure supply of raw materials, independent of complex petrochemical supply chains. Polyols can be sourced from biomass, aligning with corporate sustainability goals and mitigating risks associated with fossil fuel price fluctuations. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failures or utility shortages, as the process does not require high-pressure reactors or cryogenic cooling. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The mild operating conditions and the use of benign reagents make this process inherently scalable from gram-scale discovery to multi-ton commercial production. The absence of toxic heavy metal waste and the use of air as a green oxidant significantly simplify the environmental permitting process and reduce the burden on wastewater treatment facilities. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the company as a responsible and sustainable partner in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic methodology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional palladium methods?
A: This method utilizes earth-abundant cobalt instead of expensive precious metals like palladium, significantly reducing raw material costs. Furthermore, it uses air as the terminal oxidant, eliminating the need for hazardous stoichiometric oxidants and simplifying waste treatment.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates under relatively mild temperatures (25°C to 130°C) and atmospheric pressure, avoiding the extreme high-pressure conditions often required by conventional cyclization methods, which facilitates safer and easier commercial scale-up.
Q: What types of substrates are compatible with this synthetic route?
A: The method exhibits broad substrate applicability, tolerating various functional groups including halogens, alkoxy groups, and heterocycles on the amidine component, as well as cyclic and acyclic polyols, allowing for the synthesis of diverse imidazolidinone libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazolidinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed synthesis for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this green chemistry protocol, including the safe handling of nano-catalysts and the implementation of rigorous QC labs to guarantee stringent purity specifications for every batch. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the market through superior chemistry and supply chain excellence.
