Advanced Silver-Catalyzed Synthesis of 4-(Isochromen-1-yl)isoquinoline Derivatives for Commercial Scale-Up
Introduction to Patent CN110204533B Technology
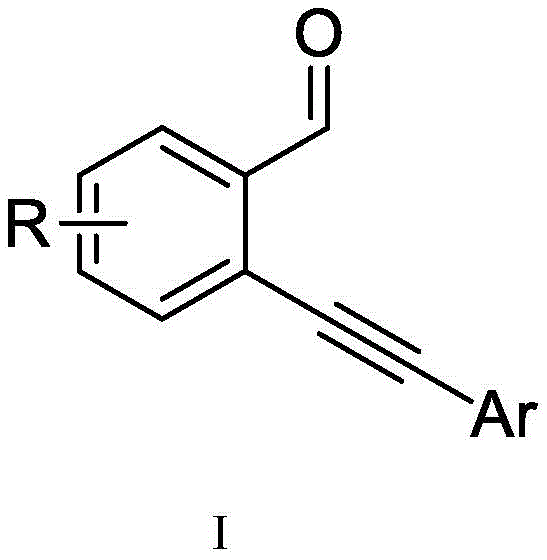
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that possess significant biological activity. Patent CN110204533B discloses a groundbreaking preparation method for 4-(isochromen-1-yl)isoquinoline derivatives, a class of compounds that merges the pharmacological potential of isochromene and isoquinoline cores. This technology represents a significant leap forward in organic synthesis, specifically addressing the challenges associated with coupling electron-deficient aromatic systems. By leveraging a monovalent silver salt catalytic system, the invention enables the efficient transformation of readily available o-alkynyl aromatic aldehydes into high-value intermediates under remarkably mild conditions.
The strategic importance of this patent lies in its ability to bypass the harsh conditions typically required for such transformations. Traditional routes often suffer from limited substrate scope and poor yields when dealing with electron-deficient nucleophiles. In contrast, this novel approach utilizes a tandem reaction sequence involving dimerization and subsequent condensation, achieving high purity and yield without the need for expensive transition metals or rigorous anhydrous environments. For R&D directors and procurement managers alike, this translates to a reliable pathway for generating diverse libraries of bioactive molecules with reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isochromene derivatives has relied heavily on metal or non-metal catalyzed reactions between o-alkynyl aromatic aldehydes and various nucleophiles such as alcohols, amines, or electron-rich aromatic hydrocarbons. While these methods have proven effective for constructing certain isochromene architectures, they exhibit severe limitations when applied to the synthesis of isoquinoline-substituted variants. The primary bottleneck is the electronic nature of the isoquinoline ring; being an electron-deficient aromatic compound, it possesses poor nucleophilic ability. Consequently, conventional electrophilic activation strategies often fail to drive the reaction to completion, resulting in low conversion rates and a narrow scope of applicable substrates. Furthermore, many existing protocols require stringent reaction conditions, including high temperatures, strong acids, or sensitive catalysts that demand inert atmospheres, thereby increasing both the cost and the safety risks associated with manufacturing.
The Novel Approach
The methodology described in CN110204533B fundamentally alters the reaction landscape by employing a monovalent silver salt catalyst to activate the alkyne moiety of the starting material. This activation facilitates a unique dimerization-cyclization cascade that generates a reactive ketone-form intermediate, which subsequently undergoes condensation with a nitrogen-hydrogen compound. This two-step continuous reaction strategy effectively circumvents the nucleophilicity issues of the isoquinoline system by pre-assembling the carbon skeleton before introducing the nitrogen source. The result is a versatile synthetic route that tolerates a wide array of functional groups, including halogens, alkyls, and alkoxy substituents, ensuring good substrate compatibility. Moreover, the reaction proceeds smoothly at room temperature in an air atmosphere, eliminating the need for costly inert gas lines and specialized pressure equipment, which significantly lowers the barrier to entry for commercial adoption.
Mechanistic Insights into Silver-Catalyzed Cyclization
The efficacy of this synthesis hinges on the precise coordination chemistry between the silver catalyst and the alkyne functionality. The mechanism initiates with the coordination of the monovalent silver cation to the triple bond of the o-alkynyl aromatic aldehyde, forming a silver-alkyne complex. This coordination activates the alkyne towards intramolecular nucleophilic attack by the carbonyl oxygen, leading to a cycloisomerization event. The resulting silver species then undergoes a self-[3+2]-cycloaddition reaction, a critical step that builds the polycyclic framework essential for the isochromene core. Following this, a series of tautomerization and cracking reactions occur, eventually yielding a hemiacetal intermediate that spontaneously eliminates to form a stable ketone-form dimer. This intricate cascade is highly selective, minimizing the formation of side products and ensuring that the majority of the starting material is channeled into the desired intermediate structure.
In the second stage of the process, the ketone-form intermediate reacts with a nitrogen source, such as ammonia or an amine, through a condensation dehydration mechanism. This step constructs the isoquinoline ring system fused to the isochromene scaffold. The use of a protic solvent like methanol in this stage facilitates the dissolution of the nitrogen source and promotes the elimination of water, driving the equilibrium towards the final product. The entire process is depicted in the reaction schemes below, highlighting the seamless transition from simple aldehydes to complex heterocyclic architectures. This mechanistic understanding allows chemists to fine-tune reaction parameters, such as catalyst loading and solvent choice, to optimize yields for specific derivatives, ensuring consistent quality across different batches.
How to Synthesize 4-(Isochromen-1-yl)isoquinoline Efficiently
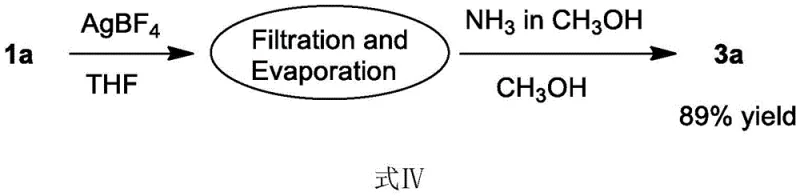
The patent outlines a streamlined protocol that combines the dimerization and condensation steps into a cohesive workflow, minimizing handling time and material loss. The process begins by dissolving the o-alkynyl aromatic aldehyde in an aprotic solvent, preferably tetrahydrofuran (THF), and adding a catalytic amount of a silver salt such as silver tetrafluoroborate (AgBF4). The mixture is stirred at room temperature for approximately 8 hours, allowing the silver-catalyzed dimerization to reach completion. Without the need for rigorous purification of the crude intermediate, the solvent is evaporated, and the residue is directly subjected to the condensation step. By dissolving the residue in methanol and adding a solution of ammonia, the reaction proceeds to form the target 4-(isochromen-1-yl)isoquinoline derivative. This telescoped approach not only simplifies the operational procedure but also enhances the overall atom economy of the synthesis.
- Dissolve the o-alkynyl aromatic aldehyde compound in an aprotic solvent such as tetrahydrofuran (THF) and add a monovalent silver salt catalyst like AgBF4.
- Stir the reaction mixture at room temperature (approx. 25°C) under air atmosphere for 6-10 hours to form the ketone-form intermediate via dimerization.
- Without isolating the intermediate, dissolve the residue in a protic solvent like methanol and react with a nitrogen source (e.g., ammonia in methanol) for 10-15 hours to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver-catalyzed methodology offers substantial benefits for supply chain stability and cost management. The reliance on inexpensive and commercially available silver salts, rather than exotic precious metal catalysts like palladium or rhodium, drastically reduces the raw material costs associated with the synthesis. Furthermore, the ability to conduct the reaction under ambient air conditions removes the necessity for expensive inert atmosphere setups, such as gloveboxes or nitrogen purging systems, which are often capital-intensive to install and maintain. This simplification of the reaction environment translates directly into lower overhead costs and reduced energy consumption, making the process highly attractive for large-scale manufacturing facilities aiming to optimize their operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of common solvents like THF and methanol significantly lower the bill of materials. Additionally, the mild reaction conditions reduce energy costs associated with heating or cooling, while the high yields minimize waste disposal fees. The process avoids the need for complex purification steps for the intermediate, further reducing solvent usage and labor time, leading to a more economical production cycle overall.
- Enhanced Supply Chain Reliability: The starting materials, o-alkynyl aromatic aldehydes, are readily accessible via standard Sonogashira coupling reactions, ensuring a stable supply of precursors. The robustness of the catalytic system against moisture and oxygen means that production is less susceptible to delays caused by equipment failure or environmental fluctuations. This reliability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing downstream partners to plan their inventory and production schedules with greater confidence.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates easy scale-up from laboratory benchtop to industrial reactor volumes without significant re-engineering. The use of less hazardous reagents and the generation of fewer toxic by-products align with green chemistry principles, simplifying regulatory compliance and waste treatment processes. This environmental friendliness not only reduces the ecological footprint but also mitigates the risk of regulatory penalties, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 4-(isochromen-1-yl)isoquinoline derivatives as described in the patent literature. These insights are derived directly from the experimental data and mechanistic explanations provided in CN110204533B, offering clarity on the feasibility and advantages of this synthetic route for potential partners and stakeholders.
Q: What are the key advantages of the silver-catalyzed method over traditional synthesis?
A: The silver-catalyzed method operates under mild conditions (room temperature, air atmosphere) without the need for inert gas protection. It utilizes cheap and easily obtainable catalysts and demonstrates excellent substrate compatibility, overcoming the limitations of traditional methods which struggle with electron-deficient isoquinoline systems.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is suitable for industrialized mass production. The reaction parameters can be adjusted by those skilled in the art, and the use of common solvents like THF and methanol facilitates scalable operations.
Q: What is the typical yield range for these derivatives?
A: The patent reports high yields for the target products. For example, in the optimized conditions using AgBF4 in THF followed by ammonia in methanol, the isolated yield for the model product reached 89%, with intermediate yields up to 93%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Isochromen-1-yl)isoquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology disclosed in CN110204533B for advancing drug discovery and development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-(isochromen-1-yl)isoquinoline derivatives meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this efficient synthesis can accelerate your pipeline. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for high-value heterocyclic intermediates.
